Scientific Achievement
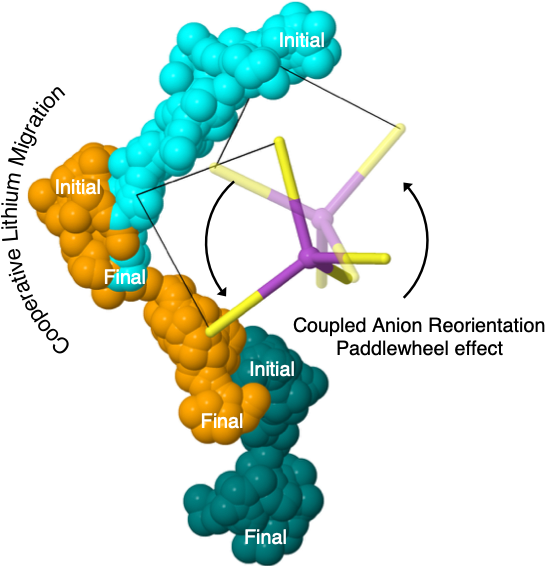
The high conductivity exhibited by glassy sulfide solid electrolytes is demonstrated to arise from the dynamic coupling of cation migration events with anion reorientations.
Significance and Impact
Glasses are promising electrolytes for solid-state lithium batteries. However, due to their amorphous structure, the mechanisms that give rise to their ionic conductivity remain poorly understood. Here, atomic-scale modeling reveals that lithium migration at practical temperatures is aided by simultaneous, quasi-permanent rotations of complex (PS43-) anions – a direct observation of the ‘paddlewheel’ effect. These observations suggest that glasses containing complex anions, and in which covalent network formation is minimized, may be fertile ground in the search for new solid electrolytes.
Research Details
- ab initio molecular dynamics was used to model the prototype glass, 75Li2S–25P2S5
- Paddlewheel contributions to Li mobility were confirmed by analyzing spatial, temporal, vibrational, and energetic correlations with anion rotations
- The dynamics in the glass differ from those in the crystalline analogue, γ-Li3PS4, where anion reorientations are negligible and ion mobility is suppressed

