Latest Updates
-
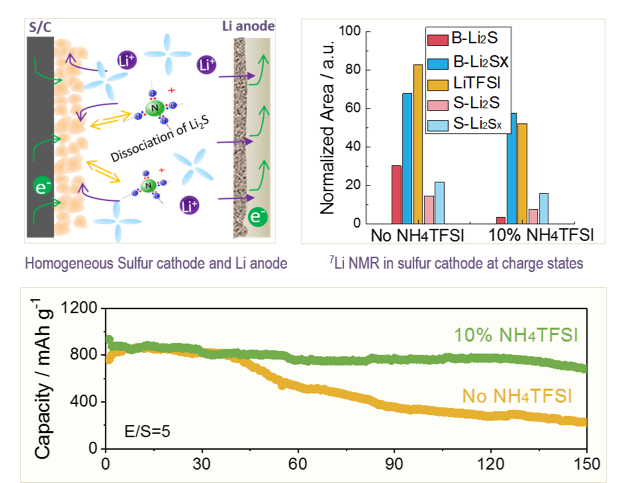
Addressing Passivation in Lithium-Sulfur Battery Under Lean Electrolyte Condition
Identification and understanding of cycle life limiting factors of Li-S batteries under lean electrolyte conditions; Identification of a NH4TFSI additive to effectively mitigate the uncontrollable passivation issue arising from accumulation of insulating Li2S. Read More
-

Nanostructured Electrolytes for Stabilizing Lithium Metal Anodes
The addition of salt to block copolymers results in the spontaneous and surprising formation of well-ordered lamellae in a sample that originally had a disordered morphology. Read More
-
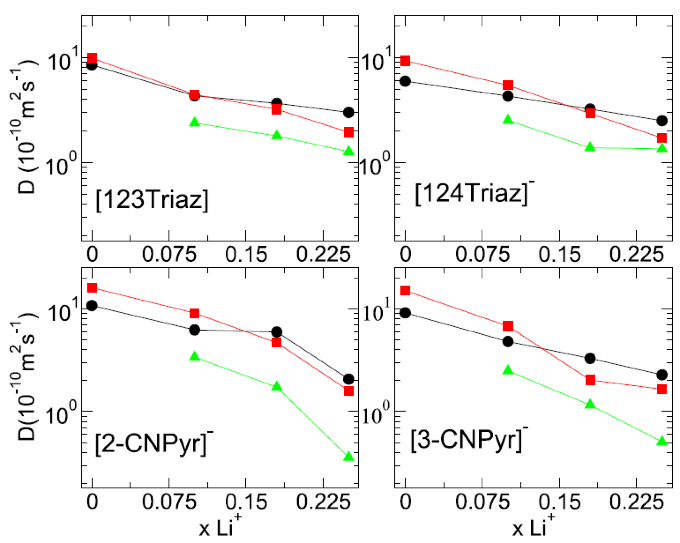
A Molecular Dynamics Study of Lithium-Containing Aprotic Heterocyclic Ionic Liquid Electrolytes
Molecular dynamics simulation was carried out on 12 aprotic heterocyclic anion ionic liquid based electrolyte systems and (methyloxymethyl)triethylphosphonium triazolide was found to have the best performance. Read More
-
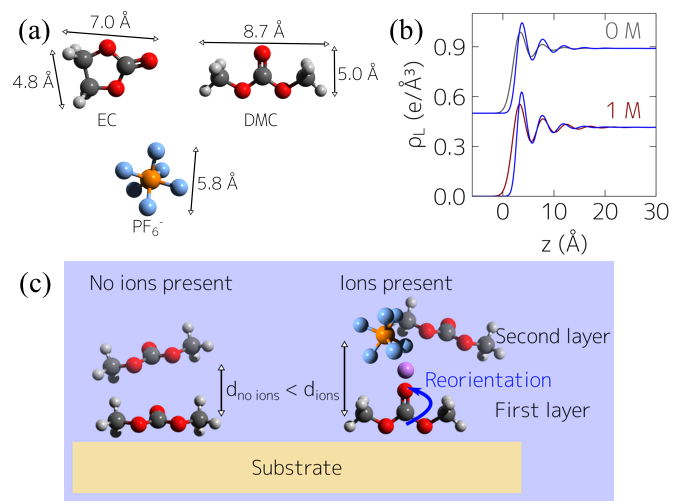
The Nanoscale Structure of the Electrolyte-metal Oxide Interface
Observation of molecular layering and insight into Li-ion salt concentration dependence of molecular orientation at metal-oxide electrolyte interfaces relevant to Li-ion batteries. Read More
-

Improved Performance through Tight Coupling of Redox Cycles of Sulfur and 2,6-Polyanthraquinone in Lithium-Sulfur Batteries
We showed that the incorporation of an all-organic redox-active polymer 2,6-polyanthraquinone (PAQ) into the cathode improves capacity retention in galvanostatic cycling and inhibits Li corrosion and S deposition. Redox reactions of this polymer are shown to be strongly coupled to S redox cycle. Read More
-
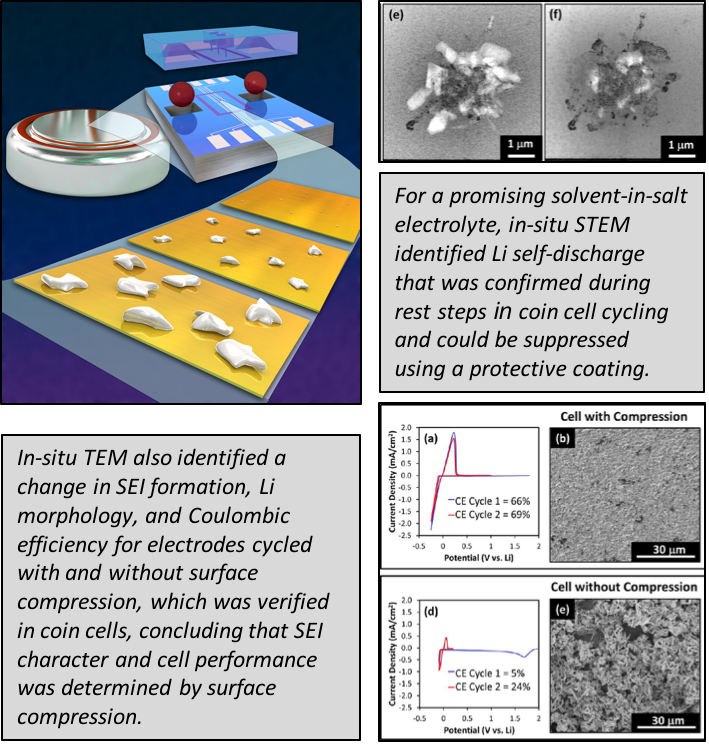
Lithium Self-Discharge and its Prevention: Direct Visualization through In-Situ Electrochemical STEM
We show that Li anode morphology and solid electrolyte interphase structure is dependent on surface compression, which affects the amount of self-discharge for an exciting solvent-in-salt electrolyte. Additionally, we show that coatings can suppress self-discharge. Read More
-

Better Batteries Hold Promise for a Sustainable Future
Better batteries for electric cars and cellphones and better batteries for industrial use that can store excess power generated by wind and solar farms and then supply it to the grid at times of peak demand are greatly needed. The ability to efficiently store energy… Read More
-
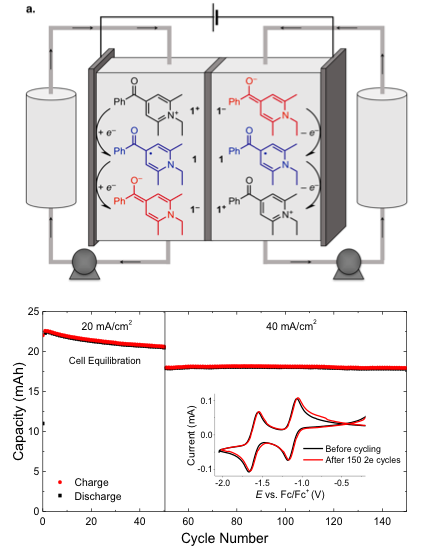
Multielectron Cycling of a Low Potential Anolyte in Alkali Metal Electrolytes for Non-Aqueous Redox Flow Batteries
High energy density organic redox flow batteries can only be realized when stable light-weight organic redox systems are developed, multi-electron redox molecules in combination with light-weight supporting electrolytes pave the way to this goal. Read More
-
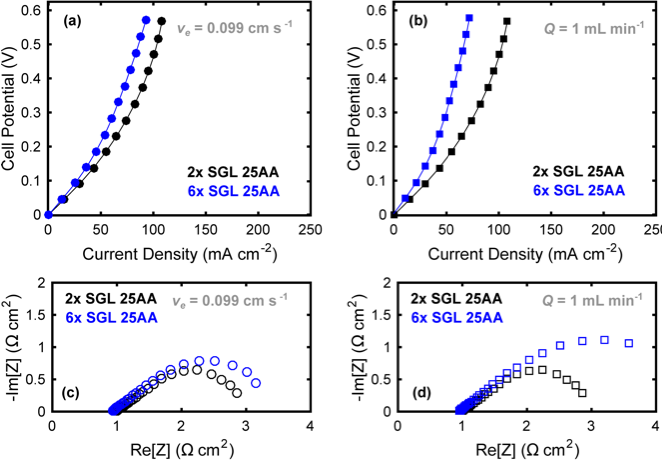
Towards Low Resistance Nonaqueous Redox Flow Batteries
Sources of resistive losses were experimentally minimized for a nonaqueous flow cell for various active species concentrations, electrolyte compositions, flow rates, separators, and electrode thicknesses. Read More
-
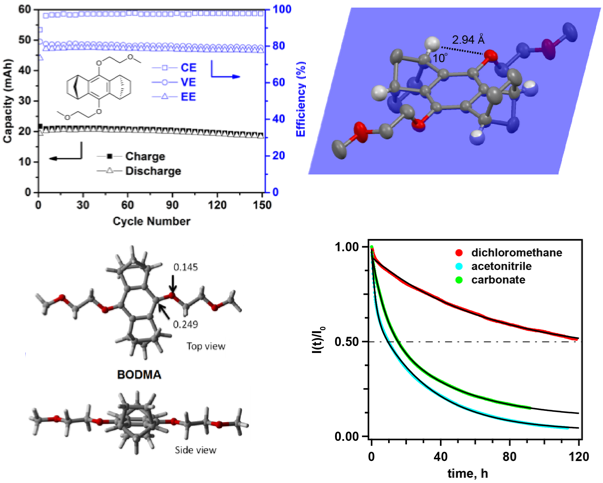
Annulated Dialkoxybenzenes as Catholyte Materials for Non-aqueous Redox Flow Batteries: Achieving High Chemical Stability Through Bicyclic Substitution
9,10-bis(2-methoxyethoxy)-1,2,3,4,5,6,7,8- octahydro-1,4:5,8-dimethanenoanthracene (BODMA) was developed for use as the catholyte in non-aqueous redox flow batteries. The bicyclic scaffolds prevent the ring-addition reaction, showing superior chemical stability in the charged state. A hybrid flow cell using this catholyte is operated for 150 charge-discharge cycles with a minimal loss of capacity. Read More
-
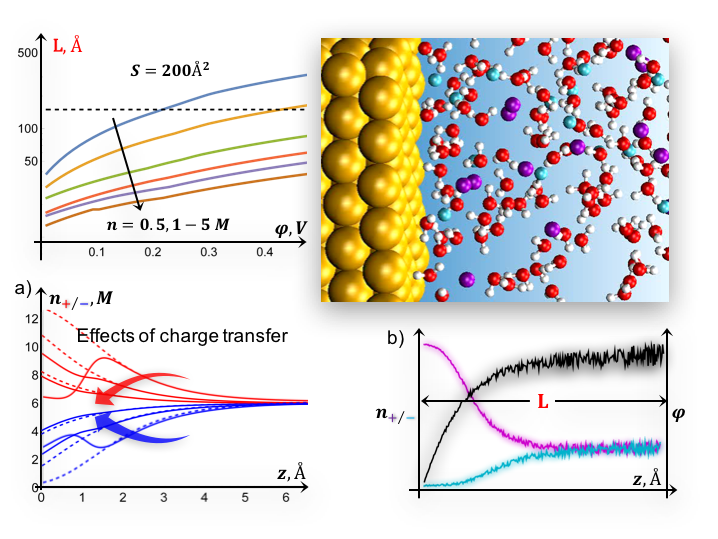
Improving Continuum Models to Define Practical Limits for Molecular Models of Electrified Interfaces
Scientific Achievement We develop a self-consistent methodology for modeling biased interfaces that combines (i) continuum theory and (ii) ab initio molecular dynamics, to explore the structure of the electric double layer under various electrochemical conditions, including effects of electron transfer and non-electrostatic interactions (e.g. Read More
-
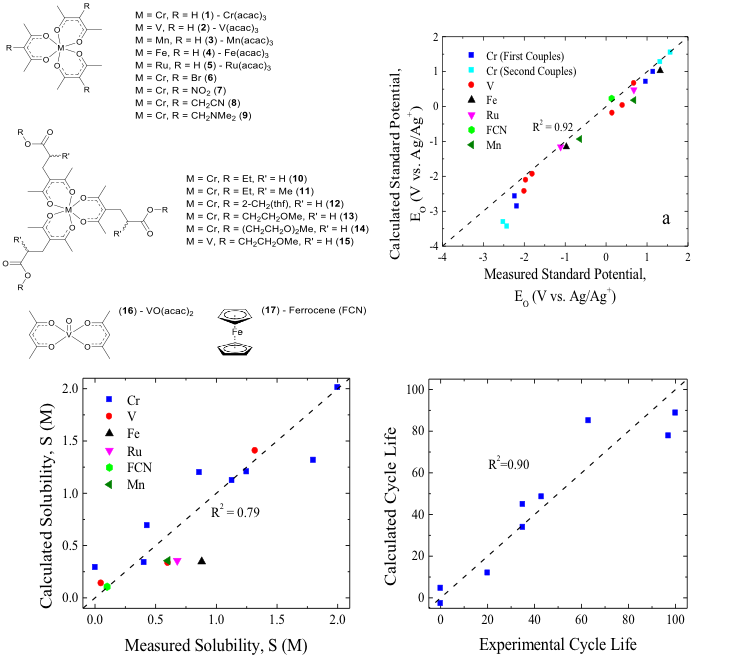
Predicting the Potentials, Solubilities and Stabilities of Metal-Acetylacetonates for Non-Aqueous Redox Flow Batteries Using Density Functional Theory Calculations
Density functional theory (DFT) was used to calculate key materials properties that were correlated with experimentally determined parameters that define the performance of redox flow battery (RFB) active materials. These include standard potentials, solubilities, and importantly stabilities. The correlations are for metal-acetylacetonate (acac) complexes, a promising class of actives, but could also be used for other materials. Read More
-
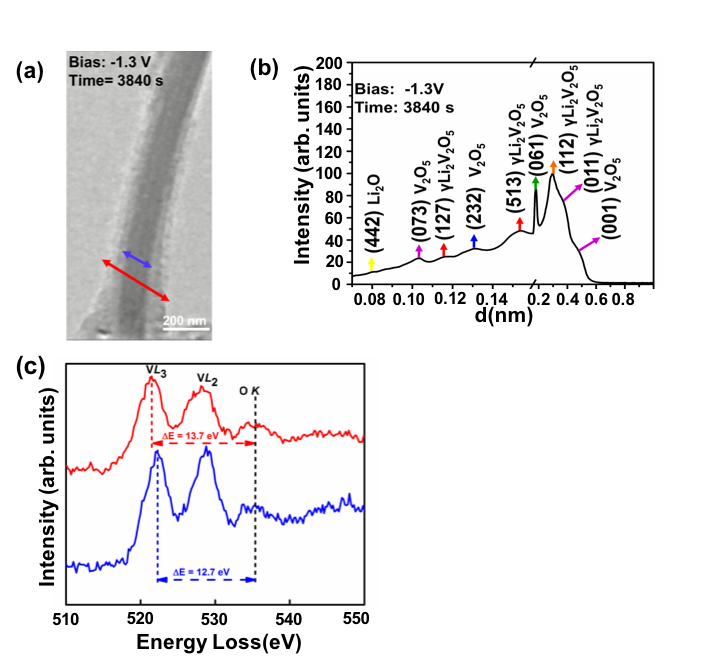
Study of Li Intercalation into α-V2O5 Nanowires using in situ Transmission Electron Microscopy
We examine the effects of Li intercalation into α-V2O5 nanowires using in situ transmission electron microscopy. Combining electron diffraction and electron energy loss spectroscopy, we conclude that the pristine V2O5 nanowires form a Li2O shell, which acts as a solid state electrolyte. Thus Li+ ions move radially into the nanowire core, and bulk of the nanowire undergoes transformation to the γ-Li2V2O5 phase at -1.3 V bias. Read More
-
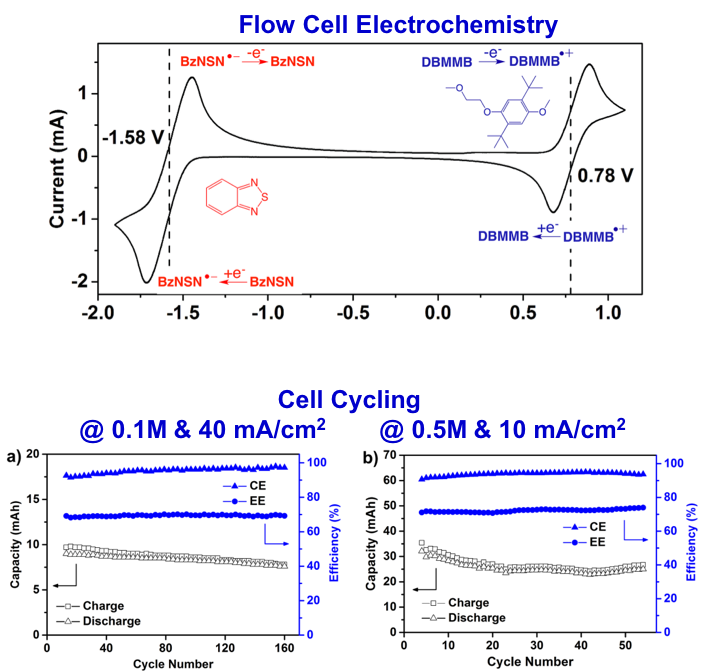
“Wine-Dark Sea” in an Organic Flow Battery: Storing Negative Charge in 2,1,3-Benzothiadiazole Radicals Leads to Improved Cyclability
Scientific Achievement 2,1,3-benzothiadiazole (BzNSN) was identified as a promising anolyte molecule for non-aqueous organic redox flow batteries. A proof-of-principle 2.36 V nonaqueous organic flow battery was developed by coupling BzNSN with DBMMB. Relatively stable performances over extended cycling were demonstrated under tested flow cell conditions,… Read More
-
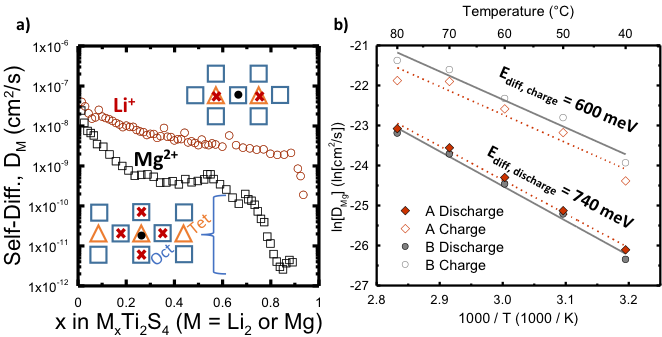
Mono vs Divalent Cation Diffusion in Thiospinel Ti2S4
We showed why the positive electrode material, MgxTi2S4, does not work well at room temperature, unlike Li2xTi2S4, which works very well. Furthermore, we confirmed that theoretical diffusion calculations predict accurate results at low x values. Read More
-
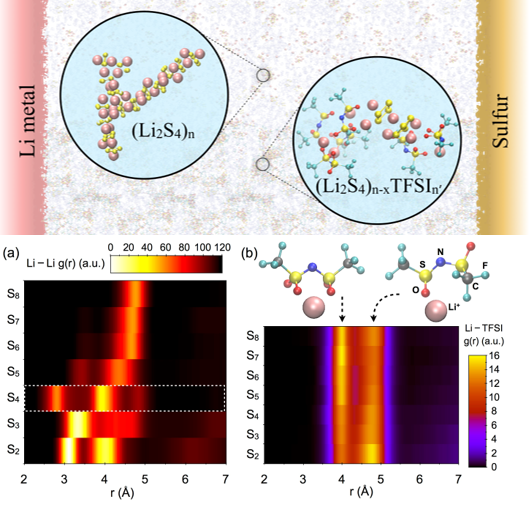
Elucidating the Solvation Structure and Dynamics of Lithium Polysulfides
A coupled theoretical and experimental study of bulk solvation structure and transport properties of lithium salt (Li-TFSI) and polysulfides species revealed extensive cluster formation in lower order polysulfides (Sx2-; x≤4), whereas the longer polysulfides (Sx2-; x>4) show high solubility and slow dynamics in the solution. Read More
-
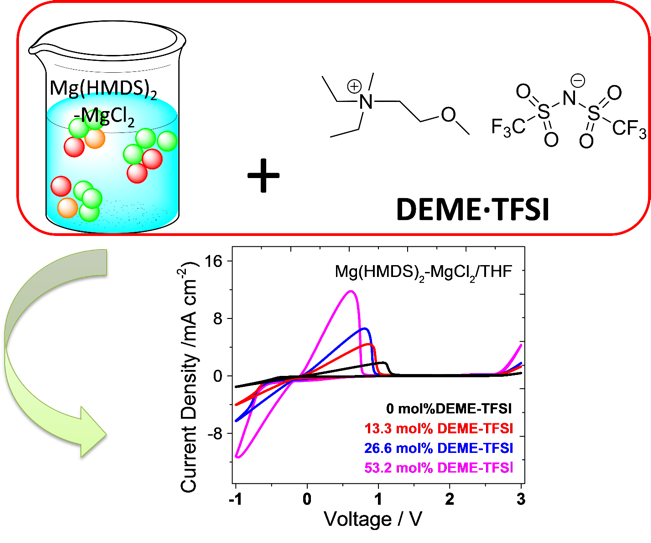
Ionic Liquid as an Effective Additive for Rechargeable Magnesium Batteries
The effect of the addition of an ionic liquid DEME·TFSI to an electrolyte solution of Mg(HMDS)2-MgCl2 in THF was spectroscopically and electrochemically studied. Reversible magnesium deposition/dissolution and cycling of Mg-Mo6S8 coin cells were achieved with the DEME·TFSI-modified magnesium electrolyte. Read More
-
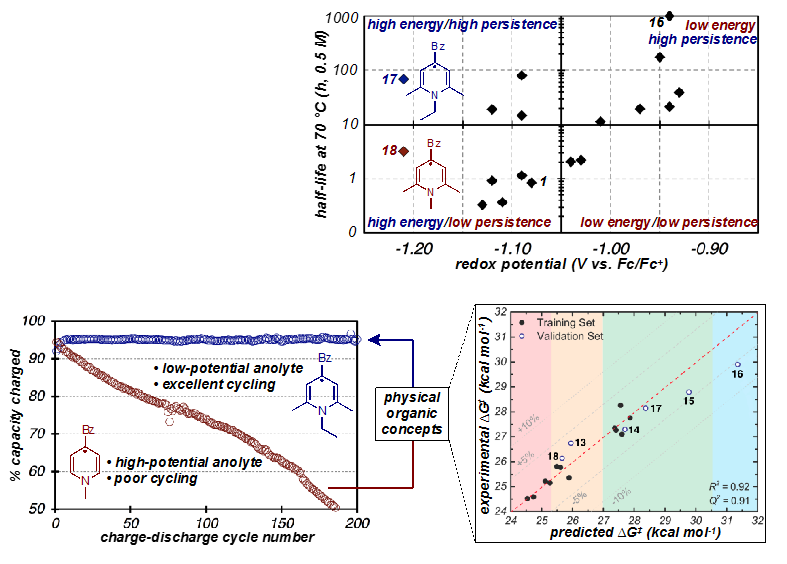
Physical Organic Approach to Persistent, Cyclable, Low-Potential Electrolytes for Flow Battery Applications
A rare example of a high energy (low potential) anolyte with excellent stability in the charged state was identified. This material undergoes cycling at potentials lower than other studied materials but does not show degradation even after 200 charge-discharge cycles. Predictive multidimensional analysis, a technique common to physical organic chemistry, was successfully applied to the development of a model for RFB electrolytes that predicts their stability in the charged state with high accuracy. Read More
-
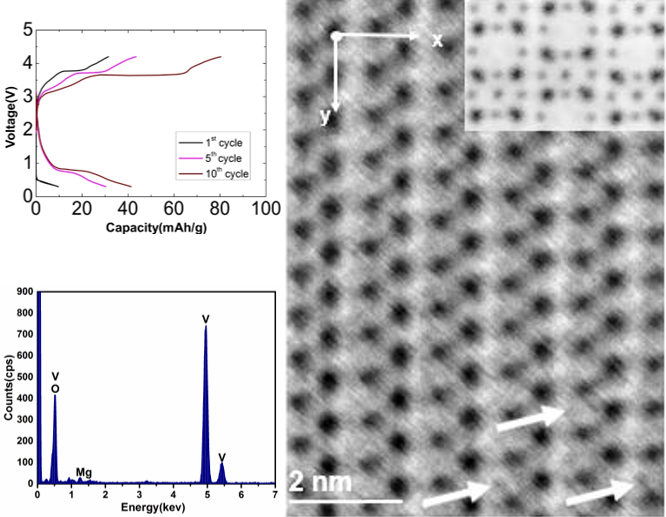
Investigation of Mg Intercalation into α-V2O5 Using Atomic Resolution Transmission Electron Microscopy
We examine the effects of Mg intercalation into α-V2O5 using atomic-resolution scanning transmission electron microscopy (STEM) and electron energy-loss spectroscopy. The position of intercalated Mg within the V2O5 unit cell is directly identified using annular bright-field imaging, and found to be in agreement with prior density-functional theory models. Read More
-
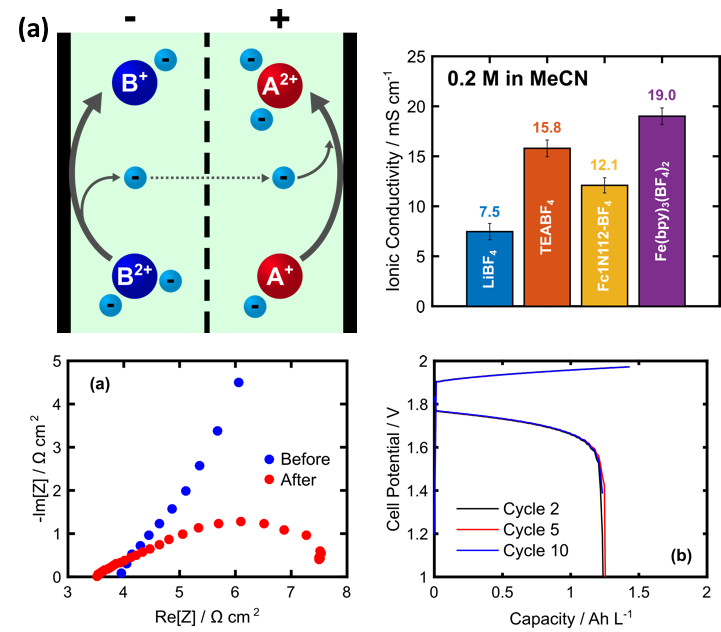
Feasibility of a Supporting Salt Free Non-aqueous Redox Flow Battery Utilizing Ionic Active Materials
We designed a non-aqueous flow battery that operates in the absence of supporting salt by utilizing redox active molecules that remain as ions across all accessible states of charge. Read More

