Latest Updates
-
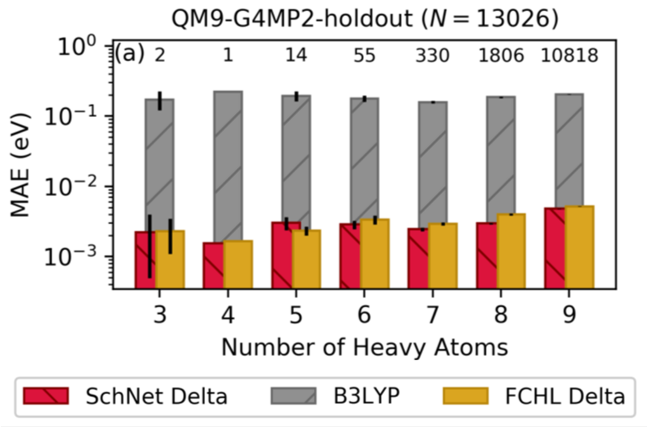
Accelerating Electrolyte Discovery for Energy Storage through Machine Learning
Utilized high performance computing to generate a database of highly accurate quantum chemical energies of 133 K organic molecules and used machine learning to enable prediction of energies from low fidelity, low cost quantum chemical calculations. Read More
-
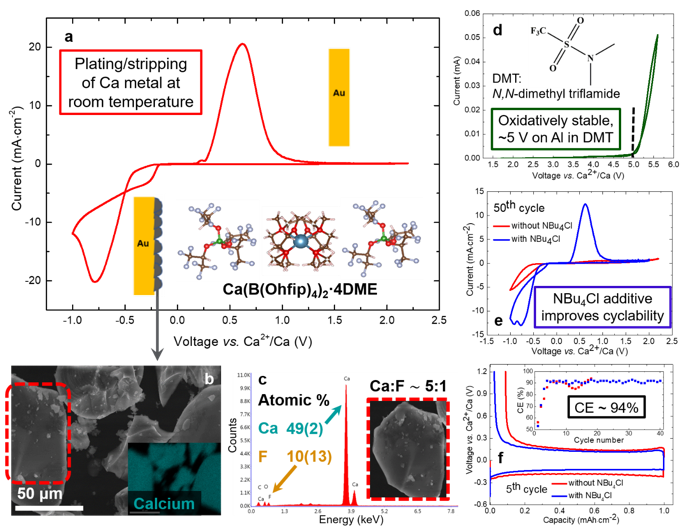
Reversible Calcium Plating and Stripping at Room Temperature Using a Borate Salt
Novel low-ion pairing calcium salt developed that enables reversible calcium plating and stripping in 1,2-dimethoxy ethane at room temperature. Read More
-
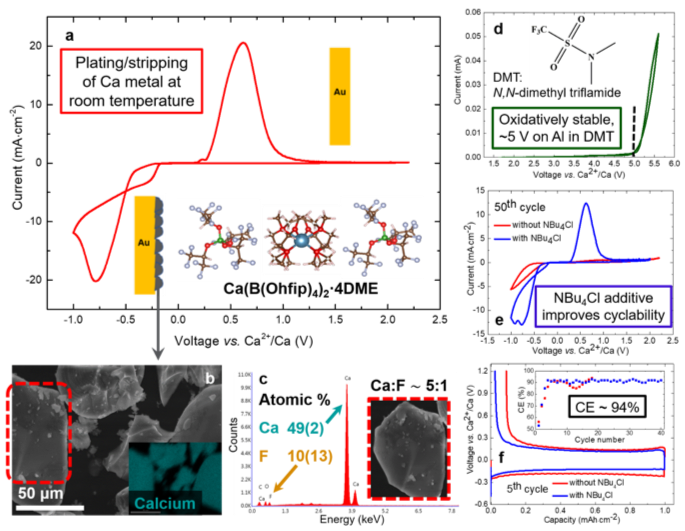
Reversible Calcium Plating and Stripping at Room Temperature Using a Borate Salt
Novel low-ion pairing calcium salt developed that enables reversible calcium plating and stripping in 1,2-dimethoxy ethane at room temperature. Read More
-
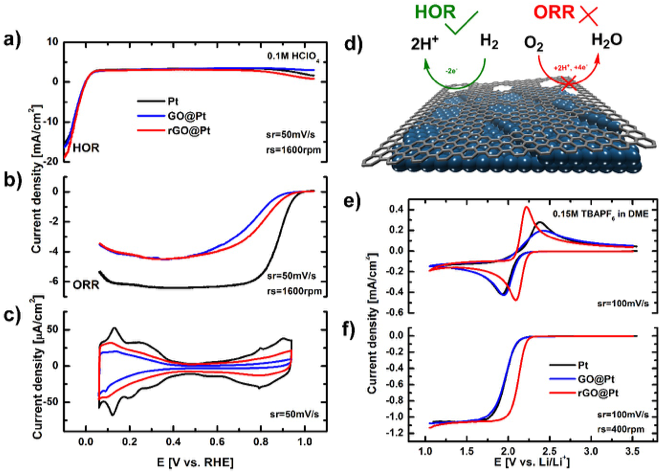
Tuning the Selectivity and Activity of Electrochemical Interfaces with Defective Graphene Oxide and Reduced Graphene Oxide
A modified Langmuir-Blodgett approach is developed for the deposition of GO and rGO films that enables ex situ engineering of the activity and selectivity of electrochemical interfaces in both aqueous and non-aqueous electrolytes. Read More
-
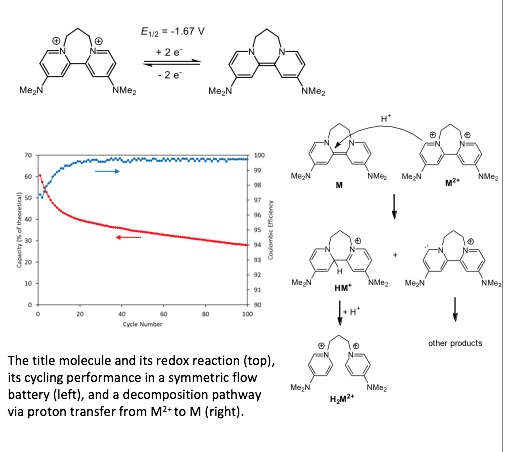
An Organic Super-Electron-Donor as a High Energy Density Negative Electrolyte for Nonaqueous Flow Batteries
A previously reported organic “super-electron-donor” was shown to have several favorable properties for application as an anolyte in nonaqueous flow batteries. Read More
-
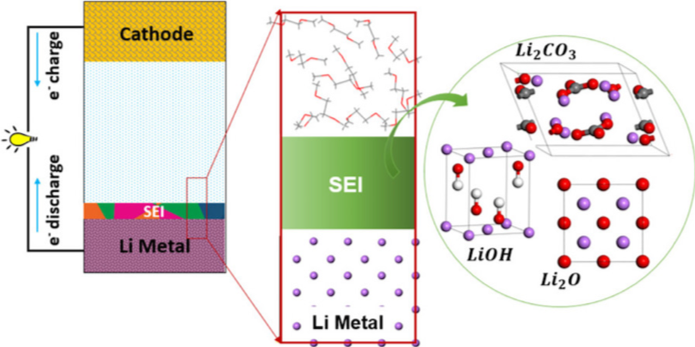
Roles of inorganic surface layers on solid electrolyte interphase evolution at Li-metal anodes
We uncovered the effects of nanometer-sized lithium oxide, lithium hydroxide, and lithium carbonate as surface passivation layers on the interfacial reactivity of Li-metal anodes. Read More
-
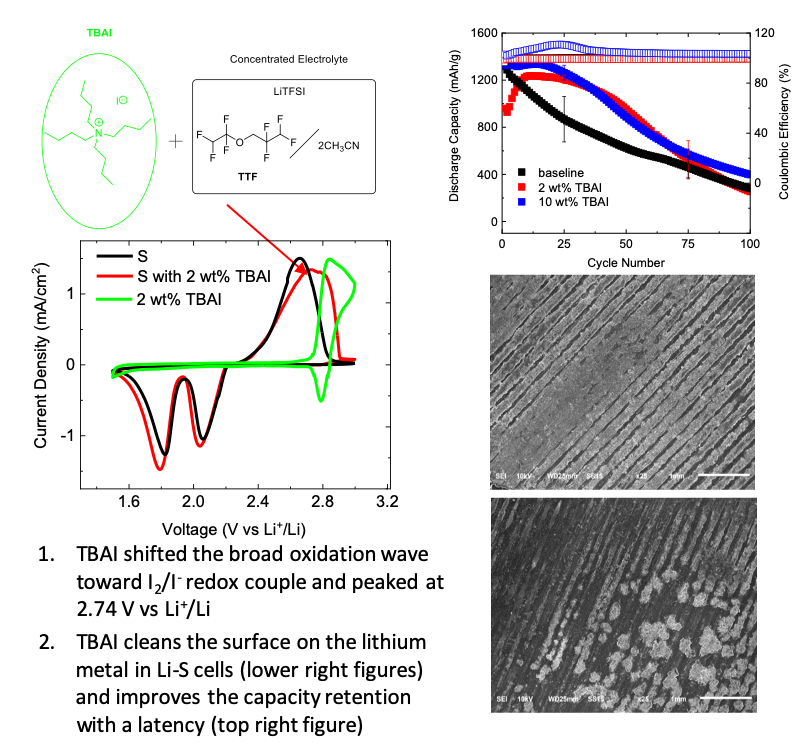
Lipophilic Additives for Highly Concentrated Electrolytes in Lithium-Sulfur Batteries
Developed a lipophilic additive, tetrabutylammonium iodide (TBAI), for highly concentrated electrolytes and tested its electrochemical performance in lithium-sulfur batteries. Read More
-
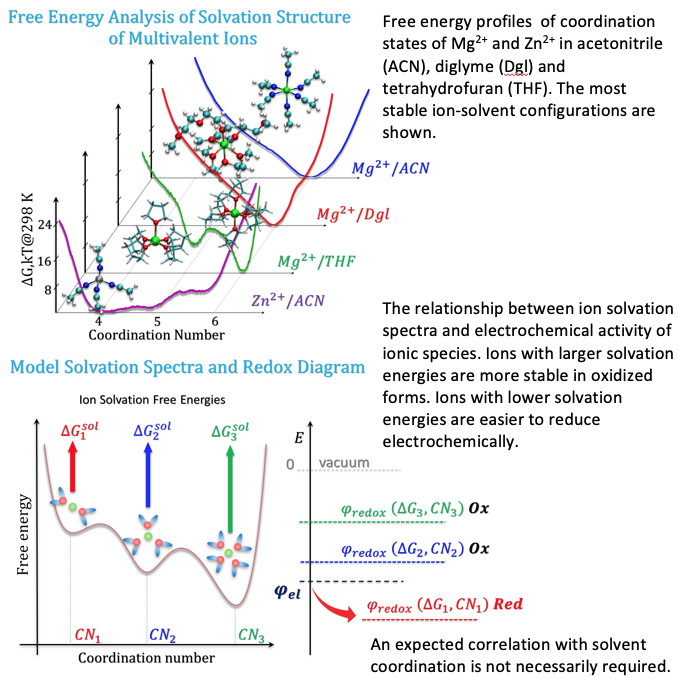
“Ion Solvation Spectra”: Free Energy Analysis of Solvation Structures of Multivalent Cations in Aprotic Solvents
Advanced free-energy sampling techniques reveal that monoatomic divalent cations in organic solvents may have multiple well-defined minima or plateaus of free energy with respect to the number of coordinating solvent molecules. We introduce the concept of “ion solvation spectra” to highlight non-trivial phenomenology related to a continuum of ion solvation environments, as opposed to a single coordination minimum. Read More
-

Modified Ohm’s Law in Lithium and Beyond-lithium Battery Electrolytes
Battery electrolytes contain two mobile charged species of opposite charge and thus the traditional Ohm’s law, which applies to one mobile charged species, must be modified. This review uses a modified Ohm’s law to rank order electrolytes. Read More
-
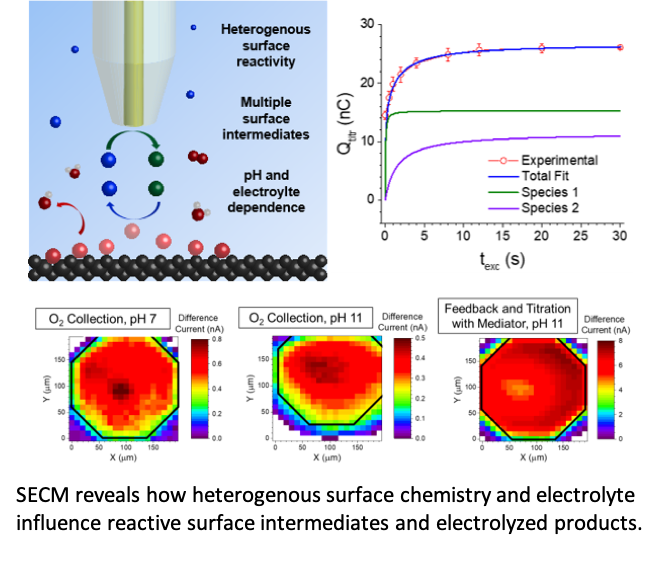
Probing Reactive Oxygen Species on Boron-Doped Diamond Anodes
Multiple surface oxygen species form and react at different rates on sp2 carbon-containing boron-doped diamond electrodes during water oxidation. Read More
-
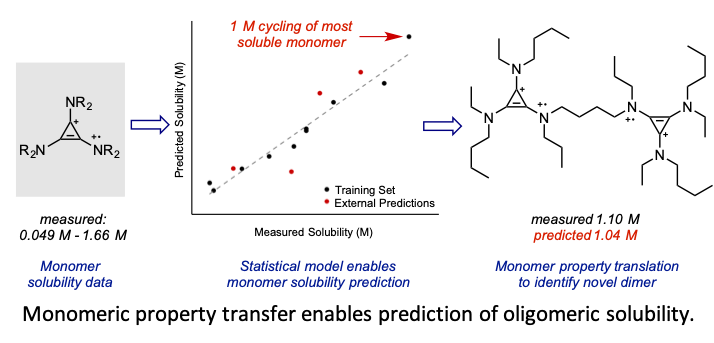
Developing a Predictive Solubility Model for Monomeric and Oligomeric Cyclopropenium-Based Flow Battery Catholytes
Two important field-wide goals are met through this study: the development of a statistical model for the solubility of conformationally flexible molecules in acetonitrile and the operation of a high concentration (1 M) nonaqueous organic symmetric flow cell. Read More
-
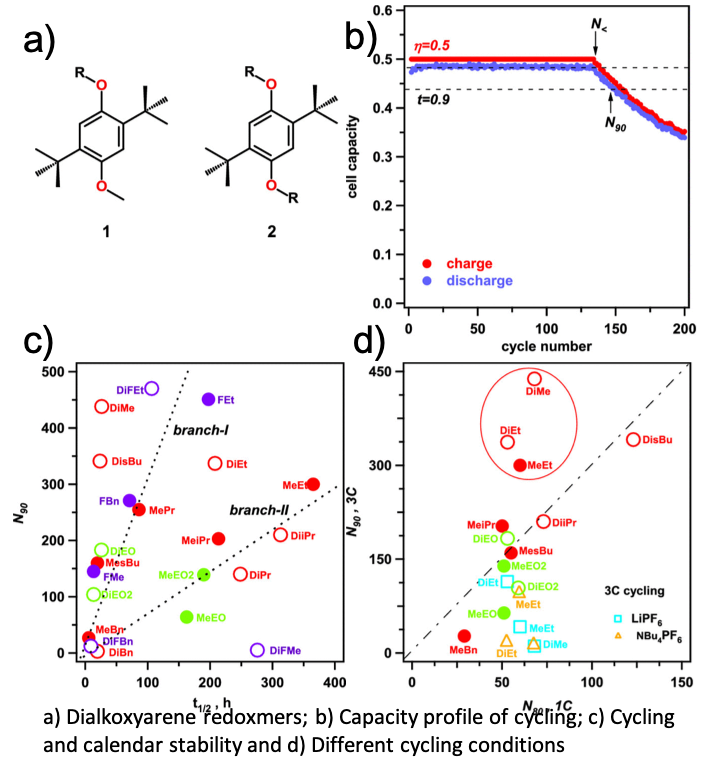
On Transferability of Performance Metrics for Redox-active Molecules
It is shown that two commonly used performance metrics for charged redoxmers in flow batteries, viz. their cycling and calendar lifetimes, generally do not correlate with each other. It is also demonstrated that the cycling stability is extremely sensitive to experimental detail; unless the conditions are tightly controlled, this metric is unsuitable as a guide for materials development. Read More
-
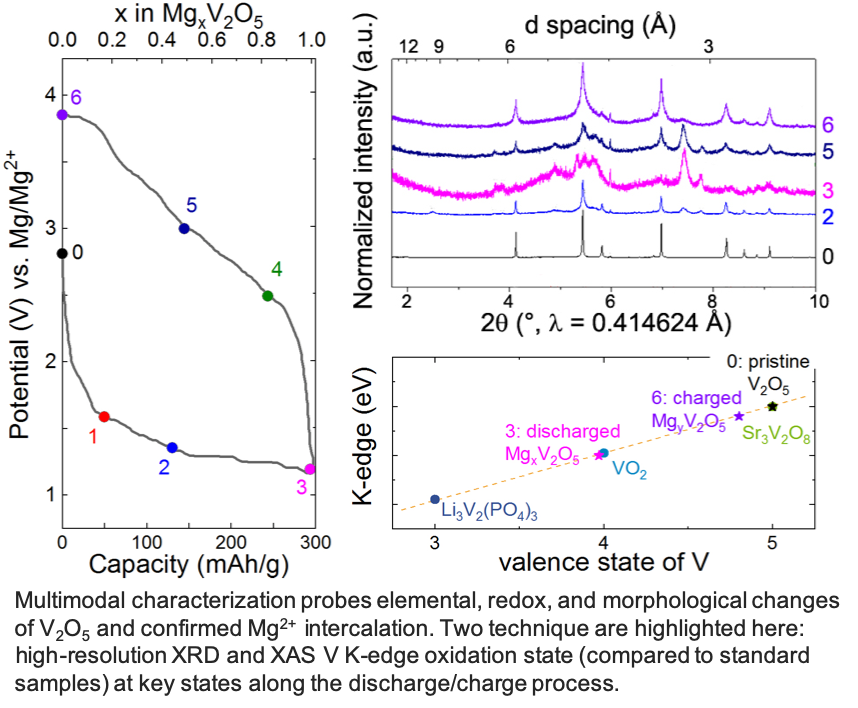
Intercalation of Magnesium into a Layered V2O5
Layered a-V2O5 reversibly intercalates 1 Mg2+ per unit formula to achieve 280 mAh/g at 110°C in a chemically and anodically stable ionic liquid electrolyte. Read More
-
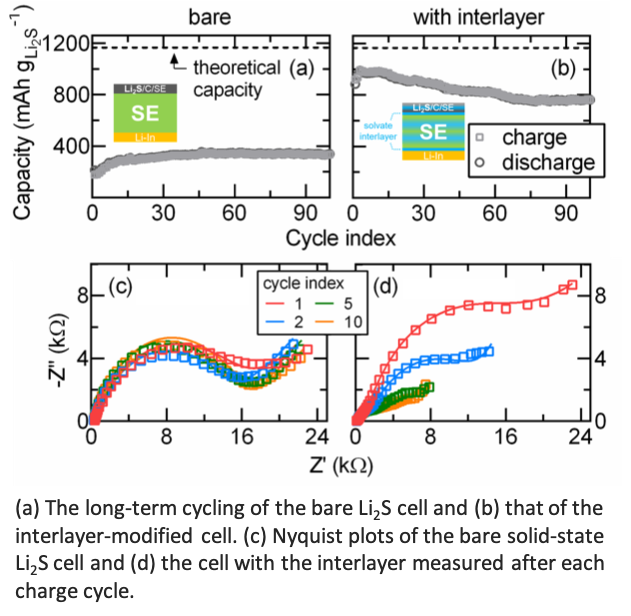
Incorporating Solvate and Solid Electrolytes for All-Solid-State Li2S Batteries with High Capacity and Long Cycle Life
Incorporating highly concentrated solvate electrolyte into all‐solid‐state Li2S batteries significantly improves the battery performance compared to its bare counterpart. The origin of the improved battery cyclability is the favorable interfacial contact, where the ion conducting network surrounding the active material remains intact even after the significant volume contraction of the Li2S cathode. Read More
-
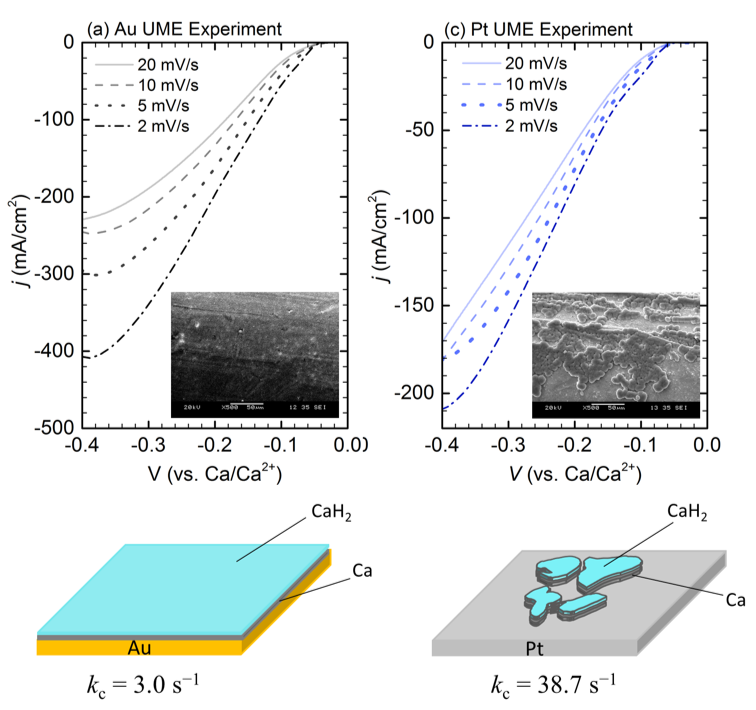
Understanding Ca Electrodeposition and Speciation Processes in Nonaqueous Electrolytes for Next-Generation Ca-Ion Batteries
Rechargeable Ca batteries have attracted little interest due to the extreme difficulty to perform reversible Ca electrodeposition. There is currently only one electrolyte, Ca(BH4)2 in THF, that exhibits reversible Ca deposition at room temperature. However, the underlying deposition mechanism and the interaction of Ca2+ ions with the surrounding molecules are still mostly unknown. Our research explores Ca deposition and speciation processes by using electrochemical and physical characterization techniques. Read More
-

Stability and Microheterogeneity in Concentrated Nonaqueous Electrolyte Solutions
We compared the molecular dynamics of two different but structurally-related electrolytes in highly concentrated solutions. One catholyte is compact and symmetrical; the other is bulkier and less symmetrical. Incoherent elastic and quasi-elastic neutron scattering measurements showed significant differences in crystallization, supercooling, and sudden microheterogeneity. Read More
-
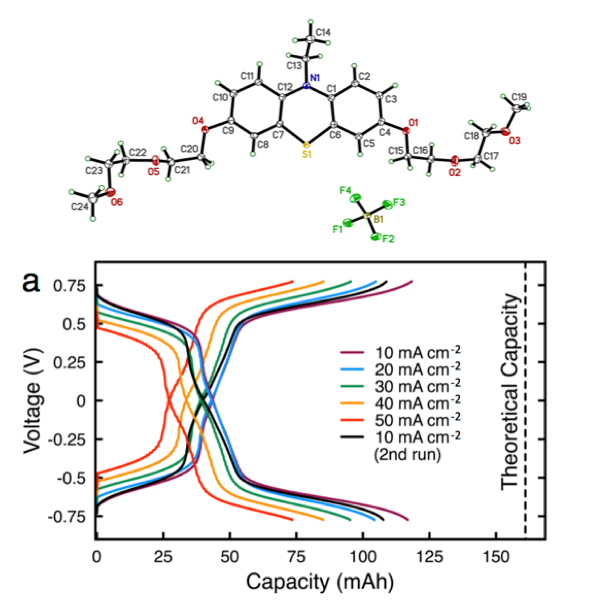
Enabling High Concentration Redox Electrolytes for Use in Nonaqueous Redox Flow Batteries
Building on prior collaborative JCESR research on iteratively advancing phenothiazines as charge storage materials, we developed a new class of phenothiazine derivatives with high equivalent charge concentration by increasing solubility and stability of multiple electron transfer events. Read More
-
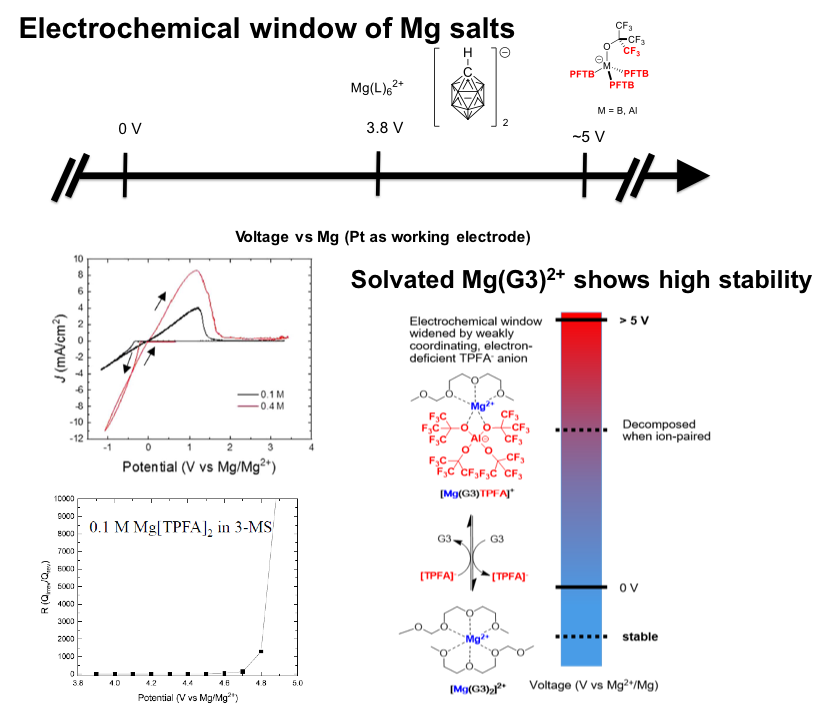
Widening Electrochemical Window of Mg Salt by Weakly Coordinating Perfluoroalkoxyaluminate Anion for Mg Battery Electrolyte
Mg[Al{OC(CF3)3}4]2 (Mg[TPFA]2 ) with perfluorinated, e- deficient anions ([TPFA]−) possesses high thermodynamic oxidative stability with high coulombic efficiency for Mg plating/stripping. Read More
-
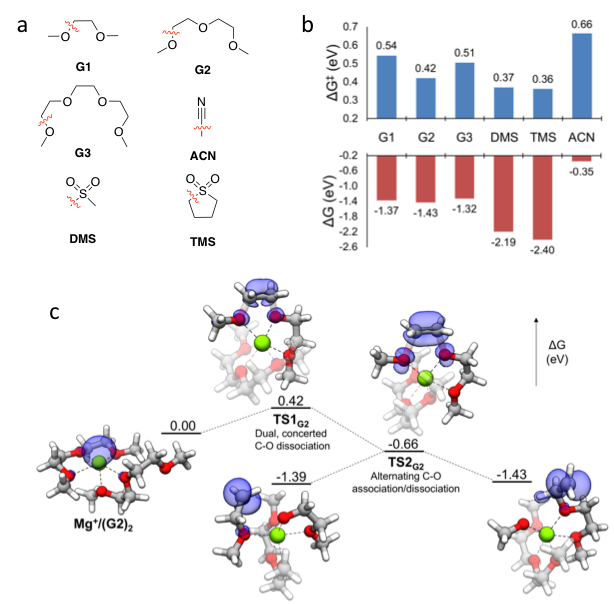
First-principles Exploration of Stability and Associated Decomposition Mechanisms of Solvents in Mg Energy Storage
Obtained a deeper understanding of the chemical and electrochemical stability governing the suitability of several organic solvents commonly considered for Mg battery electrolytes. Read More
-

Direct Observation of MgO Formation at the Cathode-Electrolyte Interface of a Spinel MgCo2O4 Cathode upon Electrochemical Mg+2 Removal and Insertion
We have been exploring the versatility and mechanism of Mg insertion into several transition metal spinel compounds and correlating the results with electron count. In this publication we established that the MgCo2O4 works by a conversion mechanism – MgO extraction and precipitation as the primary electrochemical activity with loss of oxide and reduction of cobalt. System characterization was complicated by the observed solid solution between MgCo2O4 – Co3O4 but overcome by utilizing a variety of techniques to fully characterize the materials synthesized. Read More

