Research Highlights
-
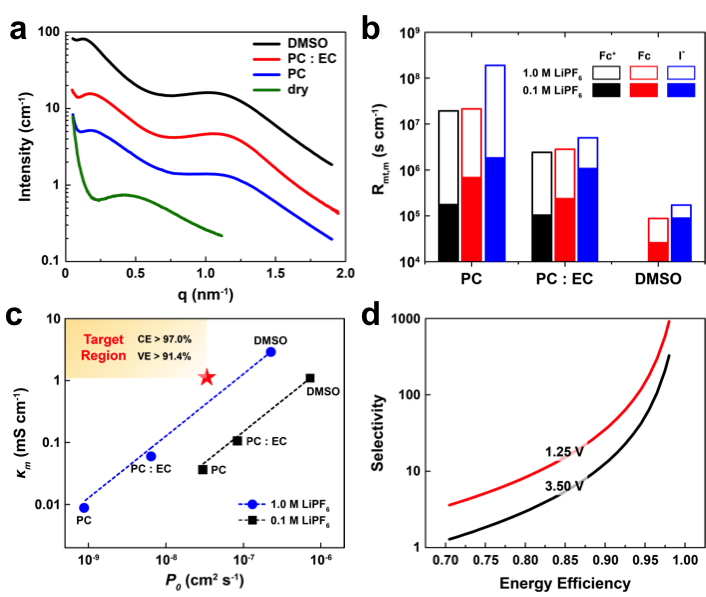
An Investigation of the Ionic Conductivity and Species Crossover of Lithiated Nafion 117 in Nonaqueous Electrolytes
Solvent volume fraction within the membrane has the greatest effect on both conductivity and crossover; as a secondary effect, the charge on redox species modifies crossover rates in accordance with Donnan exclusion. Read More
-
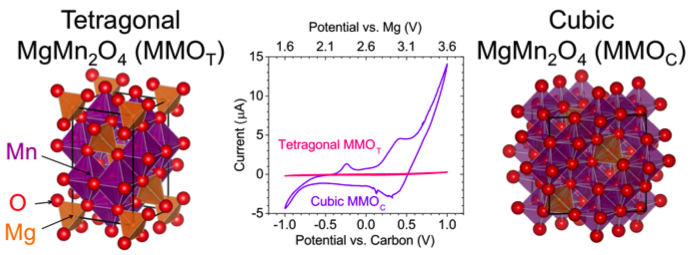
Phase-Controlled Electrochemical Activity of Epitaxial Spinel Thin films as Mg-Cathodes
Epitaxial thin film cathodes of MgMn2O4 (“MMO”) are stabilized in two distinct phases (tetragonal vs. cubic) which reveal distinct electrochemical activities for reversible Mg2+ insertion. Read More
-
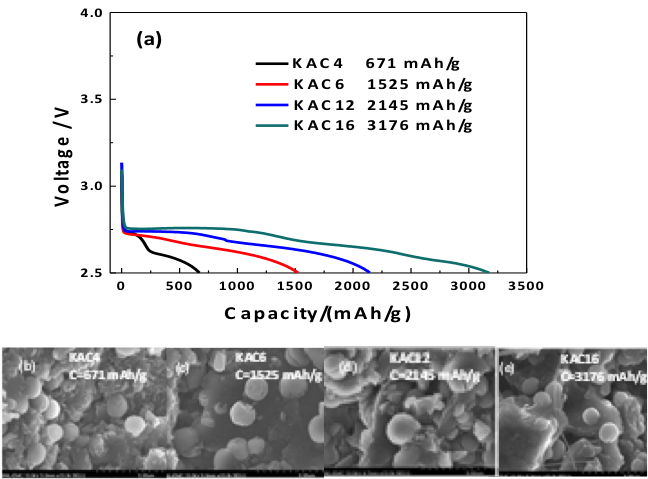
A Functional Impurity for Li-O2 Battery Cathode
Demonstrated that alkali metal can be used as a catalyst Li-O2 cell cathode design and opens the possibility of future optimization of functional K-doping in carbon cathode materials. Read More
-
Strategy of the Joint Center for Energy Storage Research to Influence Laboratory Safety Among its Funded Researchers
JCESR strives to influence safe science among its researchers. Read More
-
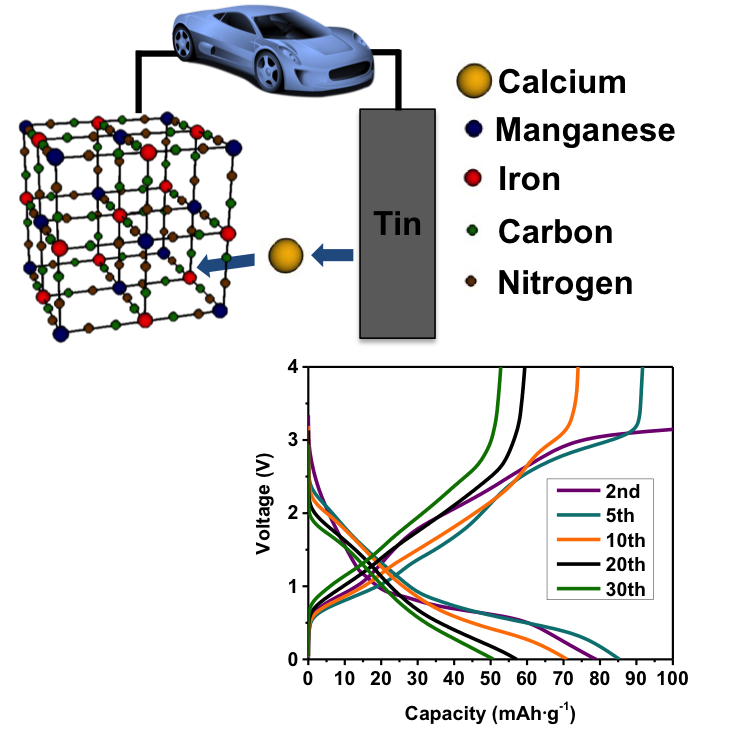
The First Ca-ion Rechargeable Battery
We have demonstrated the first ever rechargeable battery utilizing calcium as the working ion. Read More
-
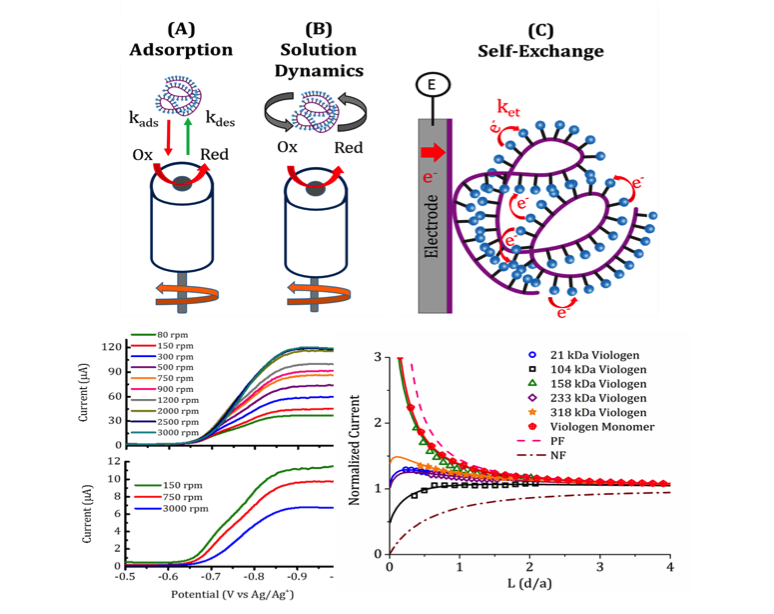
Investigation of Charge Transfer Mechanisms on Redox Active Polymers Using RDE and SECM
This study is a first step to evaluate rate determining processes on the electrochemical activity of RAPs. Using different electrochemical techniques we determined the presence of several preceding chemical steps. We also studied how the resulting RAP film mediates the charge transfer process and affects the electrochemical behavior of RAPs. Read More
-
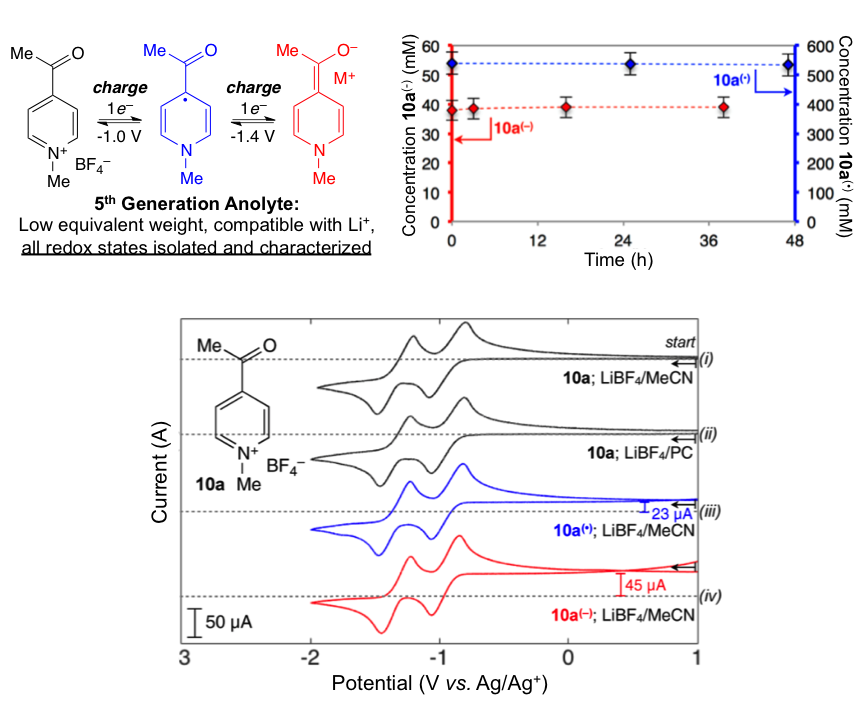
Evolutionary Design of Low Molecular Weight Organic Anolyte Materials for Applications in Nonaqueous Redox Flow Batteries
All-organic anolyte materials for nonaqueous redox flow batteries with high stability at all redox states were designed through an iterative study. Anolyte materials exhibit two chemically reversible reductions, have low equivalent weights of only 111 g/mol/e–, undergo reversible electro-chemistry in Li-ion supporting electrolytes, and are stable in solution for greater than 2 months without detectable decomposition. Read More
-
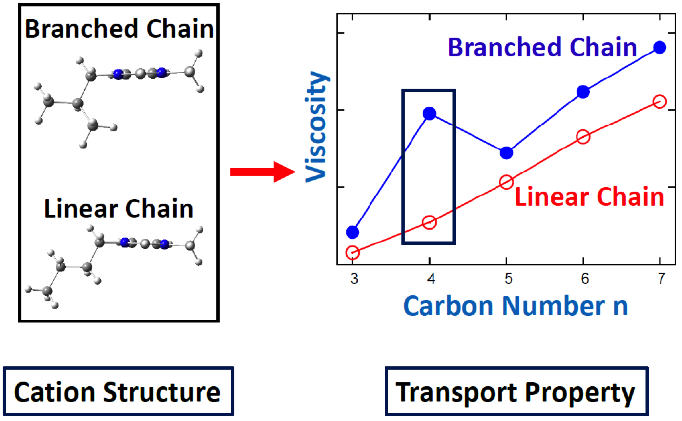
Molecular Topology and Local Dynamics Govern the Viscosity of Imidazolium-Based Ionic Liquids
A direct connection between molecular structure and liquid viscosity was made, and the latter was explained based on the former feature. Read More
-
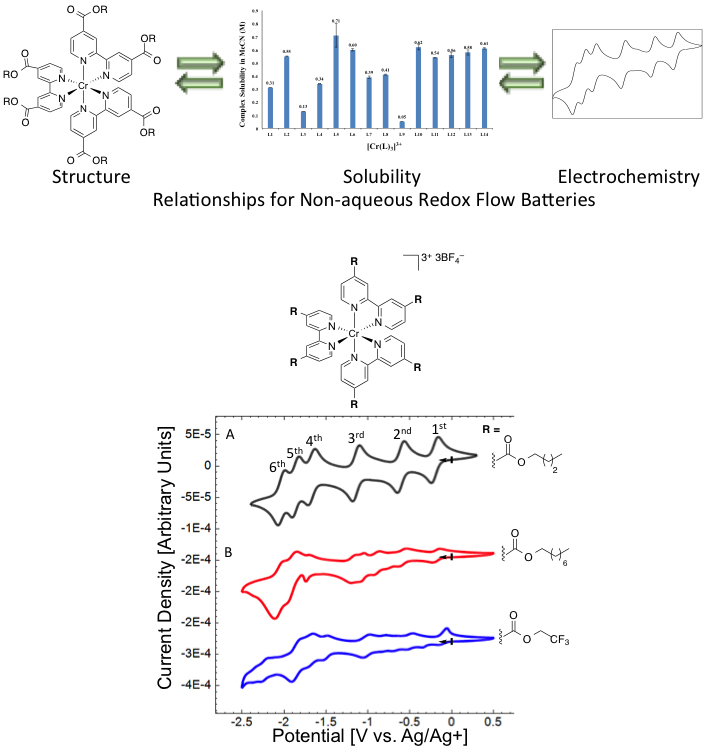
Evaluation of Tris-Bipyridine Chromium Complexes for Flow Battery Applications: Impact of Bipyridine Ligand Structure on Solubility and Electrochemistry
Synthesis of 24 Cr-bipyridine complexes across two different oxidation states; their solubility varies from insoluble to 0.7 M. Most of the complexes undergo reversible electrochemistry demonstrating up to six reversible redox couples. Read More
-
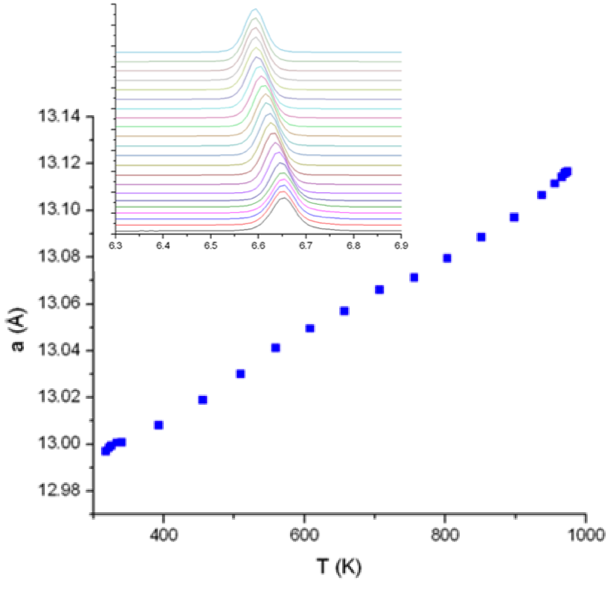
Thermal Expansion in the Garnet-Type Solid Electrolyte (Li7-xAlx/3)La3Zr2O12 as a Function of Al Content
The study identified the lattice expansion lithium lanthanum zirconium oxide (LLZ), which is an important lithium-metal stable solid-state Li-ion diffusion ceramic membrane as a function of Al-content. Read More
Latest Updates
-
You’re Invited - JCESR and Beyond: Translating the Basic Science of Batteries
Please join us at Argonne National Laboratory on Tuesday, April 4, 2023 for JCESR and Beyond: Translating the Basic Science of Batteries. Registration is now open. This in-person event will celebrate 10 years of research from the Joint Center… Read More
-
A Message from JCESR: In Memory of George Crabtree
It is with heavy hearts that we say goodbye to George Crabtree, a Senior Scientist and Distinguished Fellow at Argonne National Laboratory, and Director of the Joint Center for Energy Storage Research (JCESR), who passed away unexpectedly on January 23. Dr. Read More
-
Cyanopyridines As Extremely Low-Reduction-Potential Anolytes for Nonaqueous Redox Flow Batteries
Discovery of a cyanophenylpyridine derivative with a very low reduction potential and good stability during cycling. Read More
-
Characterizing Redoxmer – Electrode Kinetics Using a SECM-Based Spot Analysis Method
Identified asymmetries in electron transfer (ET) kinetics between the reduction and oxidation of ferrocene-based redoxmers by measuring the ET rate constants (kf/kb) as a function of electrode potential. Read More
-
Benzotriazoles as Low Potential Anolytes for Non-Aqueous Redox Flow Batteries
We developed an easy-to-synthesize benzotriazole-based anolyte with a high energy redox potential (-2.3 V vs Fc/Fc+) and high solubility that demonstrates stable electrochemical cycling performance. Read More

