Latest Updates
-
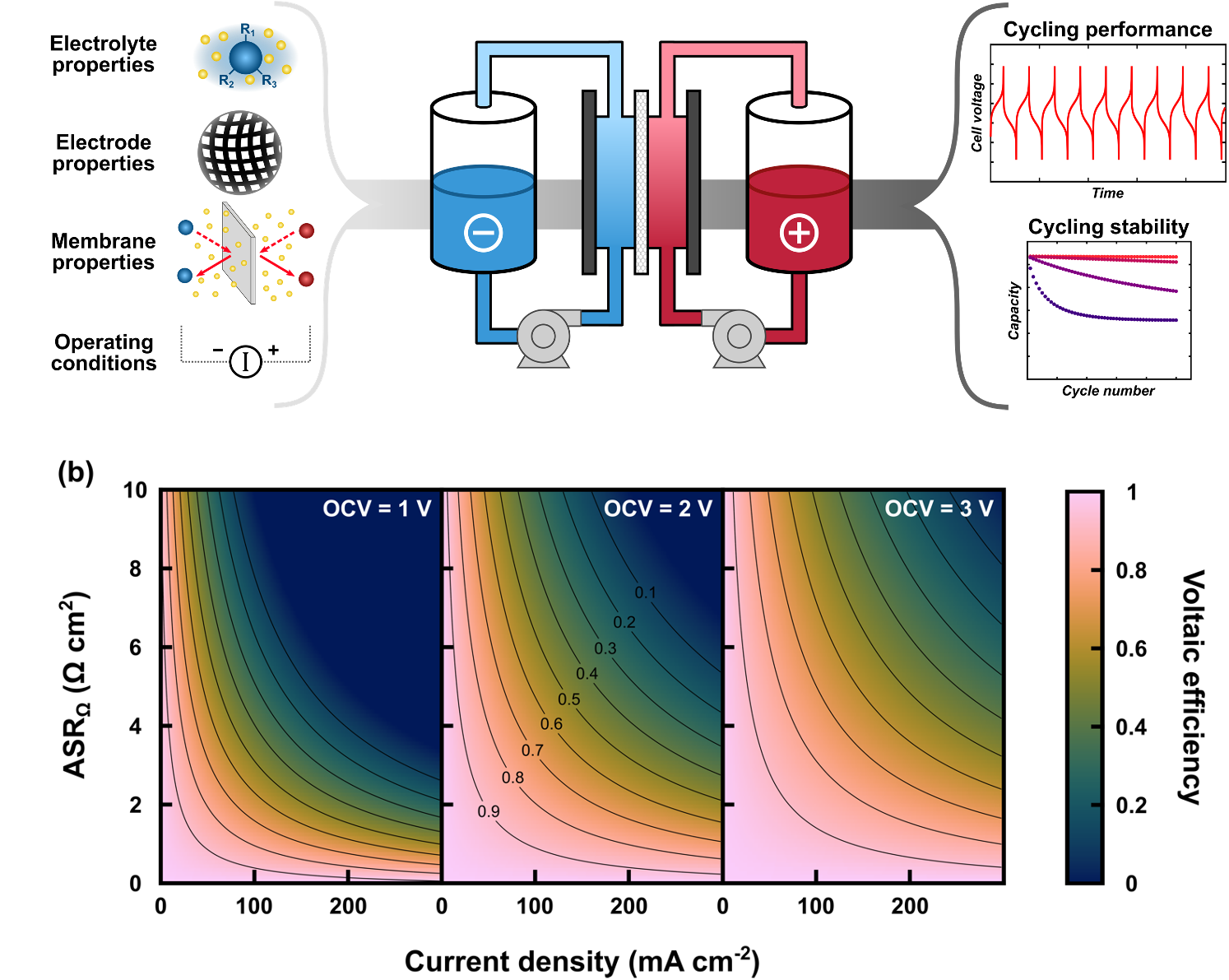
Connecting material properties and redox flow cell cycling performance through zero-dimensional models
We derived a zero-dimensional, analytical model for describing mass balances and cell voltages in redox flow batteries (RFBs), enabling direct connections between material / electrolyte properties, cell operating conditions, and resulting performance metrics (e.g., energy efficiency, capacity fade). Read More
-
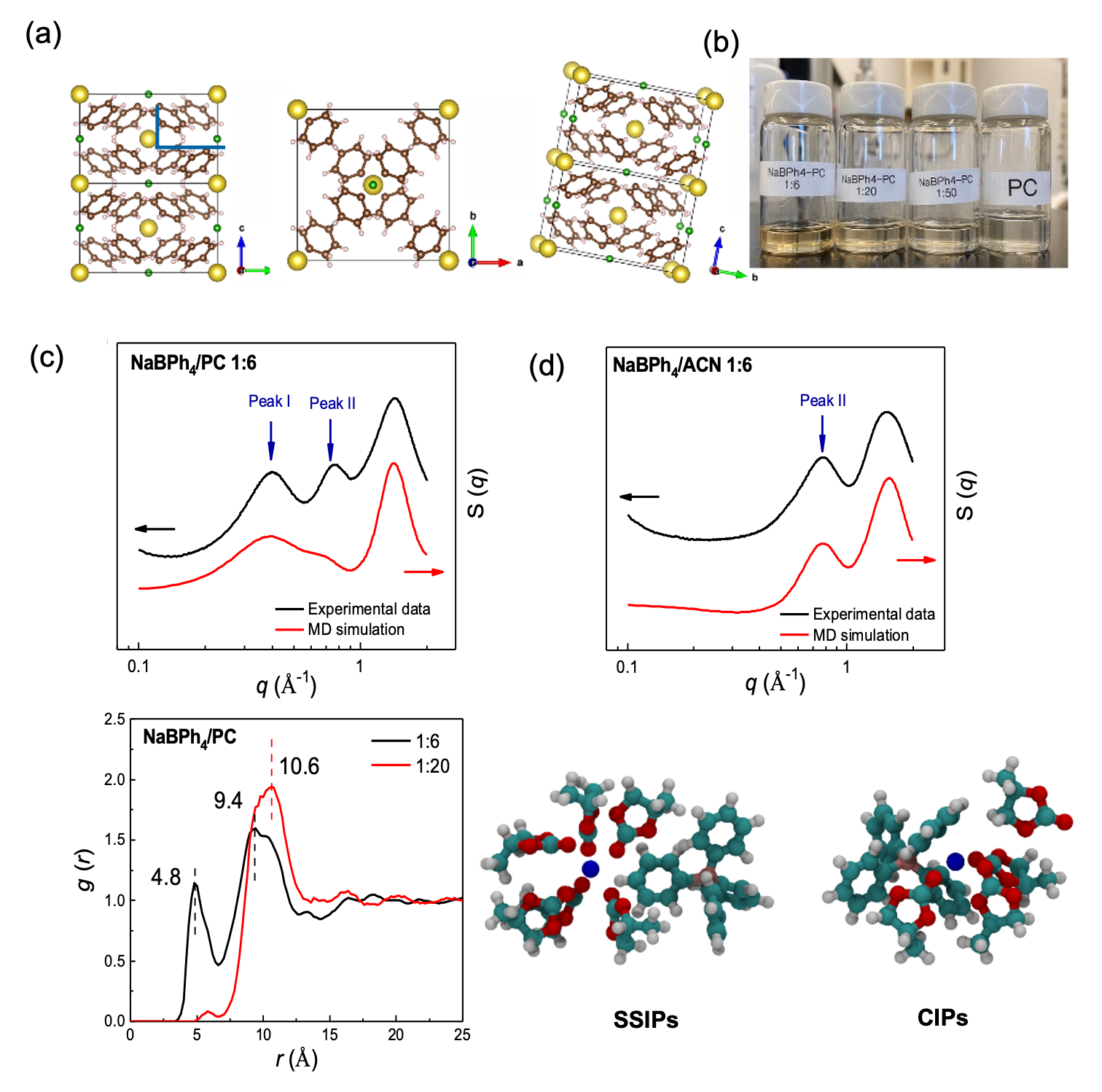
Understanding fluorine-free electrolytes via small-angle X-ray scattering
We compare the solvation phenomenon of sodium tetraphenylborate (NaBPh4) salt dissolved in organic solvents of propylene carbonate (PC), 1,2-dimethoxyethane (DME), acetonitrile (ACN) and tetrahydrofuran (THF) by SAXS/WAXS measurement and MD simulation. Read More
-
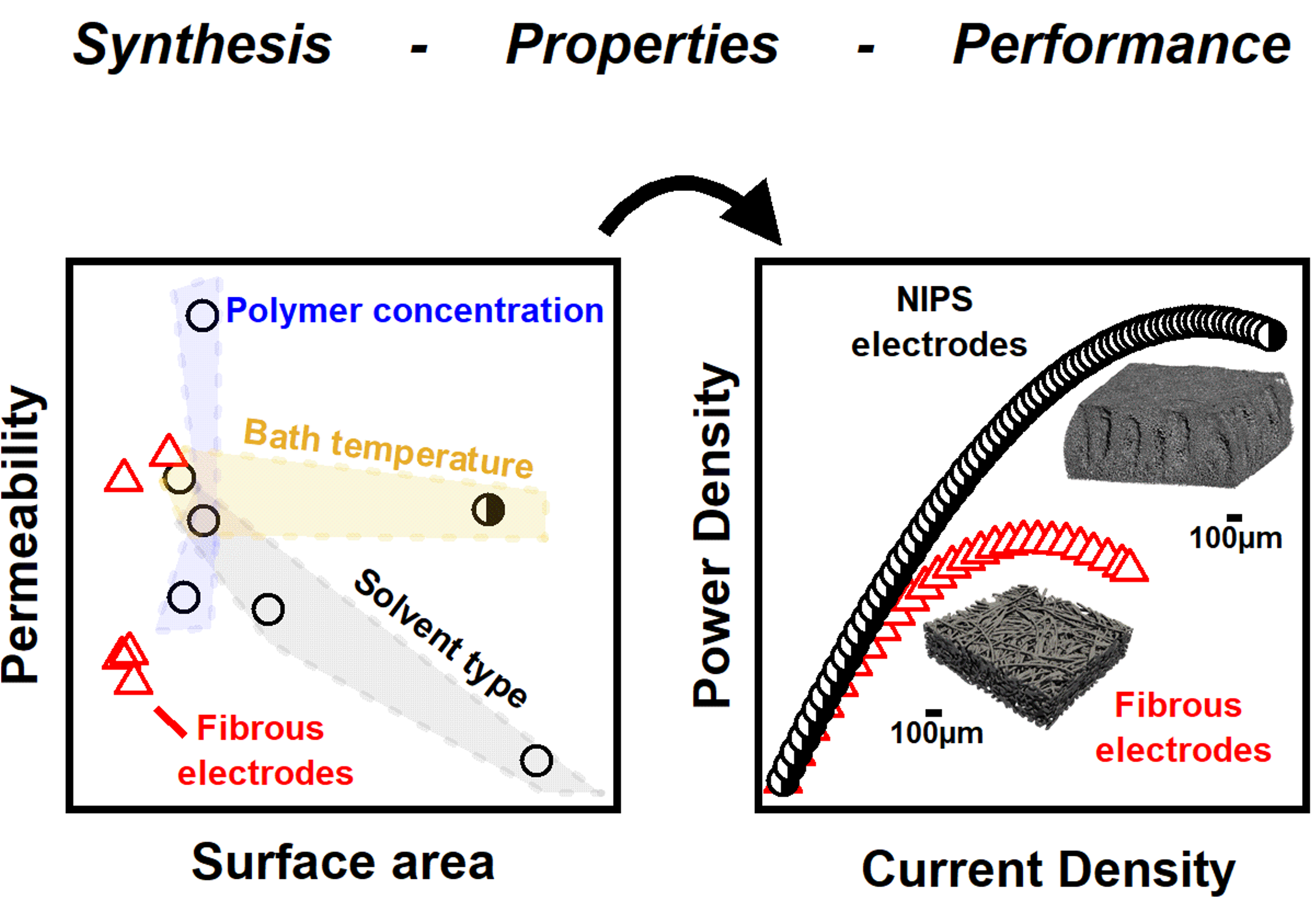
Microstructural engineering of high-power redox flow batteries via non-solvent induced phase separation
Non-solvent induced phase separation (NIPS) is used to develop electrode synthesis-structure-performance relationships, and select samples are incorporated in redox flow batteries (RFBs) which exhibit high power density. Read More
-
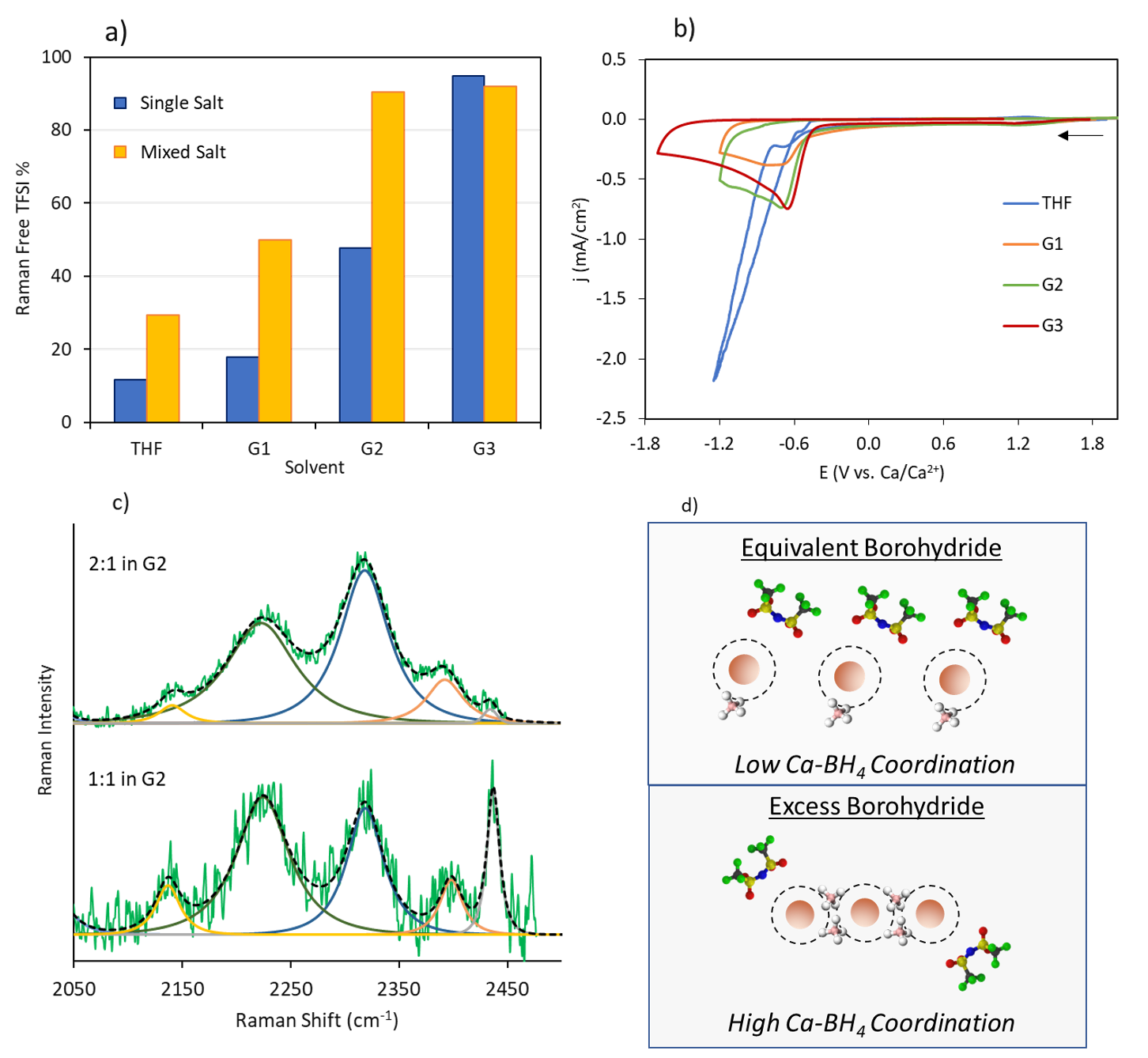
Efficacy of Stabilizing Calcium Battery Electrolytes through Salt-Directed Coordination Change
The factors determining the extent to which salt anions can be stabilized during Ca plating by eliminating their coordination with Ca2+ through co-salt addition were elucidated. Contrary to expectation, the exemplar bis(trifluoromethylsulfonyl)imide (TFSI-) anion is unstable whether in the coordinated or free state. Instead, the type of Ca2+ coordination structure formed with the co-salt anion determines whether Ca deposition can be achieved. Read More
-
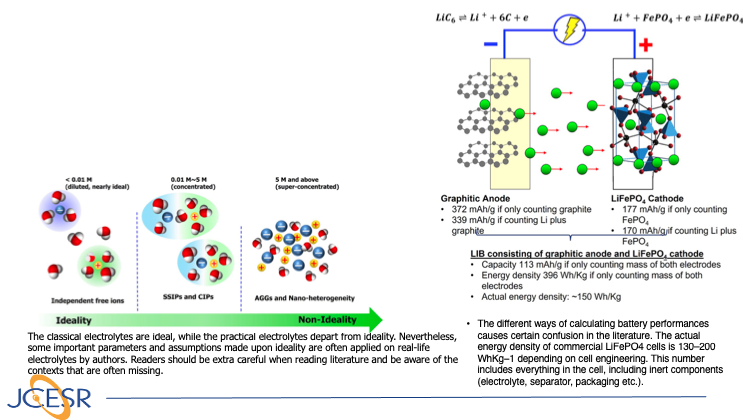
Navigating the Minefield of Battery Literature
This is an invited perspective aiming to help researchers new to the field of battery research to circumvent certain recurring misconceptions and inaccuracies in the current battery literature. It covers the electrolyte ideality and practical situation in batteries, the difficulty in accurately determining ion transference… Read More
-
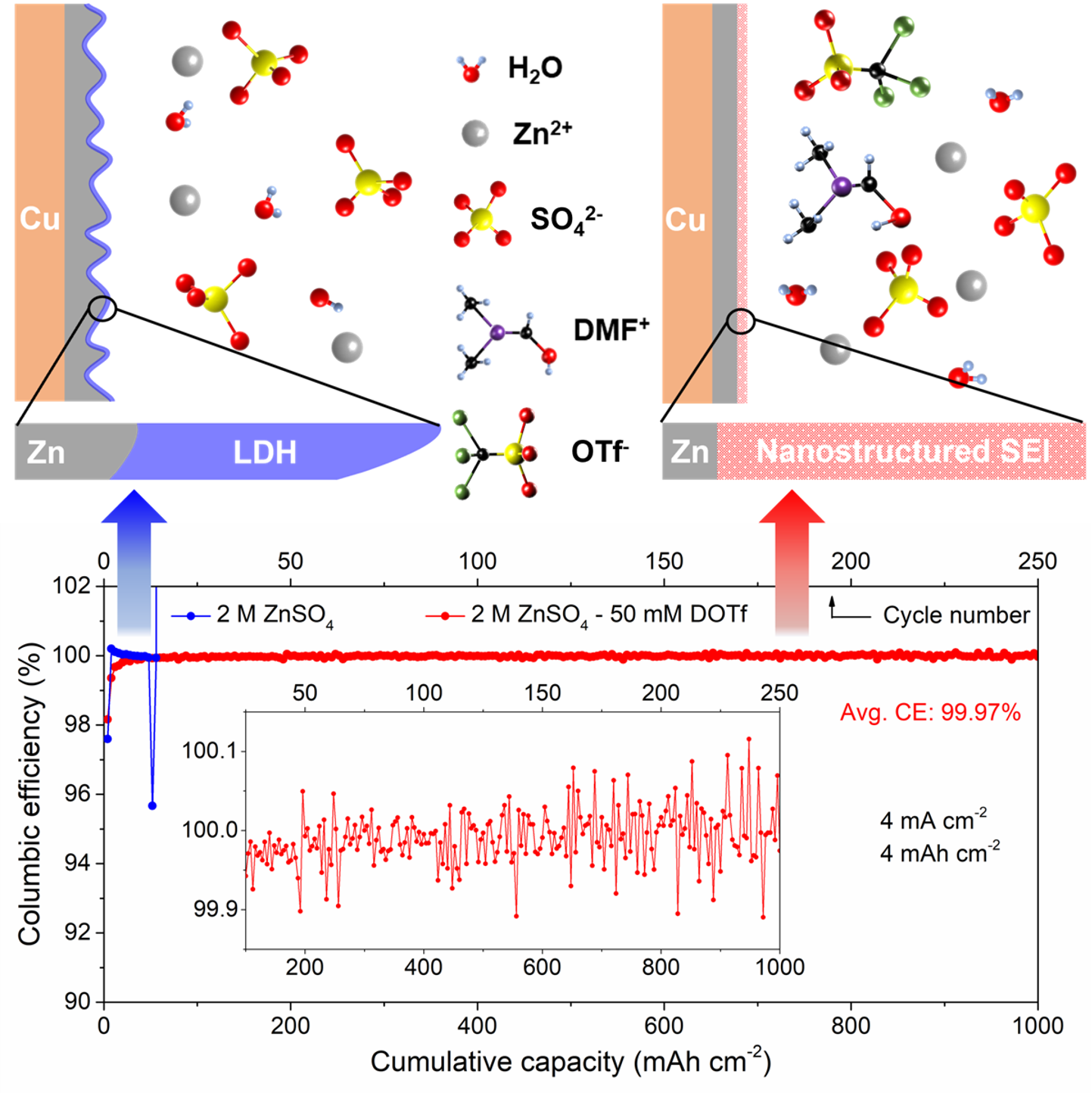
Highly reversible Zn anode with a practical areal capacity enabled by a sustainable electrolyte and superacid interfacial chemistry
We introduced - N,N-dimethylformamidium trifluoromethanesulfonate (DOTf) - as a novel low-concentration electrolyte additive for aqueous Zn metal batteries. This electrolyte leads to dendrite-free and highly reversible Zn plating/stripping with close-to-100% average CE at practical cycling conditions (current density of 4 mA cm-2 and areal capacity of 4 mAh cm-2) with long cycle life. Read More
-
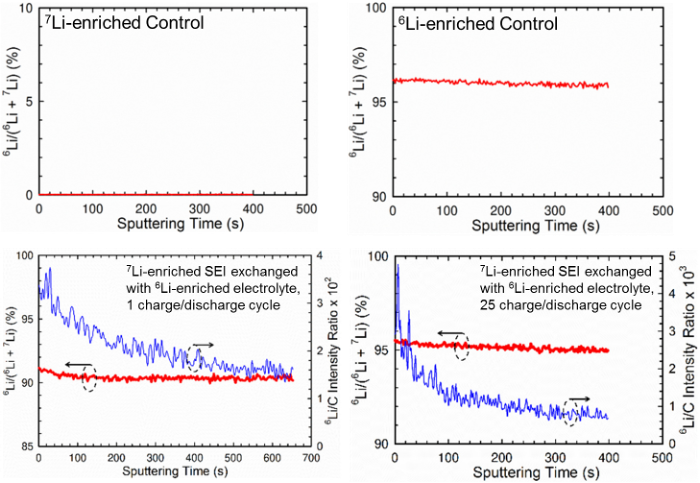
Quantifying Lithium Ion Exchange in Solid Electrolyte Interphase (SEI) on Graphite Anode Surfaces
By using Li isotopic labelling of SEIs and electrolytes followed by time-of-flight secondary-ion mass spectroscopy and solid-state NMR analyses, we found that the majority of Li+ “immobilized” in the chemical ingredients were exchanged after 1 SEI formation cycle. Ion exchange by diffusion based on concentration gradient without applied potential also occurred simultaneously. Read More
-
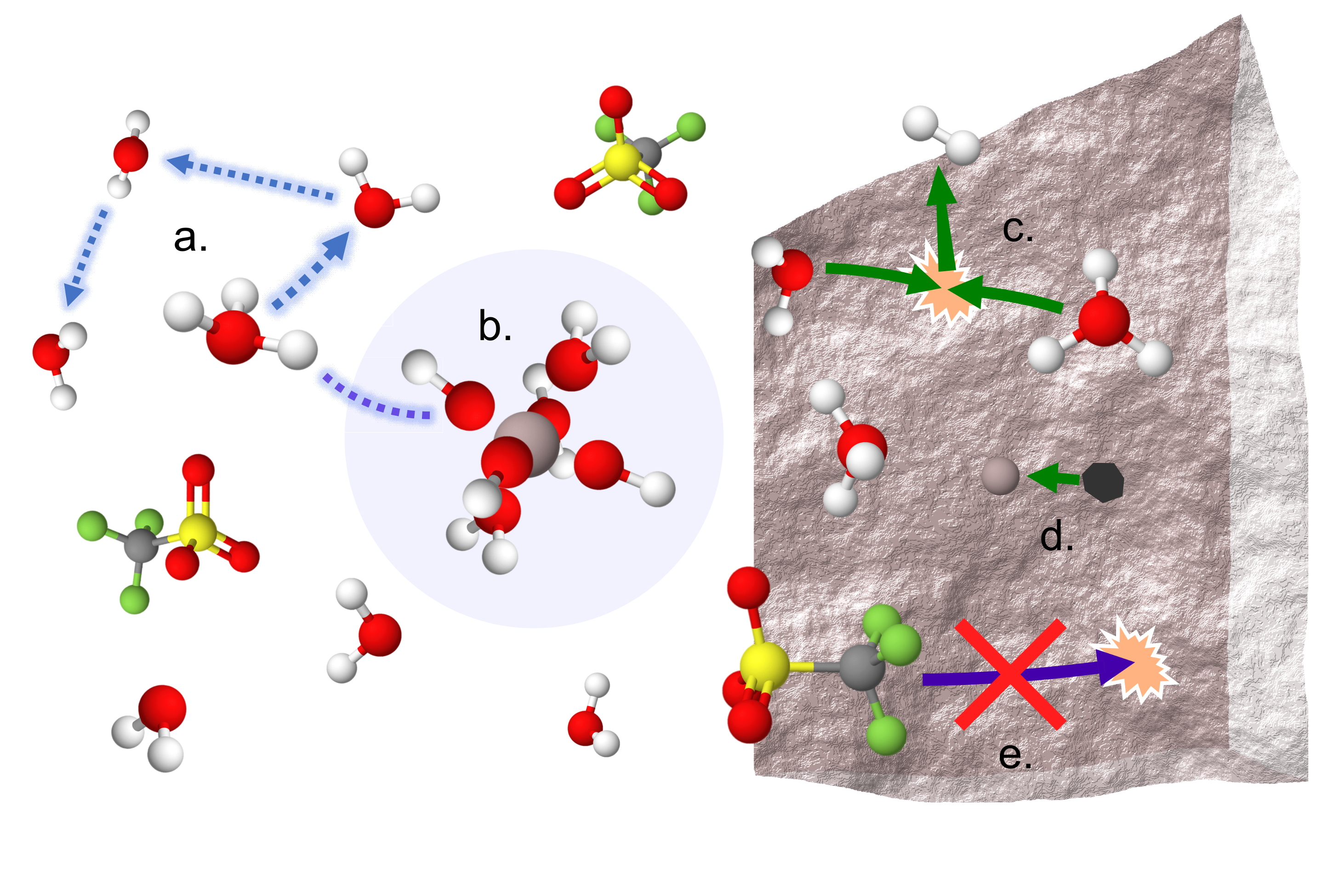
A Sobering Examination of the Feasibility of Aqueous Aluminum Batteries
We revealed the first compelling evidence for a dynamic octahedral solvation structure around Al3+ dominated by labile water and OH-, without Al-OTf contact ion pairs, at high salt concentrations. High proton activity is observed in transport and electrochemical measurements which relates well with the proposed solvation environment. Read More
-
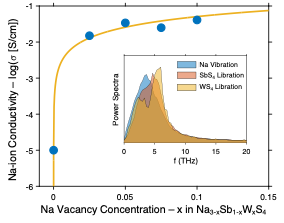
Ion Migration Mechanisms in the Sodium Sulfide Solid Electrolyte Na3-xSb1-xWxS4
The atomic-scale mechanisms that underlie the exceptionally high ionic conductivity of Na3-xSb1-xWxS4 are elucidated. The conductivity is well explained by a combination of vacancy-related effects and a strong overlap of cation vibrational modes with anion librations. Read More
-
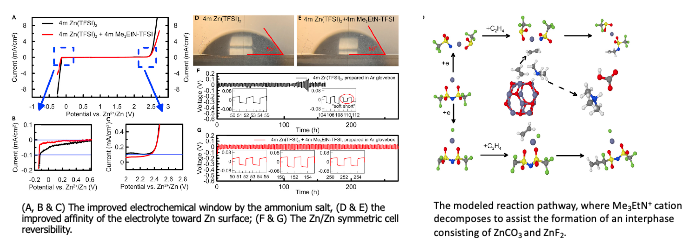
Ammonium enables reversible aqueous Zn battery chemistries by tailoring the interphase
As a continued effort to make Zn metal anode reversible at lower cost, researchers at ARL explored a new supporting salt based on ammonium salt to alter the interfacial structure and interphasial chemistries. The salt was synthesized at ARL and much cheaper than the phosphonium… Read More
-
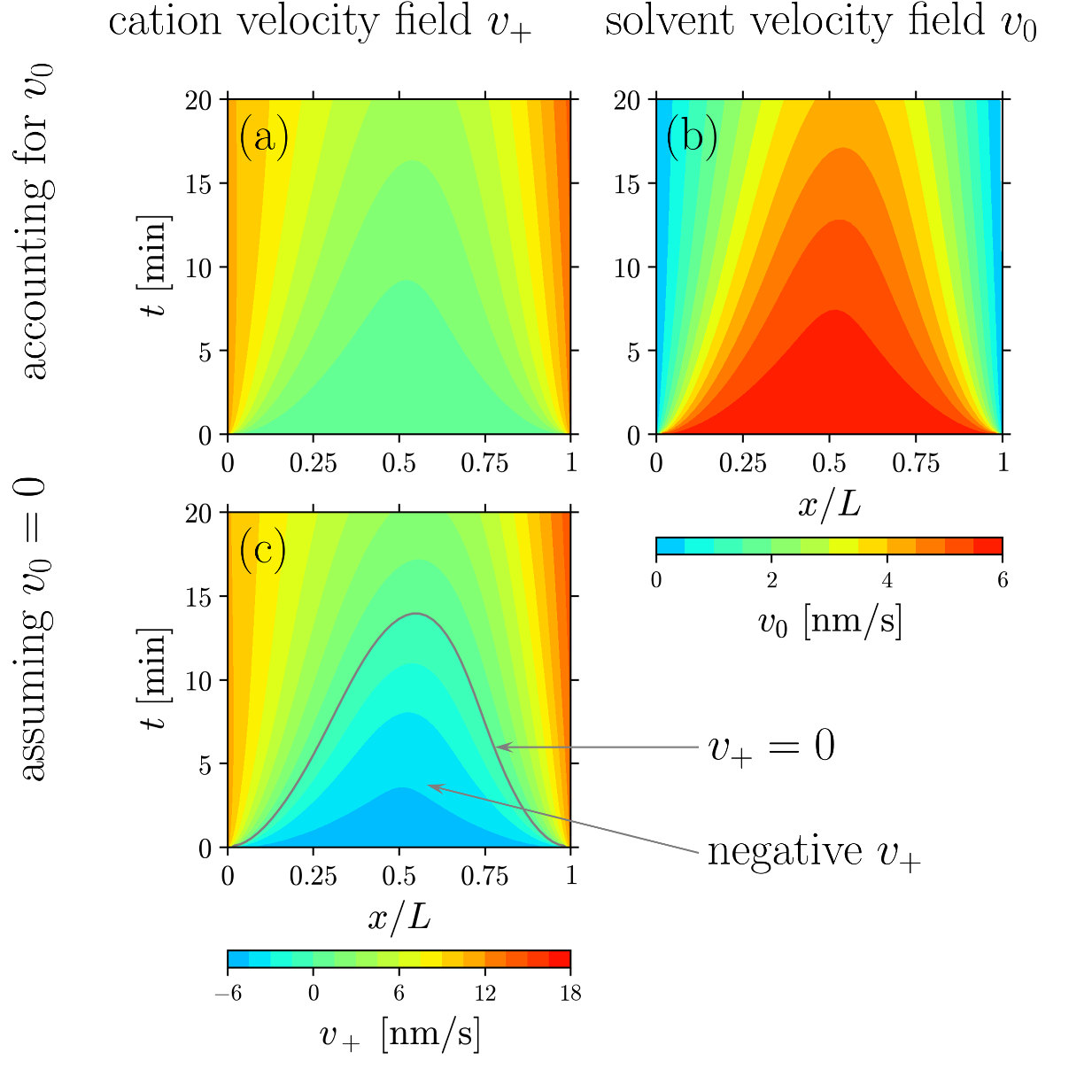
Effect of Solvent Motion on Ion Transport in Electrolytes
Typically, the solvent is assumed to be immobile in a polarized electrolyte. However, recent JCESR measurements show non-zero solvent velocity. This article proposes a continuum theory to predict such solvent motion and its influence on other electrolyte fields. Read More
-
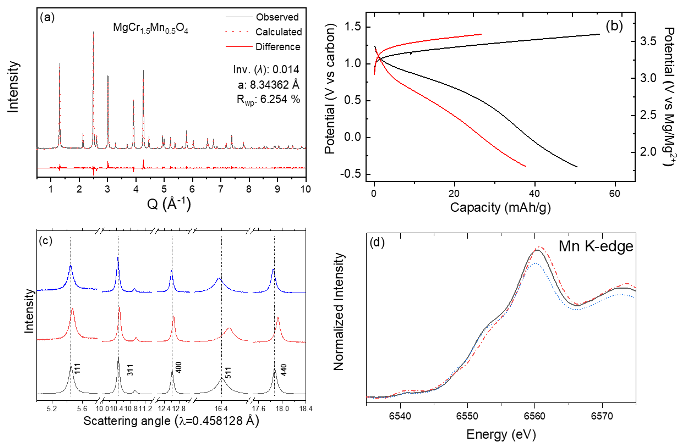
Facile Electrochemical Mg-Ion Transport in a Defect-Free Spinel Oxide
Inversion-free spinel MgCr1.5Mn0.5O4 was successfully synthesized. The tailored spinel showed reversible (de)intercalation of Mg2+ at high redox potentials. It was found that the overpotentials and, thus, overall hysteresis was reduced when the inversion ratio in the spinel lattice was minimized. The experimental evidence emphasizes the influence of structural defects, in this case inversion, on electrochemical Mg2+ activity and provides a design rule toward a building functional Mg cathode for a high-energy Mg-ion battery. Read More
-
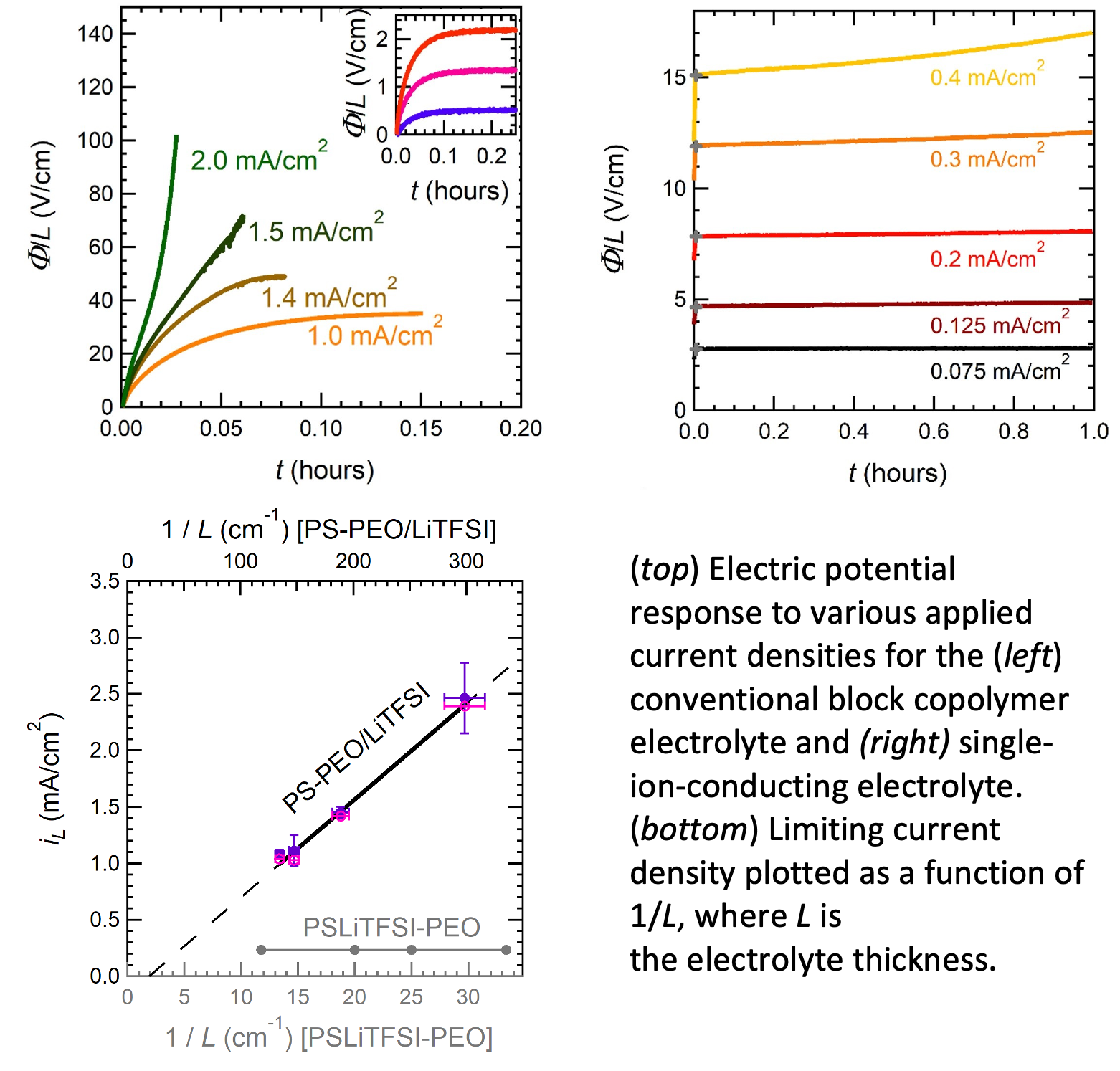
Limiting Current in Single-Ion-Conducting and Conventional Block Copolymer Electrolytes
The performance of an electrolyte in a battery is determined by the limiting current – the maximum allowable current before detrimental irreversible side-reactions take over. We have developed a methodology for determining limiting current in electrolytes with mobile cations and anions (conventional liquid electrolytes) and electrolytes with only one mobile ion (single-ion conductors such as inorganic ceramics and glasses). Read More
-
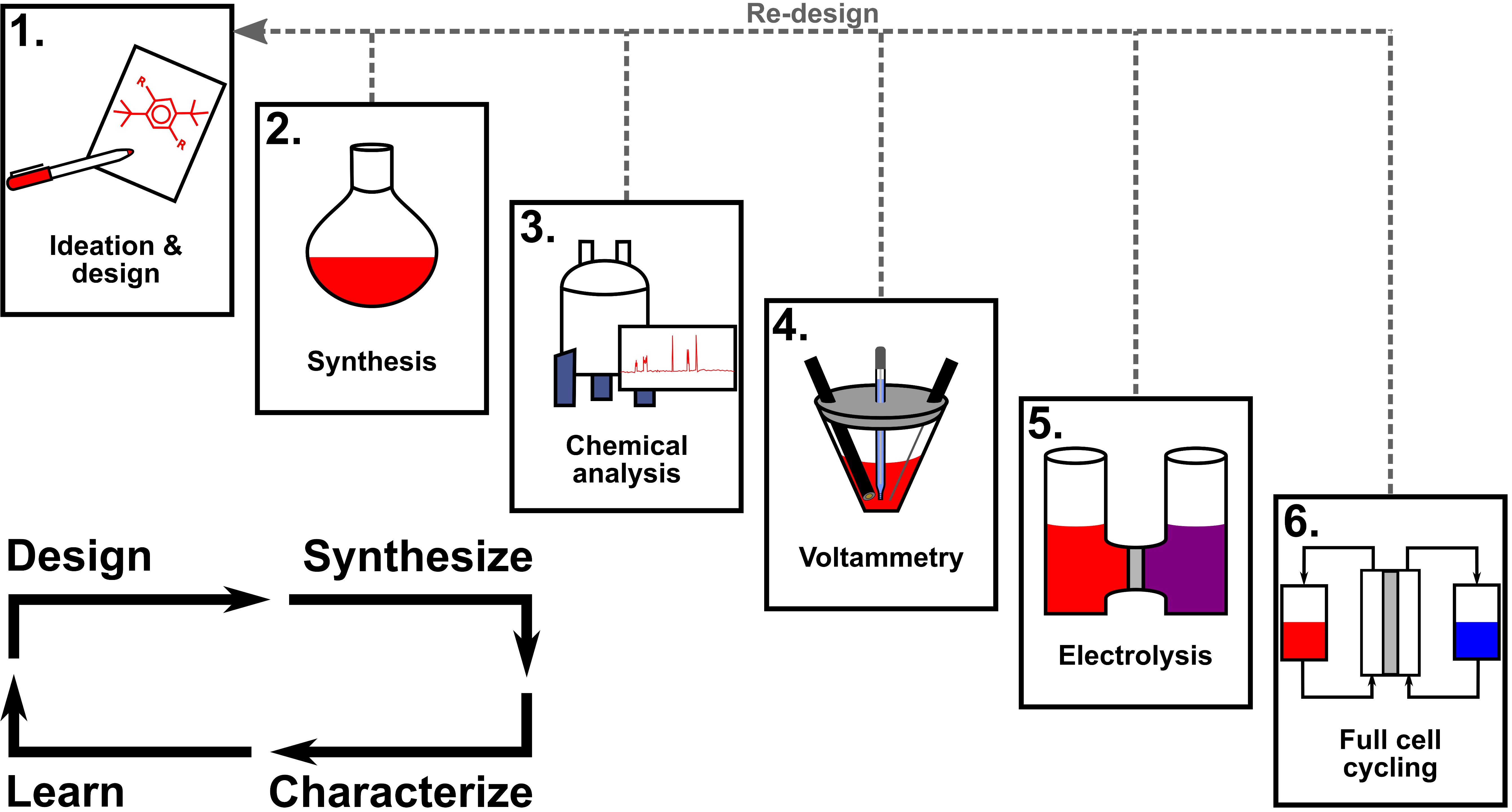
From the synthesis vial to the full cell: Electrochemical methods for characterizing active materials for redox flow batteries
We describe foundational electrochemical methods in the context of redox flow batteries, discussing proper experimental methodology, data interpretation, and limitations for evaluating key properties that translate to device performance. Read More
-

Ethylene oxide functionalization enhances the ionic conductivity of a MOF
A synergistic approach, combining methods known to enhance solid-state conductivity, is applied to a MOF resulting in a true solid-state lithium conducting MOF system with ionic conductivity similar to reported MOF/Li salt/solvent electrolytes. Read More
-

A Nonaqueous Redox-Matched Flow Battery with Charge Storage in Insoluble Polymer Beads
Demonstration of a “redox-matched” flow battery. Read More
-

A Comparative Study of Compressive Effects on the Morphology and Performance of Carbon Paper and Cloth Electrodes in Redox Flow Batteries
We systematically investigate the performance of carbon paper and cloth electrodes, which are of interest for redox flow batteries, as a function of compression. Read More
-

Understanding the Solvation-Dependent Properties of Cyclic Ether Multivalent Electrolytes Using High Field NMR and Quantum Chemistry
The influences of cation size and cyclic ether solvent structure on multivalent electrolyte solvation structure and dynamics were elucidated through theory and experiment. Read More
-

Exploration of reduced graphene oxide microparticles as electrocatalytic materials in vanadium redox flow batteries
Reduced graphene oxide (rGO) microparticles with nearly identical chemical composition but different surface areas and pore structures are evaluated in vanadium redox flow batteries (VRFBs). Read More
-

Austen Angell’s Legacy in Electrolyte Research
This is an article to remember the late Prof Austen Angell (1933~2021) Prof Angell was the guru in non-crystalline solid materials, an authority in glass-transition phenomenon. His brief and tangential interest in battery electrolytes in 1990s discovered the abnormal ion-solvation behavior at high concentrations, which… Read More

