Latest Updates
-

Inside Tesla's Deal to Buy SolarCity for $2.6 Billion
Tesla Motors Inc. has agreed to purchase SolarCity for $2.6 billion deal that combines Elon Musk’s electric-car maker and clean-energy companies. Argonne National Laboratory’s George Crabtree, Techonomy’s David Kirkpatrick and Consumer Edge Research’s James Albertine speak on “Bloomberg West.”… Read More
-
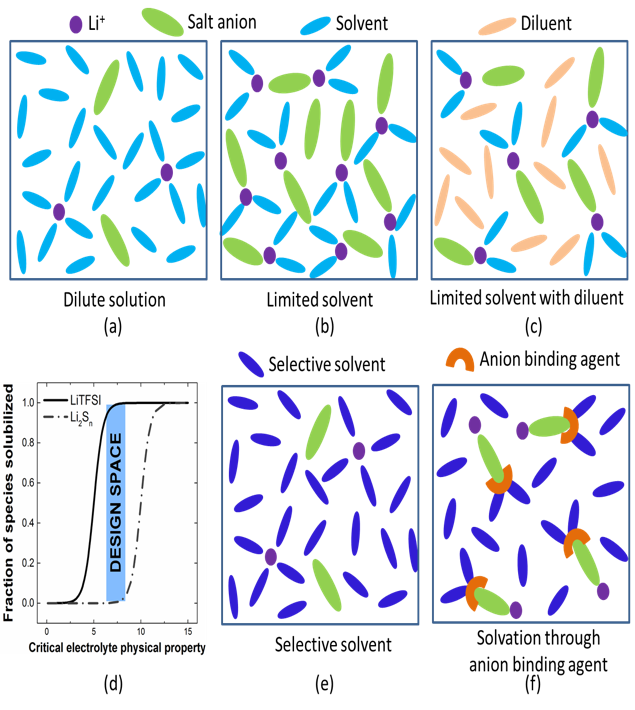
Sparingly Solvating Electrolytes for High Energy Density Lithium-Sulfur Batteries
This work presents the promising new concepts of using sparingly solvating electrolyte to enable Li-S battery operation at lean electrolyte condition, as well as the design rules for discovering new electrolyte systems. Read More
-
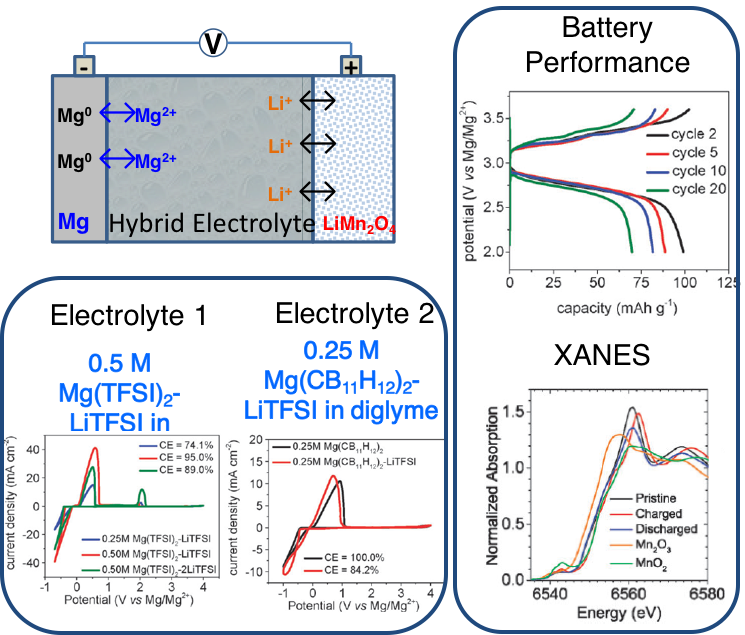
Advanced Hybrid Batteries with a Magnesium Metal Anode and Spinel LiMn₂O₄ Cathode
Two Mg-Li dual salt hybrid electrolytes were developed with excellent oxidative stability up to around 3.8 V (vs Mg/Mg2+) on a aluminum current collector, enabling the successful coupling of several state-of-the-art lithium-ion intercalation cathodes (LiMn2O4, LiCoO2 and LiNi1/3Mn1/3Co1/3O2) with magnesium metal anodes. Read More
-
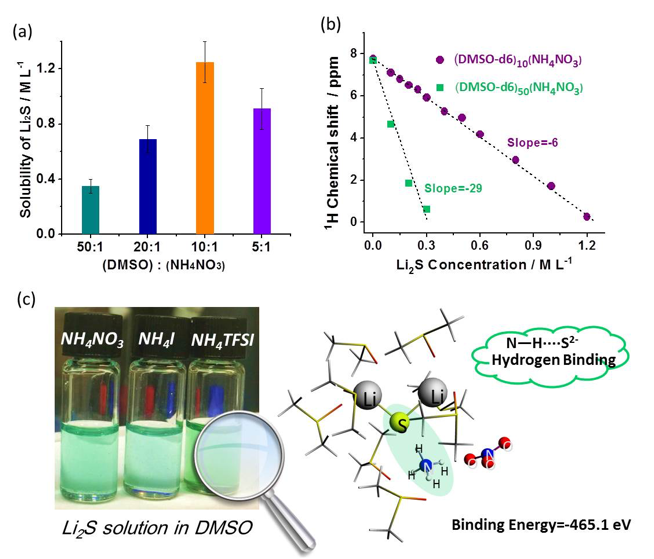
Ammonium Additives to Dissolve Li2S through Hydrogen Binding for High Energy Li-S Batteries
Ammonium salts are demonstrated as effective additives to promote the dissolution of Li2S (up to concentrations of 1.25 M) in DMSO solvent at room temperature through hydrogen binding between N-H groups and S2- anions. Read More
-
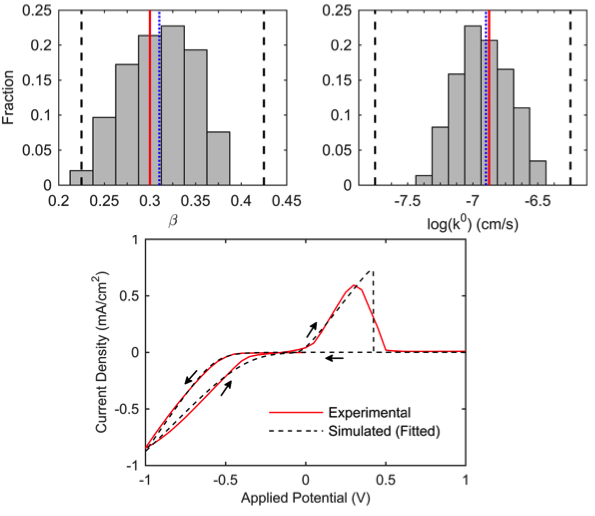
Computational Model of Magnesium Deposition and Dissolution for Property Determination via Cyclic Voltammetry
A computationally inexpensive model has been developed to parametrically study the cyclic voltammetry behavior of magnesium-based battery electrolytes. Read More
-
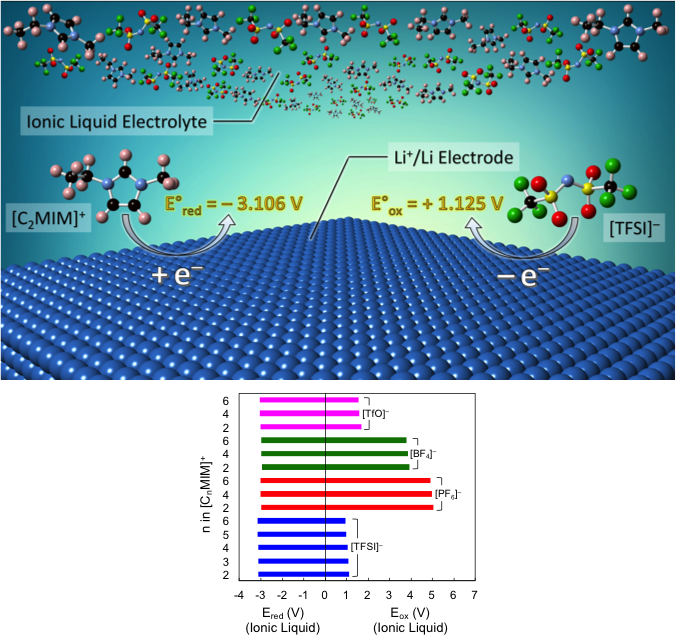
Electrochemical Stability Window of Imidazolium-Based Ionic Liquids as Electrolytes for Lithium Batteries
The oxidation and reduction potentials of the constituent cations and anions of a series of imidazolium-based ionic liquids with respect to a Li+/Li reference electrode were calculated using density functional theory (DFT) following the method of thermodynamic cycles. Read More
-
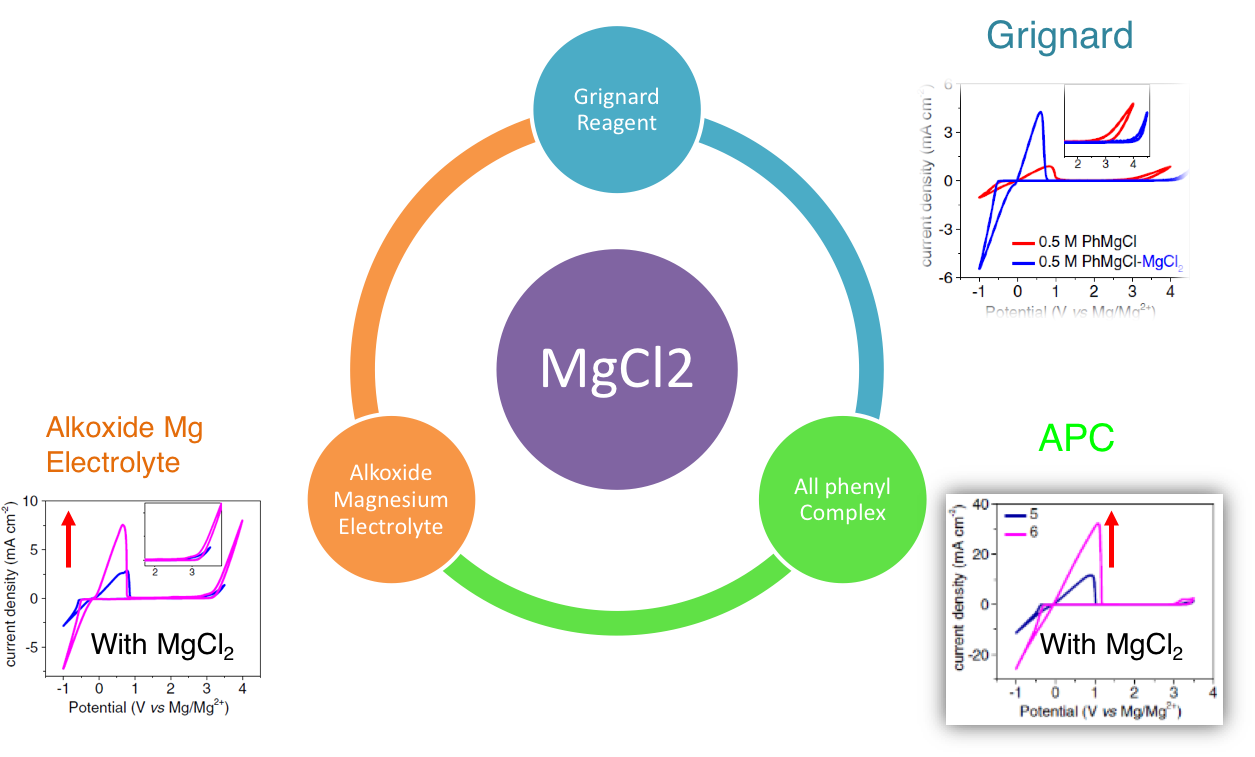
MgCl2: The Key Ingredient to Improve Chloride Containing Electrolytes for Rechargeable Magnesium-Ion Batteries
The effect of MgCl2 on a series of chloride containing magnesium electrolytes has been experimentally investigated. The incorporation of MgCl2 into these electrolytes results in the significant improvement in all aspects of electrochemical properties. Read More
-
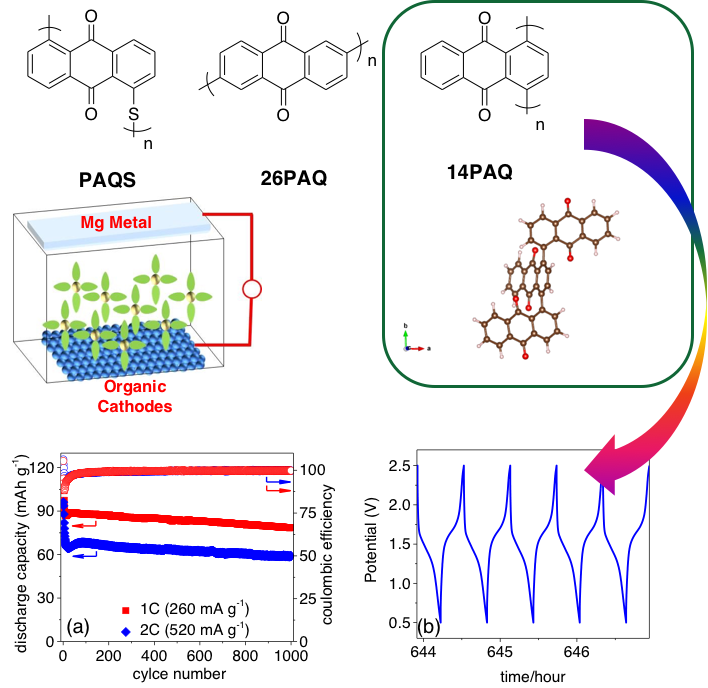
A Polyanthraquinone-based Organic Cathode for High-performance Rechargeable Magnesium-ion Batteries
A series of anthraquinonyl-based polymers (PAQS, 14PAQ, and 26PAQ) have been prepared and evaluated as high-performance organic cathodes for rechargeable Mg-ion batteries. Mg-14PAQ system shows the best cycling stability among the series: at the current rate of 0.5C, 104.9 mAh g-1 discharge capacity can be obtained in the 100th cycle; successful 1000 cycles with a small amount of capacity loss at 1C. Read More
-
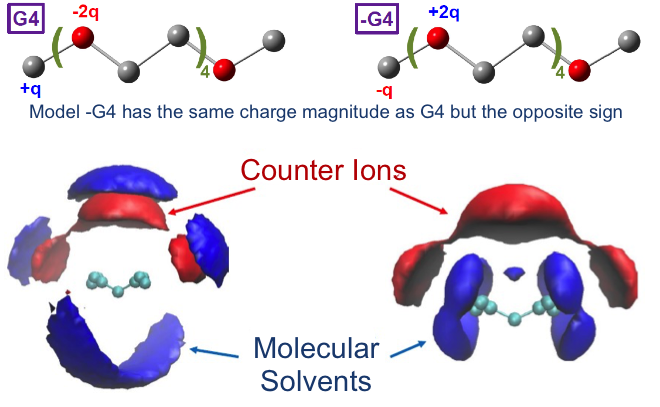
How Mixing Tetraglyme with Ionic Liquid Changes Volumetric and Transport Properties
Charge localization of the solvent molecules affects the liquid phase structure and transport properties in electrolyte solutions. Read More
-
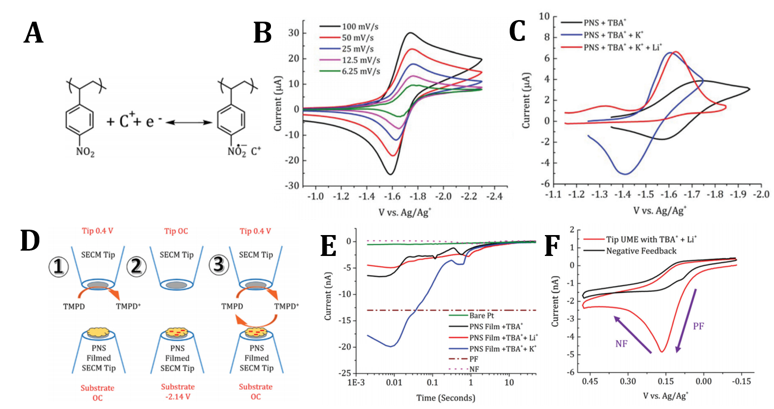
Enhancing the Performance of a Redox Active Polymer via Specific Electrolyte Interactions
A redox active polymer (RAP) with nitro- substituents was synthesized and studied via voltammetric analysis in combination with surface interrogation and ionic sensitive scanning electrochemical microscopy to reveal specific electrolyte interactions on the performance of the material. Read More
-

Effect of the Anion Activity on the Stability of Li Metal Anodes in Lithium-Sulfur Batteries
Discovered why the salt LiTFSI -- when added to the electrolyte of a Li-S battery -- allows the battery to hold a charge much longer than other salts Read More
-
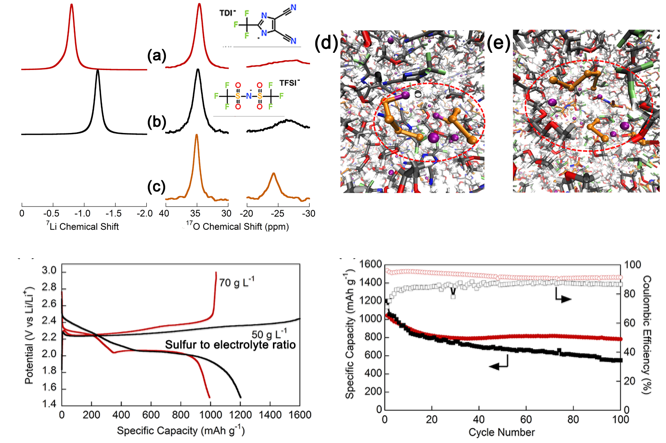
Restricting the Solubility of Polysulfides in Li-S Batteries Via Electrolyte Salt Selection
Lithium 2-trifluoromethyl-4,5-dicyanoimidazole (LiTDI) as a supporting salt in electrolytes suppresses the maximum solubility of Li2S8 by forming a Li4S8 dimer rather than the Li2S3 and Li2S5 observed in a LiTFSI electrolyte, which enables a cell with a high sulfur loading (3 mg-S cm-2) to deliver a 1.67 mAh cm-2 areal capacity after 300 stable cycles at a high current density (2.4 mA cm-2). Read More
-
The Electrochemical Discovery Laboratory
The Electrochemical Discovery Laboratory (EDL) — a key JCESR discovery tool located at Argonne — synthesizes high-quality materials for testing in beyond-lithium-ion batteries and characterizes their properties with state-of-the-art analytical techniques. Read More
-
The Electrolyte Genome Project
Traditional chemistry relies on intuition and experience to select a few materials that might work well for new electrolytes. The Electrolyte Genome streamlines this process by evaluating thousands of materials by simulation on the computer and choosing the most promising few for synthesis in the laboratory. Read More
-
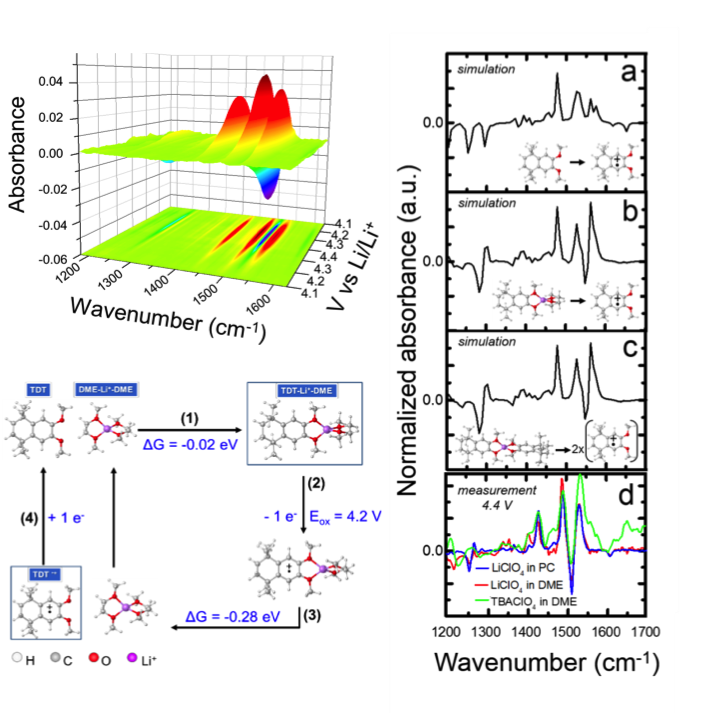
Controlling degradation pathways in organic electrochemistry via redox-mediated Li+ coordination
Exhaustive DFT experiments are well-matched to in-situ spectroscopic data showing Li+ coordination to basic methoxy groups. Li+ coordination promotes improved redox reversibility within Li+ electrolytes. Read More
-
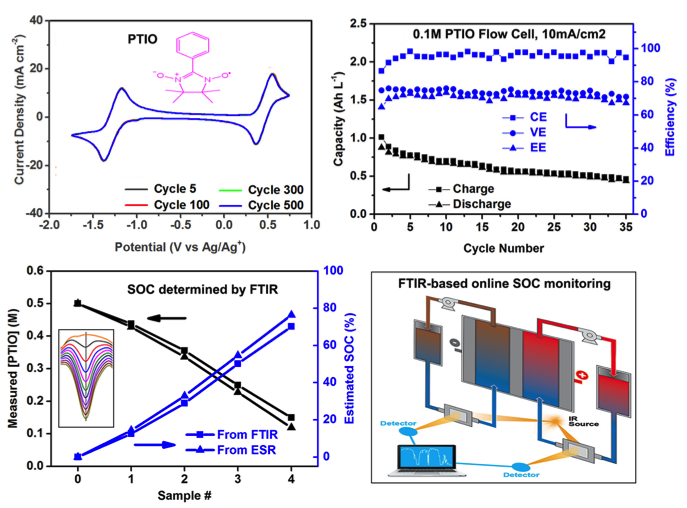
A Symmetric Organic - Based Nonaqueous Redox Flow Battery and Its State of Charge Diagnostics by FTIR
A symmetric nonaqueous flow battery based on the highly soluble, ambipolar PTIO achieved a cell voltage of ~1.7V and decent cyclability. We demonstrated FTIR as an effective method to monitor the state of charge (SOC) of this flow battery. Read More
-
Water as a Catalyst – Improving how Batteries Function
Anyone who has ever dropped a cell phone in the sink will tell you that electrical devices and water do not go together. However, a new study has shown that conventional wisdom may not hold on the molecular scale in some beyond-lithium-ion batteries. Read More
-
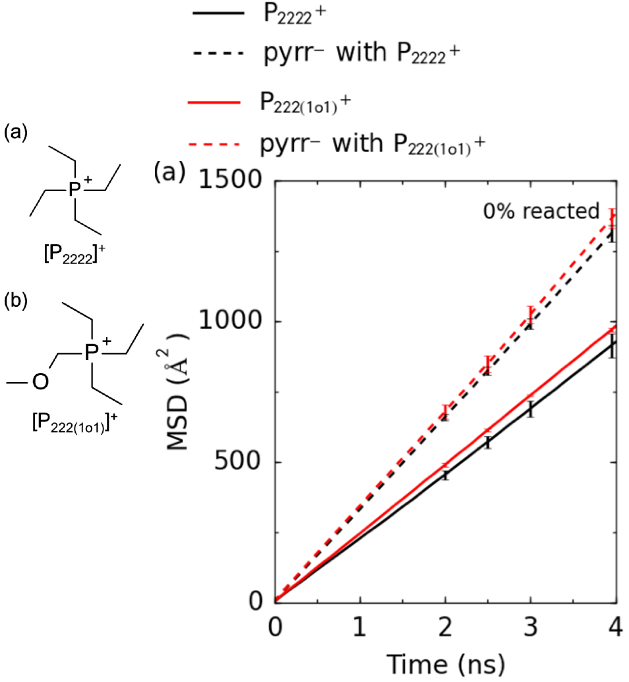
Molecular Design of Low Viscosity Ionic Liquid for High Performance Electrolyte
Computationally predicted viscosities confirmed by experimental measurements. Read More
-

JCESR Scientific Sprints - Speed through Collaboration
JCESR supplements its traditional project management approach with scientific “Sprints.” Sprints take a single question from JCESR’s catalog of prioritized scientific challenges and dedicate a small, multidisciplinary team of 5-15 members to answer it, enabling us to move forward more rapidly in our research. Sprints empower early-career scientists to show their leadership qualities in the Sprints they lead. Once a Sprint is completed, the outcome is documented within JCESR and shared with the research community. The resulting new knowledge then informs and inspires subsequent research challenges. Read More
-

JCESR Scientific Sprints - Better Polymers for Better Batteries
JCESR supplements its traditional project management approach with scientific “Sprints.” The sprint described in this video involved a multidisciplinary team from Argonne, the University of Illinois at Urbana-Champaign, Massachusetts Institute of Technology, and the University of Michigan. As they studied how polymers in solution… Read More

