Scientific Achievement
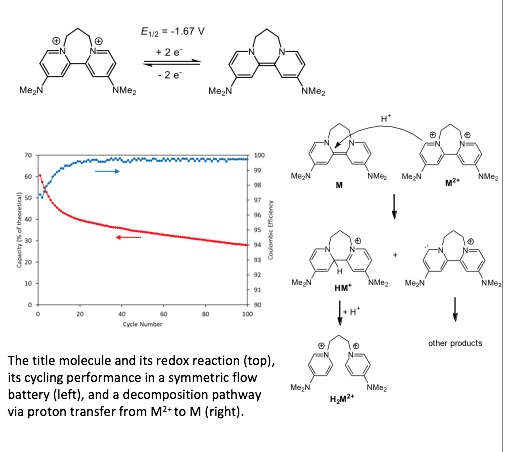
A previously reported organic “super-electron-donor” was shown to have several favorable properties for application as an anolyte in nonaqueous flow batteries.
Significance and Impact
To realize the full potential of nonaqueous flow batteries, catholytes with high oxidation potentials and anolytes with low reduction potentials (and high solubility) must be discovered/synthesized, as is the case for the molecule described herein.
Research Details
- The dication of the title molecule M2+ undergoes a 2-electron reduction to M at -1.67 V (vs ferrocene) and [M2+][PF6]2 is soluble to 0.60 M in CH3CN, providing a Faradaic concentration of up to 1.20 M.
- A decomposition route via proton transfer from M2+ to M was discovered, providing a clear path to derivatives of M with enhanced stability

