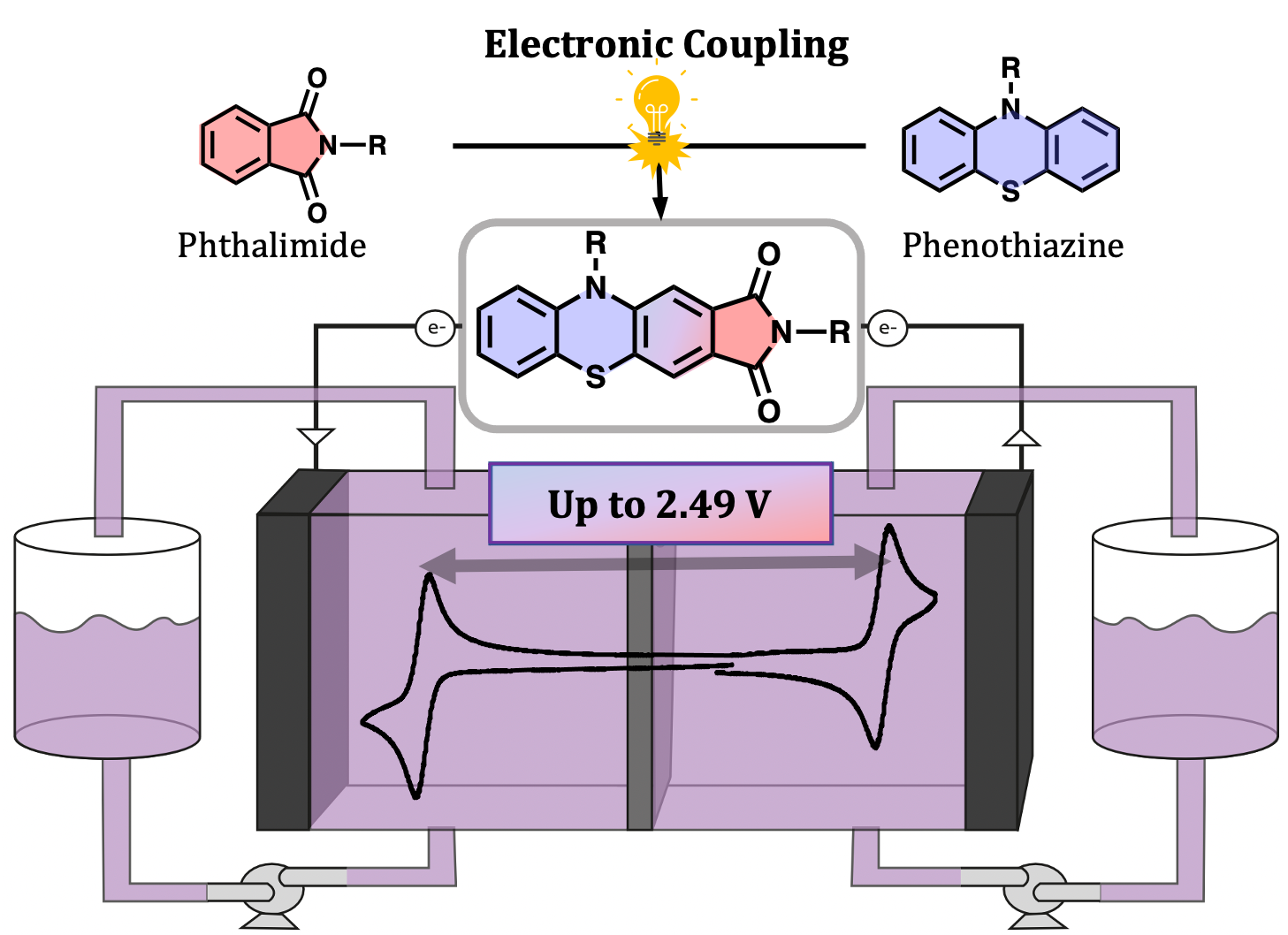
Scientific Achievement
Phenothiazine catholytes and phthalimide anolytes have been electronically coupled to create a new class of bipolar redox-active molecules (i.e. molecules that can be both reversibly oxidized and reduced) with enhanced voltages as compared to what can be achieved by their uncoupled building blocks.
Significance and Impact
The demonstrated strategy of electronically couplings anolyte and catholyte structures provides a new tool to improve battery voltages and serves as a roadmap to construct new classes of bipolar redox-active molecules.
Research Details
- Five members of the new class of molecules were synthesized and tested for redox potentials, solubility, rates of diffusion, rates of electron transfer, and electrochemical reversibility.
- The performance of the most promising molecule was demonstrated under relevant flow battery conditions and showed high levels of cycling stability over the course of 275 charge-discharge cycles.

