Scientific Achievement
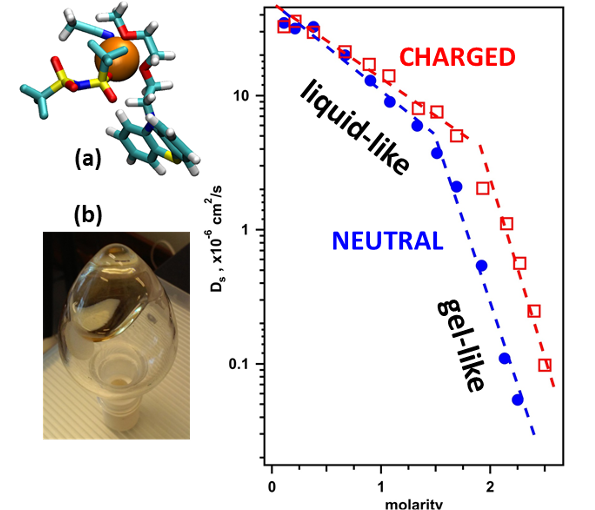
Neutral and charged crowded electrolytes containing redox active organic molecules (redoxmers) are shown to suffer exponential decline in their dynamic properties as they approach the gel-like regime that sets in between 1 and 2 M.
Significance and Impact
In crowded all-organic electrolyte solutions there are hard limits for increasing energy density that cannot be surpassed regardless of the redoxmer nature or electrolyte composition. Only select systems in hot ionic liquid like solutions can have favorable dynamic properties allowing 2.5-3 M operation of electrochemical cells.
Research Details
- Phenothiazine redoxmers miscible with acetonitrile based electrolyte were used to study crowding effects
- Viscosity, ionic conductivity, diffusivity, and x-ray scattering were studied
- Liquid-like and gel like behaviors were observed and rationalized using molecular dynamics modeling.

