Scientific Achievement
This study demonstrates a comprehensive computational approach to predicting multiple electrode/electrolyte interfacial properties in solid-state batteries.
Significance and Impact
The wettability and the electrochemical window at interfaces between electrodes and solid electrolytes are shown to be strongly impacted by chemical interactions at these interfaces. This suggests a potential trade-off between wettability and reductive stability.
Research Details
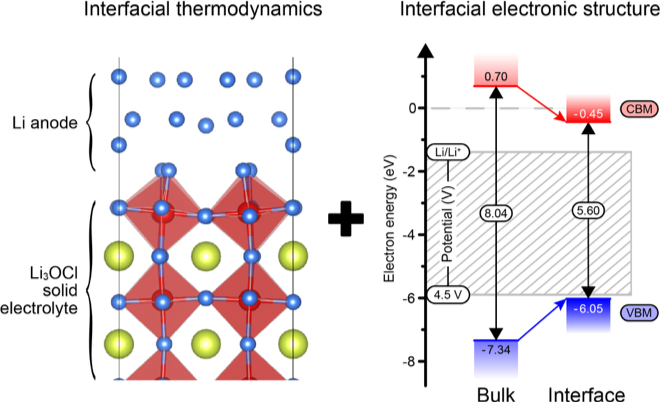
- DFT calculations were used to predict several properties of relevance for battery performance at a model interface between a Li3OCl solid electrolyte and a Li-metal anode.
- The most stable interface was predicted to be oxygen-terminated. Additional calculations of the interfacial work of adhesion and application of the Young-Dupree equation found that Li will strongly wet Li3OCl, suggesting the possibility for low interfacial resistance.
- The strong interfacial interaction narrows the band gap of Li3OCl significantly (30%) in the vicinity of the interface, potentially degrading its electrochemical stability. Nevertheless, the large bandgap of Li3OCl and the high-energy of its conduction band minimum implies that stability against reduction by Li metal is maintained overall.

