Redox Flow
-
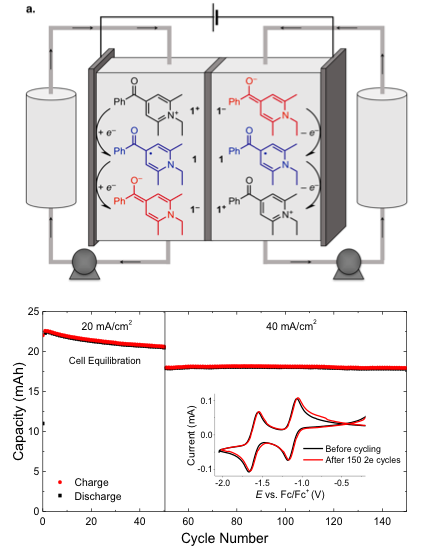
Multielectron Cycling of a Low Potential Anolyte in Alkali Metal Electrolytes for Non-Aqueous Redox Flow Batteries
High energy density organic redox flow batteries can only be realized when stable light-weight organic redox systems are developed, multi-electron redox molecules in combination with light-weight supporting electrolytes pave the way to this goal. Read More
-
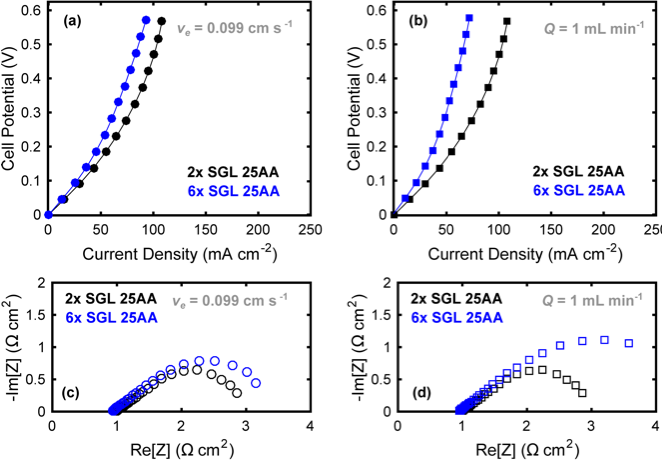
Towards Low Resistance Nonaqueous Redox Flow Batteries
Sources of resistive losses were experimentally minimized for a nonaqueous flow cell for various active species concentrations, electrolyte compositions, flow rates, separators, and electrode thicknesses. Read More
-
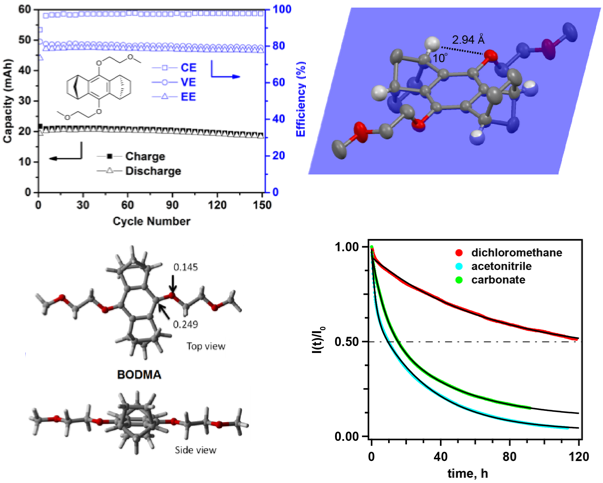
Annulated Dialkoxybenzenes as Catholyte Materials for Non-aqueous Redox Flow Batteries: Achieving High Chemical Stability Through Bicyclic Substitution
9,10-bis(2-methoxyethoxy)-1,2,3,4,5,6,7,8- octahydro-1,4:5,8-dimethanenoanthracene (BODMA) was developed for use as the catholyte in non-aqueous redox flow batteries. The bicyclic scaffolds prevent the ring-addition reaction, showing superior chemical stability in the charged state. A hybrid flow cell using this catholyte is operated for 150 charge-discharge cycles with a minimal loss of capacity. Read More
-
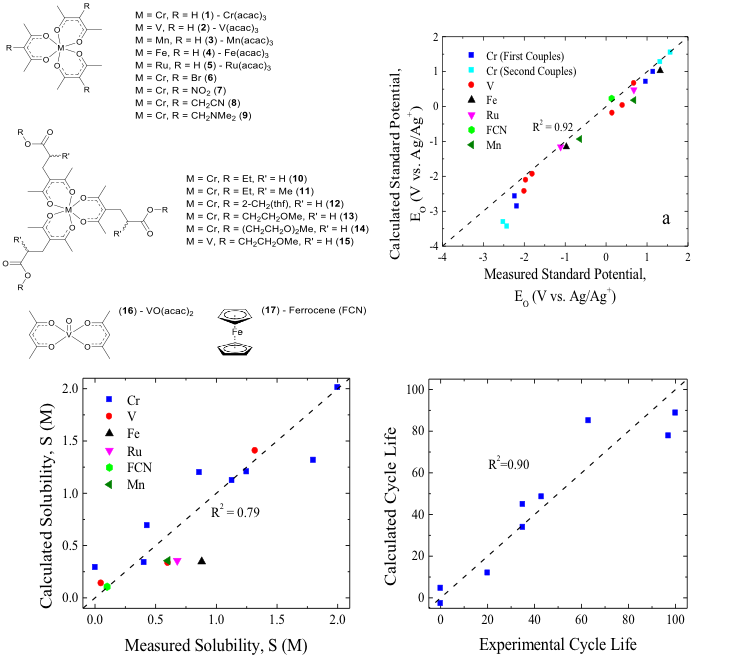
Predicting the Potentials, Solubilities and Stabilities of Metal-Acetylacetonates for Non-Aqueous Redox Flow Batteries Using Density Functional Theory Calculations
Density functional theory (DFT) was used to calculate key materials properties that were correlated with experimentally determined parameters that define the performance of redox flow battery (RFB) active materials. These include standard potentials, solubilities, and importantly stabilities. The correlations are for metal-acetylacetonate (acac) complexes, a promising class of actives, but could also be used for other materials. Read More
-
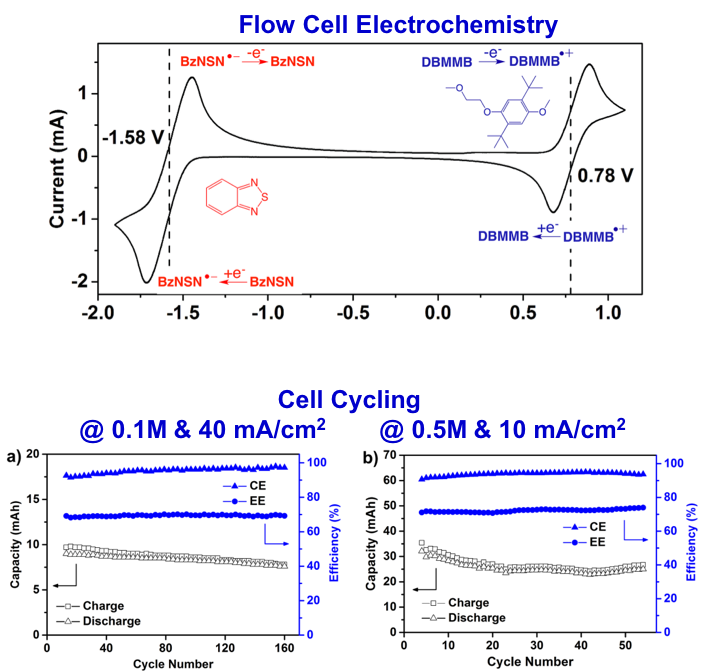
“Wine-Dark Sea” in an Organic Flow Battery: Storing Negative Charge in 2,1,3-Benzothiadiazole Radicals Leads to Improved Cyclability
Scientific Achievement 2,1,3-benzothiadiazole (BzNSN) was identified as a promising anolyte molecule for non-aqueous organic redox flow batteries. A proof-of-principle 2.36 V nonaqueous organic flow battery was developed by coupling BzNSN with DBMMB. Relatively stable performances over extended cycling were demonstrated under tested flow cell conditions,… Read More
-
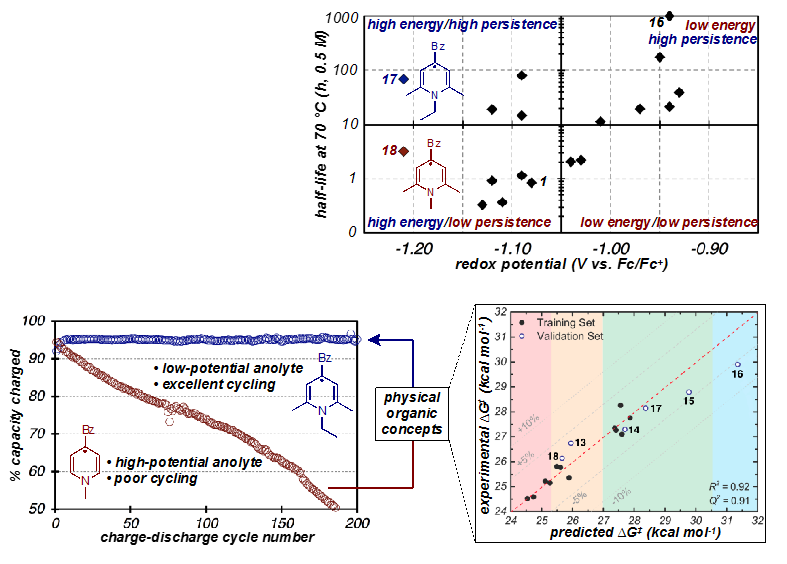
Physical Organic Approach to Persistent, Cyclable, Low-Potential Electrolytes for Flow Battery Applications
A rare example of a high energy (low potential) anolyte with excellent stability in the charged state was identified. This material undergoes cycling at potentials lower than other studied materials but does not show degradation even after 200 charge-discharge cycles. Predictive multidimensional analysis, a technique common to physical organic chemistry, was successfully applied to the development of a model for RFB electrolytes that predicts their stability in the charged state with high accuracy. Read More
-
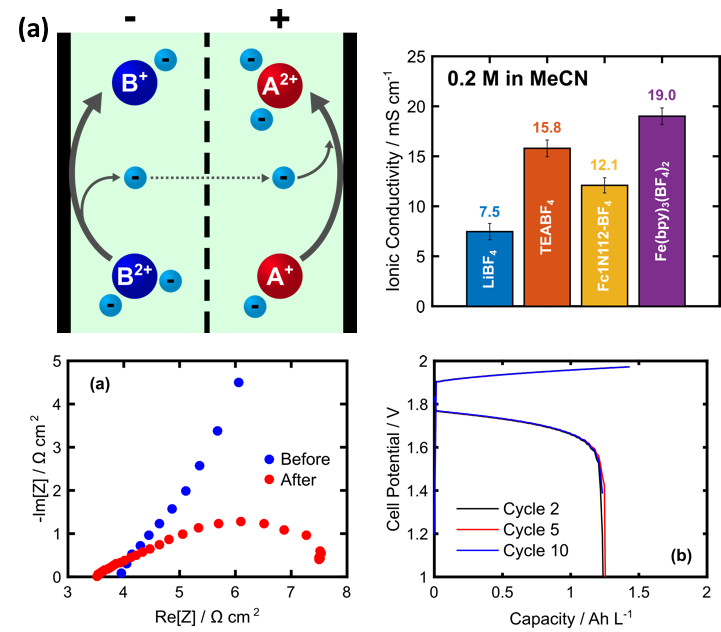
Feasibility of a Supporting Salt Free Non-aqueous Redox Flow Battery Utilizing Ionic Active Materials
We designed a non-aqueous flow battery that operates in the absence of supporting salt by utilizing redox active molecules that remain as ions across all accessible states of charge. Read More
-
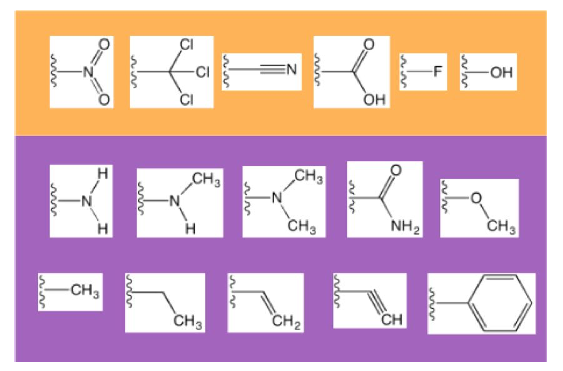
The Effects of Functional Groups in Redox-Active Organic Molecules: A High-Throughput Screening Approach
This work utilized high-throughput screening of thousands of molecules to explore the relationship between molecular structure and the reduction/oxidation potentials (ROPs) of organic electrolytes. Read More
-
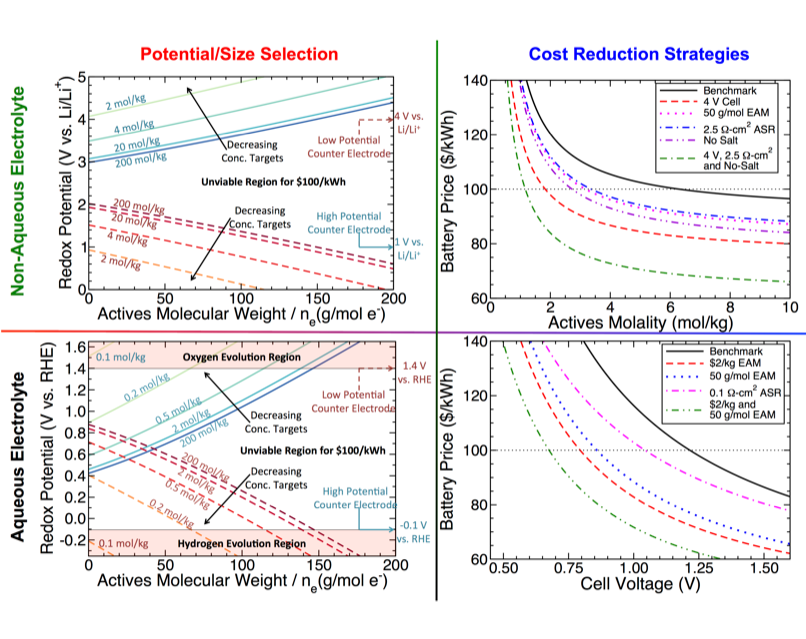
Cost-Driven Materials Selection Criteria for Redox Flow Battery Electrolytes
Techno-economic analysis shows that design challenges for non-aqueous batteries include minimizing salt content and dropping redox-active species concentration to moderate levels, while aqueous batteries are sensitive to only redox-active material cost and cell voltage due to low area-specific resistance and supporting electrolyte costs. Read More
-
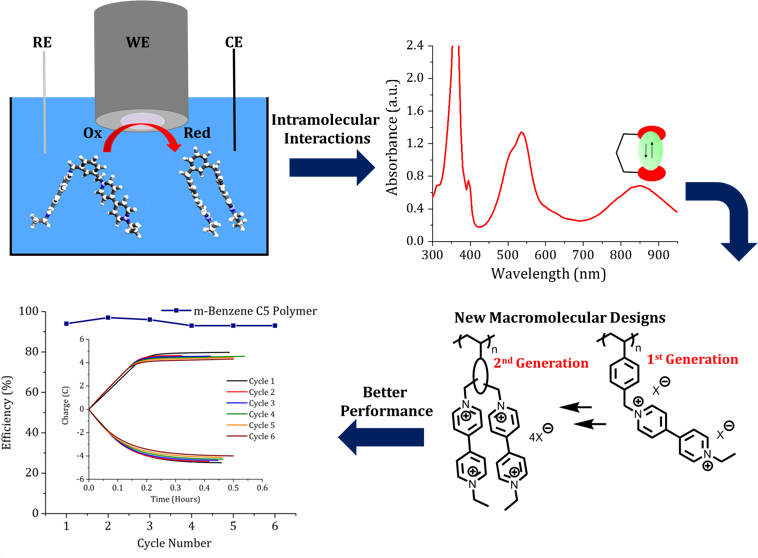
Impact of Backbone Tether Length and Structure on the Electrochemical Performance of Viologen RAPs
Fundamental inter-valence interactions are used as starting point for improving the electrochemical properties of redox active polymers and explore structure/activity relationships. Read More
Latest Updates
-
You’re Invited - JCESR and Beyond: Translating the Basic Science of Batteries
Please join us at Argonne National Laboratory on Tuesday, April 4, 2023 for JCESR and Beyond: Translating the Basic Science of Batteries. Registration is now open. This in-person event will celebrate 10 years of research from the Joint Center… Read More
-
A Message from JCESR: In Memory of George Crabtree
It is with heavy hearts that we say goodbye to George Crabtree, a Senior Scientist and Distinguished Fellow at Argonne National Laboratory, and Director of the Joint Center for Energy Storage Research (JCESR), who passed away unexpectedly on January 23. Dr. Read More
-
Cyanopyridines As Extremely Low-Reduction-Potential Anolytes for Nonaqueous Redox Flow Batteries
Discovery of a cyanophenylpyridine derivative with a very low reduction potential and good stability during cycling. Read More
-
Characterizing Redoxmer – Electrode Kinetics Using a SECM-Based Spot Analysis Method
Identified asymmetries in electron transfer (ET) kinetics between the reduction and oxidation of ferrocene-based redoxmers by measuring the ET rate constants (kf/kb) as a function of electrode potential. Read More
-
Benzotriazoles as Low Potential Anolytes for Non-Aqueous Redox Flow Batteries
We developed an easy-to-synthesize benzotriazole-based anolyte with a high energy redox potential (-2.3 V vs Fc/Fc+) and high solubility that demonstrates stable electrochemical cycling performance. Read More

