Scientific Achievement
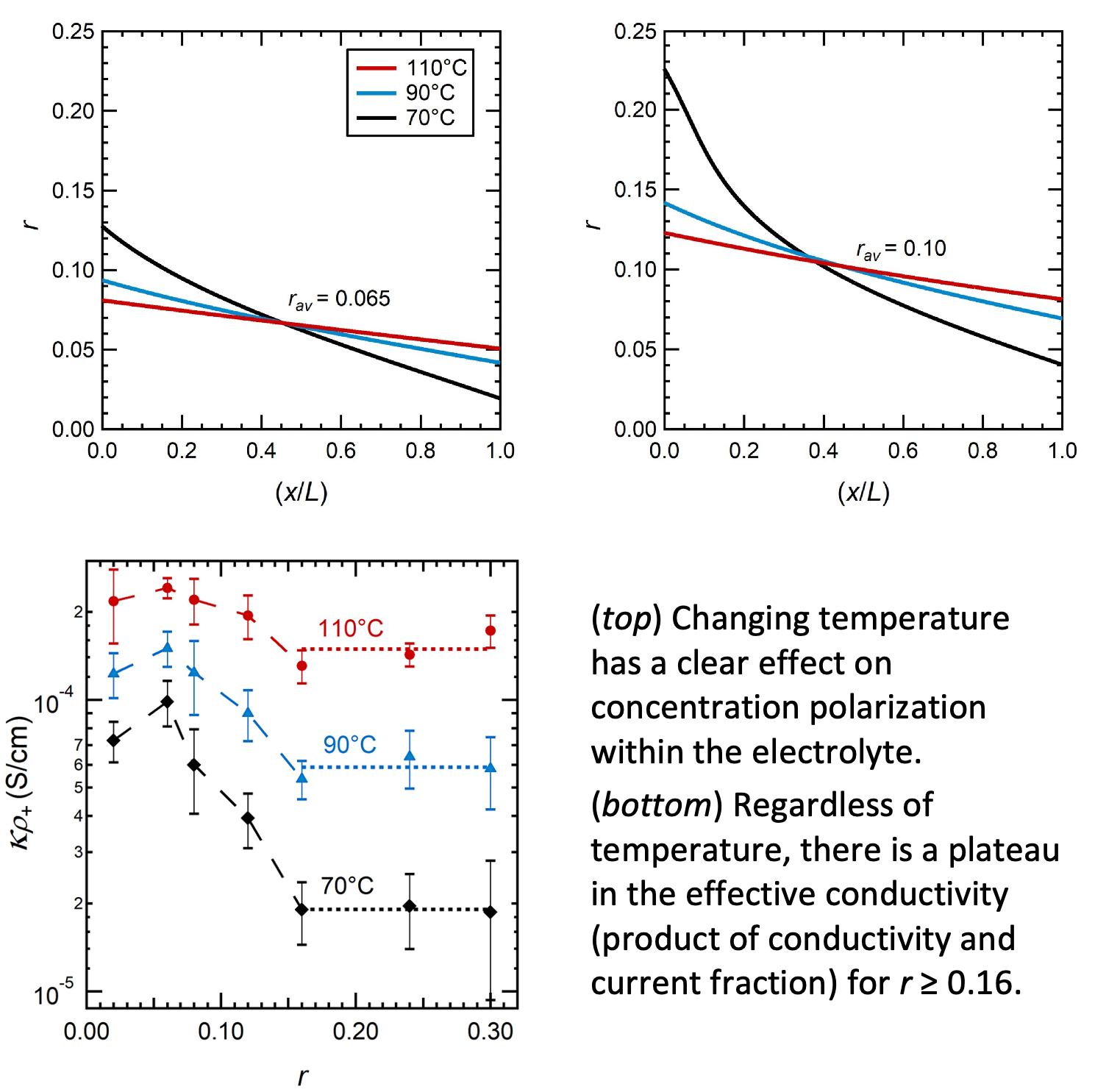
We measured the temperature dependence of ion transport properties in PEO/LITFSI electrolytes across a range of salt concentrations. Using these results, we were able to predict how concentration polarization changes with temperature.
Significance and Impact
Quantifying temperature effects on electrolyte properties is important to understand how a battery’s performance may change during fast charging and discharging during which the temperature of the battery may change drastically. Of particular importance is the observation of a plateau in effective conductivity at high salt concentrations.
Research Details
- AC Impedance spectroscopy was used to measure the electrochemical properties of PEO/LiTFSI electrolytes across a range of temperatures and concentrations
- Concentration gradients were modeled using concentrated solution theory and the measured transport properties.

