Research Highlights
-
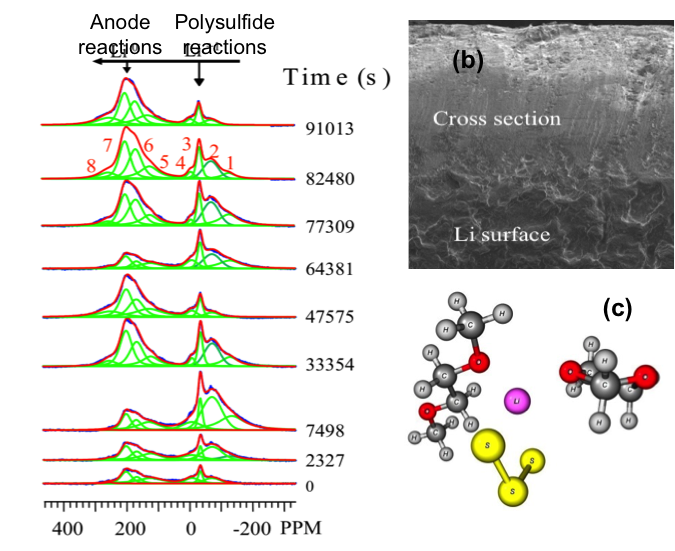
Following the Transient Reactions in Lithium-Sulfur Batteries Using an In Situ Nuclear Magnetic Resonance (NMR) Technique
First in situ NMR characterization of the full cell electrochemical reactions in Li-S batteries using a microbattery design Read More
-
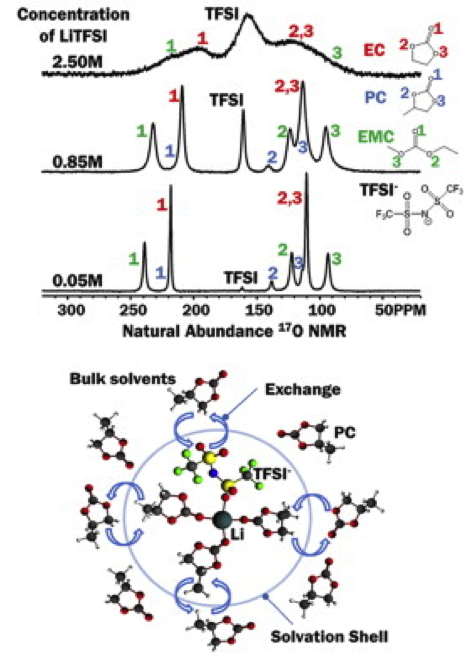
Natural Abundance 17O Nuclear Magnetic Resonance and Computational Modeling Studies of Lithium Based Liquid Electrolytes
A high sensitivity NMR probe was developed at ultra-high field, allowing 17O NMR spectra on electrolytes acquired at concentration as low as about 20 mM. Read More
-
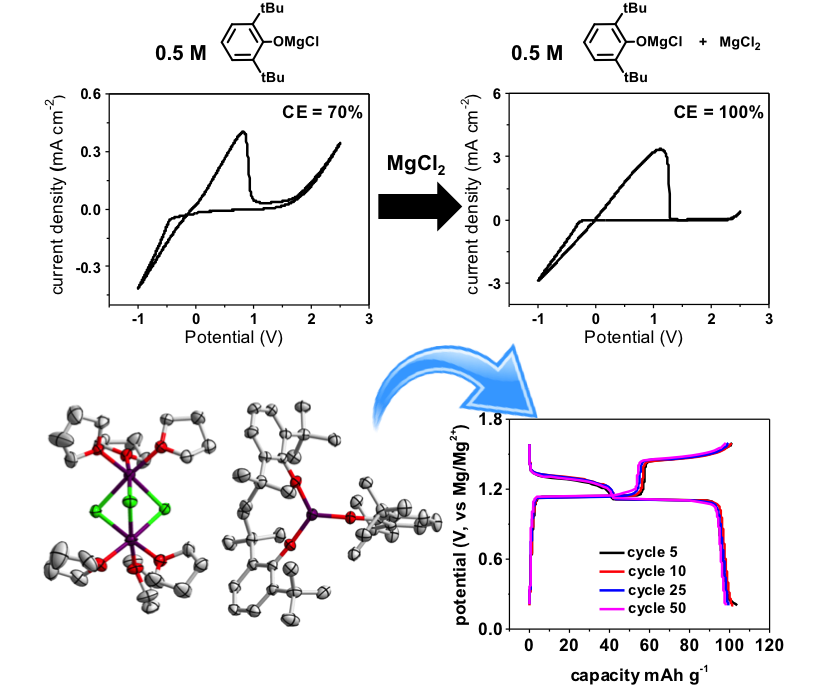
A Lewis Acid-free and Phenolate-based Magnesium Electrolyte for Rechargeable Magnesium Batteries
A novel Lewis acid-free all magnesium electrolyte containing 2,6-di-tert-butylphenoxidemagnesium chloride ((DTBP)MgCl + MgCl2) has been deliberately developed. The obtained electrolyte possesses the oxidative stability of up to 2.3 V (vs Mg/Mg2+) with 100% coulombic efficiency, which is comparable to its AlCl3 counterpart. Read More
-
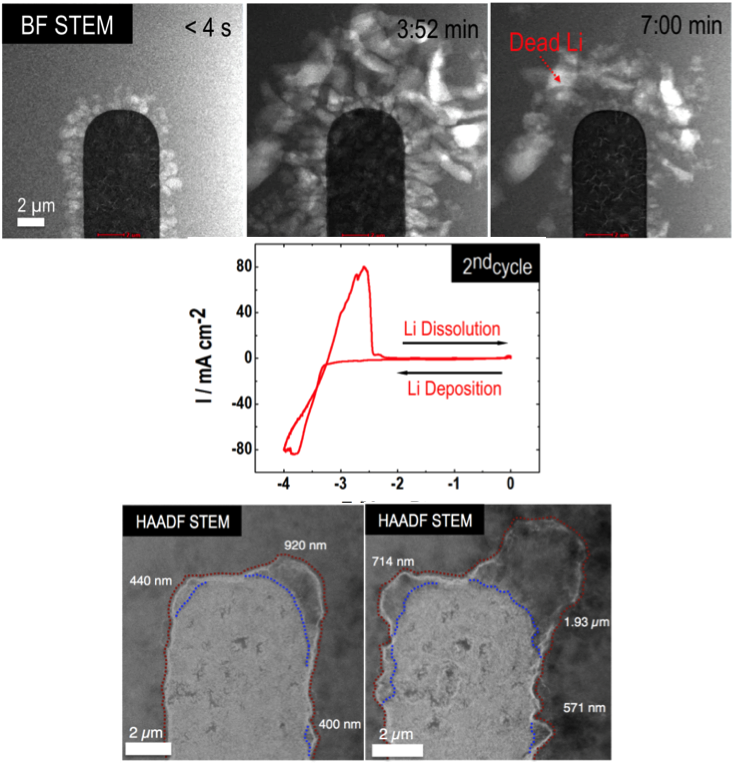
Direct Observation of SEI and Dendrite Dynamics by Operando Electrochemical Scanning Transmission Electron Microscopy
Implemented “battery in a STEM” - an operando electrochemical stage inside an Cs-corrected scanning transmission electron microscope (STEM) Read More
-
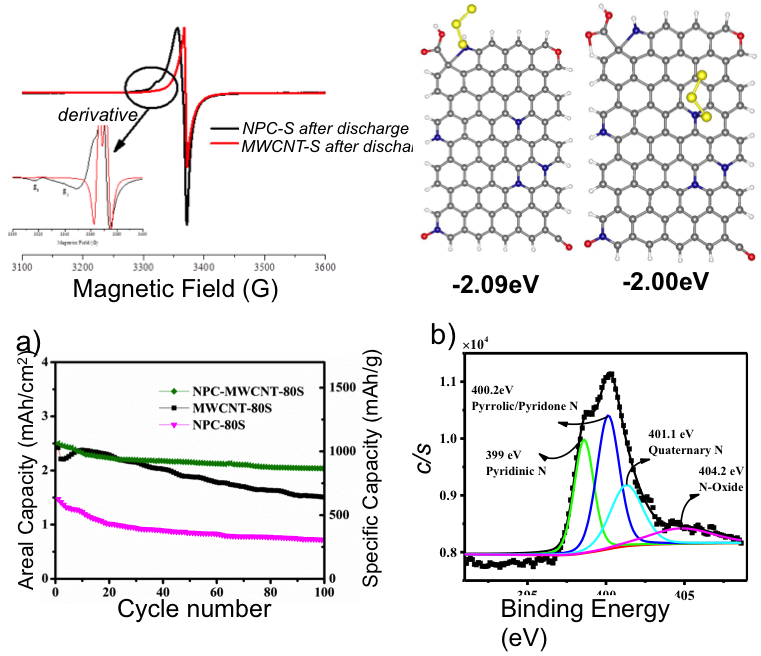
Molecular‐Confinement of Polysulfide within HybridElectrodes for High Mass Loading in Lithium Sulfur Batteries
N-doped carbon stabilizes the S3 radicals, which are rarely detected in ether-based electrolyte due to their high reactivity; this also restrains the polysulfide loss during battery cycling. Read More
-
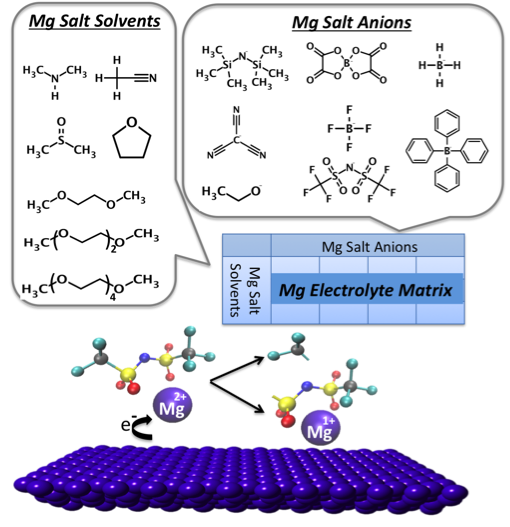
Electrolyte Genome Reveals New Instability Mechanism in Mg Electrolytes
imulations of a matrix of Mg salt and solvent combinations revealed a strong tendency to ion pair formation. Close association of the salt anion and cation within the first solvation shell, even at modest concentrations. Read More
-
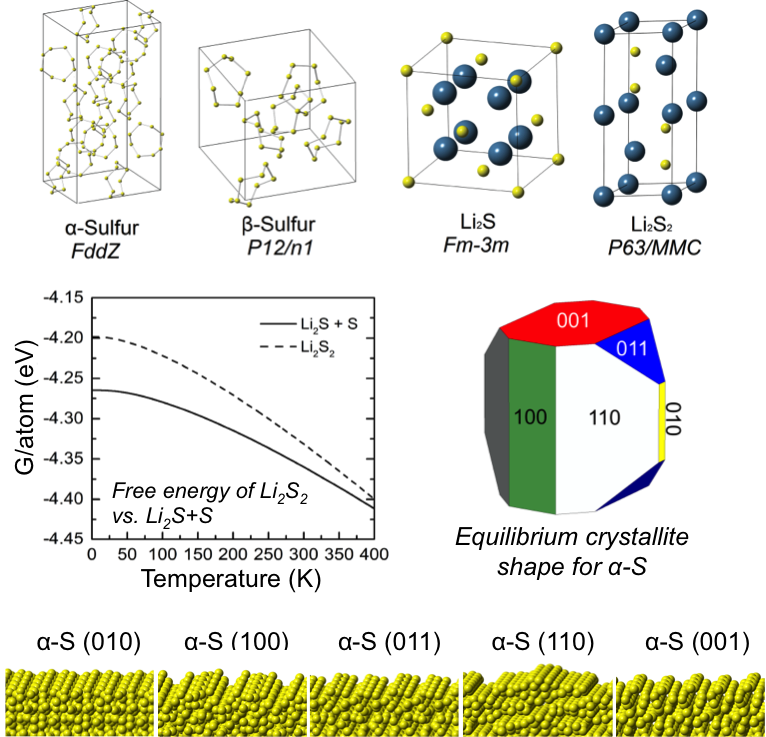
First-Principles Study of Redox End-Members in Li-Sulfur Batteries
Van der Waals-augmented density functional theory (vdW-DF), quasi-particle methods (G0W0), and continuum solvation techniques were used to predict several structural, thermodynamic, spectroscopic, electronic, and surface characteristics of solid-phase redox end-members in Li-S batteries. Read More
-
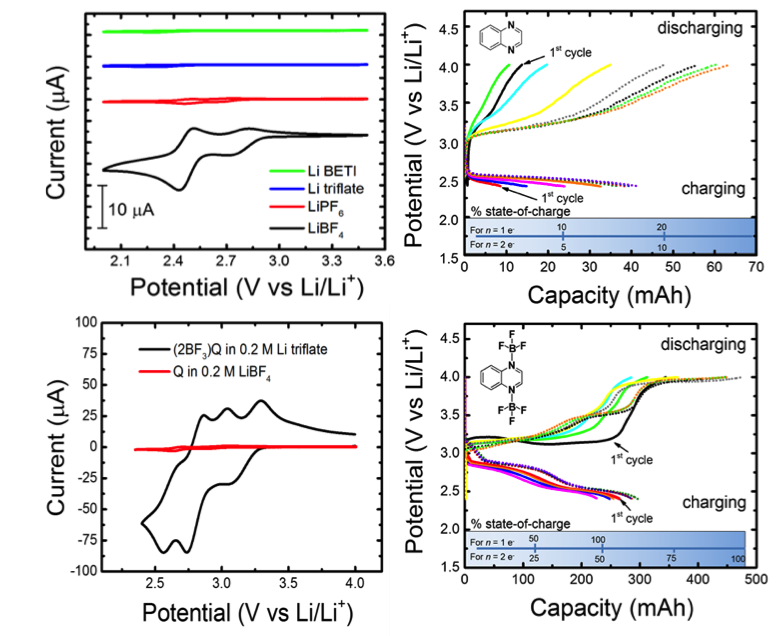
Modified Electrochemical Properties of Organic Quinoxaline via Electrolyte Interactions in Propylene Carbonate
Quinoxalines are highly sensitive to solvent and electrolyte interactions. For example, bare quinoxaline is active in acetonitrile at DFT-predicted potentials but is effectively dead in propylene carbonate. Electrochemical activation is achieved by addition of BF3-ligands. Read More
-
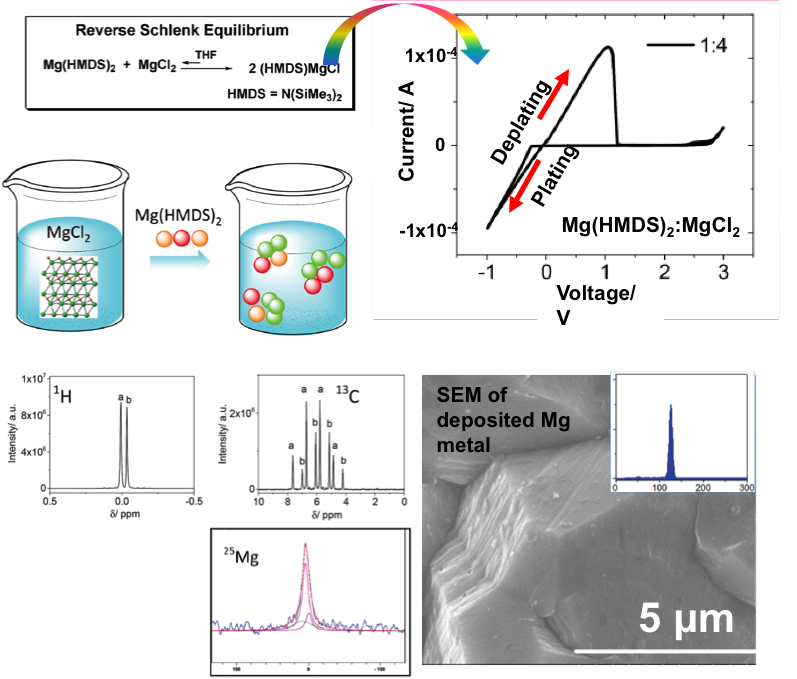
The Unexpected Discovery of the Mg(HMDS)2/MgCl2 Complex as a Magnesium Electrolyte for Rechargeable Magnesium Batteries
A simple mixture of magnesium compounds: magnesium hexamethyldisilazide (Mg(HMDS)2) and magnesium chloride (MgCl2) was prepared to achieve reversible Mg deposition/dissolution, a wide electrochemical window, and a coulombic efficiency of 99%. Read More
-
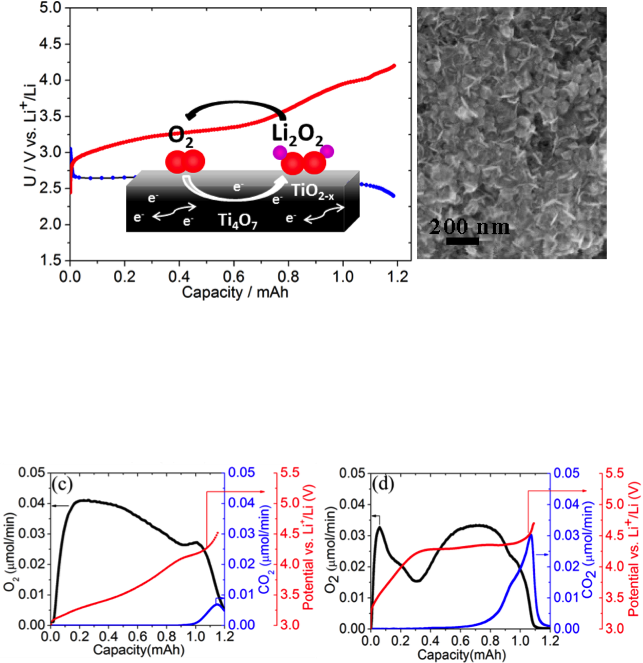
A Highly Active Nanostructured Metallic Oxide Cathode for Li-O2 Batteries
Synthesis of a low-cost nanocrystalline metallic metal oxide that shows very good ORR/OER properties in a Li-O2 cell, highlighting the importance of controlling the cathode interface to achieve better round-trip efficiency Read More
Latest Updates
-
You’re Invited - JCESR and Beyond: Translating the Basic Science of Batteries
Please join us at Argonne National Laboratory on Tuesday, April 4, 2023 for JCESR and Beyond: Translating the Basic Science of Batteries. Registration is now open. This in-person event will celebrate 10 years of research from the Joint Center… Read More
-
A Message from JCESR: In Memory of George Crabtree
It is with heavy hearts that we say goodbye to George Crabtree, a Senior Scientist and Distinguished Fellow at Argonne National Laboratory, and Director of the Joint Center for Energy Storage Research (JCESR), who passed away unexpectedly on January 23. Dr. Read More
-
Cyanopyridines As Extremely Low-Reduction-Potential Anolytes for Nonaqueous Redox Flow Batteries
Discovery of a cyanophenylpyridine derivative with a very low reduction potential and good stability during cycling. Read More
-
Characterizing Redoxmer – Electrode Kinetics Using a SECM-Based Spot Analysis Method
Identified asymmetries in electron transfer (ET) kinetics between the reduction and oxidation of ferrocene-based redoxmers by measuring the ET rate constants (kf/kb) as a function of electrode potential. Read More
-
Benzotriazoles as Low Potential Anolytes for Non-Aqueous Redox Flow Batteries
We developed an easy-to-synthesize benzotriazole-based anolyte with a high energy redox potential (-2.3 V vs Fc/Fc+) and high solubility that demonstrates stable electrochemical cycling performance. Read More

