Scientific Achievement
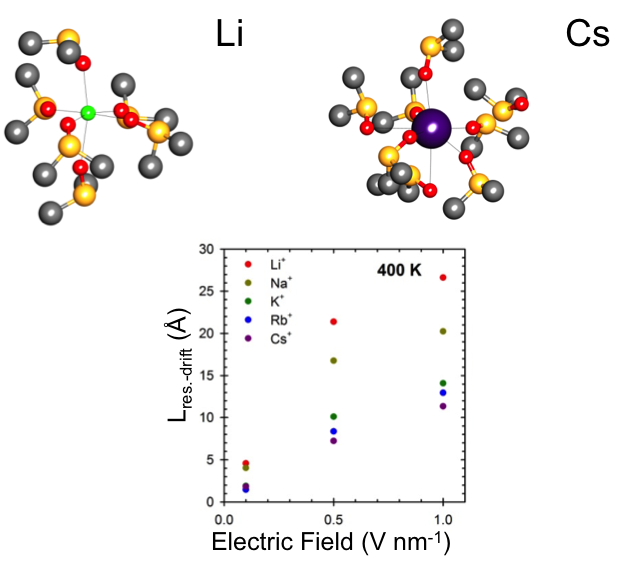
To predict the behavior of battery electrolyte components, we investigated the role of exogenous electric fields on the solvation structure and dynamics of alkali ions in dimethyl sulfoxide (DMSO).
Significance and Impact
Although exogenous electric fields induce a drift displacement, their presence does not significantly affect the random diffusive displacement of alkali ions in DMSO. Temperature is found to generally have a stronger influence on dynamical properties, such as the DMSO exchange dynamics and the ion mobilities.
Research Details
- Pulsed field gradient nuclear magnetic resonance spectroscopy was used to quantify the dynamics of solvent molecules and ions in solution via determination of self-diffusion coefficients.
- Molecular dynamics simulations explored the interplay between electric fields and temperature and their effects on the solvation structure and dynamics of alkali ions in DMSO.
Work performed at Pacific Northwest National Laboratory (JCESR partner) by S.N. Kerisit, M. Vijayakumar, K.S. Han, and K.T. Mueller, J. Chem. Phys., 2015, 142, 224502.
DOI: 10.1063/1.4921982

