Scientific Achievement
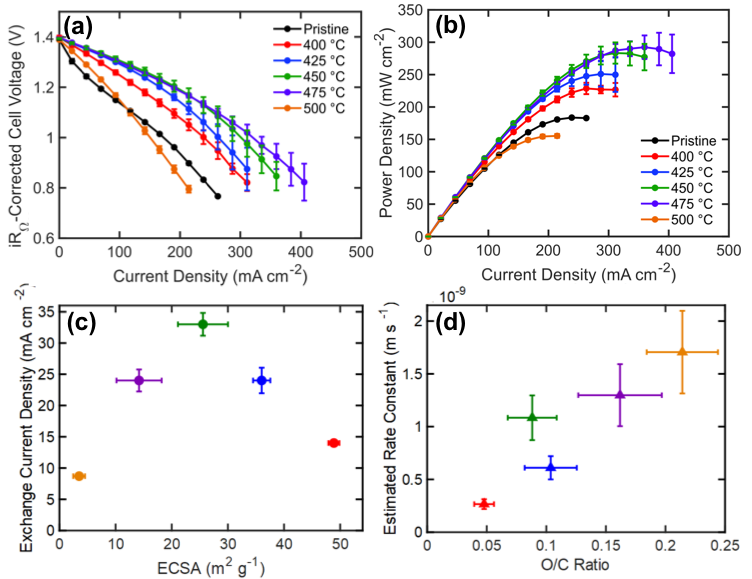
The chemical and physical properties of carbon paper electrodes treated at temperatures between 400-500 °C for 30 hours were quantified, and the subsequent impact of these properties on the performance of vanadium redox flow batteries explained. We find that the oxygen content and electrochemically active surface area, two parameters that govern the kinetic performance of the vanadium flow battery, follow opposing temperature trends; thus, optimal performance is achieved at a balance of these properties rather than a maximization of each.
Significance and Impact
Thermal pretreatment of electrodes is a common technique to improve flow battery performance but its effect on relevant electrode properties and the subsequent effect of these properties on performance remain poorly understood. The results of this work can be used to inform the design of electrodes with optimal properties for next-generation flow batteries.
Research Details
- Ex situ characterization of material and chemical electrode properties was performed using SEM, XTM, EDX, XRD, Raman spectroscopy, and goniometry.
- In situ electrode characterization was performed to identify the electrode capacitance, and electrode efficacy in vanadium flow batteries was evaluated using electrochemical impedance spectroscopy, polarization, and cell cycling.

