Scientific Achievement
We have successfully developed non-flammable hybrid single-ion-conducting electrolytes comprising inorganic sulfide glass particles covalently bonded to a perfluoropolyether polymer. These electrolytes present high transference numbers, unprecedented ionic conductivities at room temperature, excellent electrochemical stability, and limit the dissolution of lithium polysulfides.
Significance and Impact
This study describes new hybrid electrolytes that provide a compelling alternative to the traditional glass, ceramic, or polymer battery electrolytes. These hybrid electrolytes represent a significant step toward addressing the challenges of enabling the next generation cathodes.
Research Details
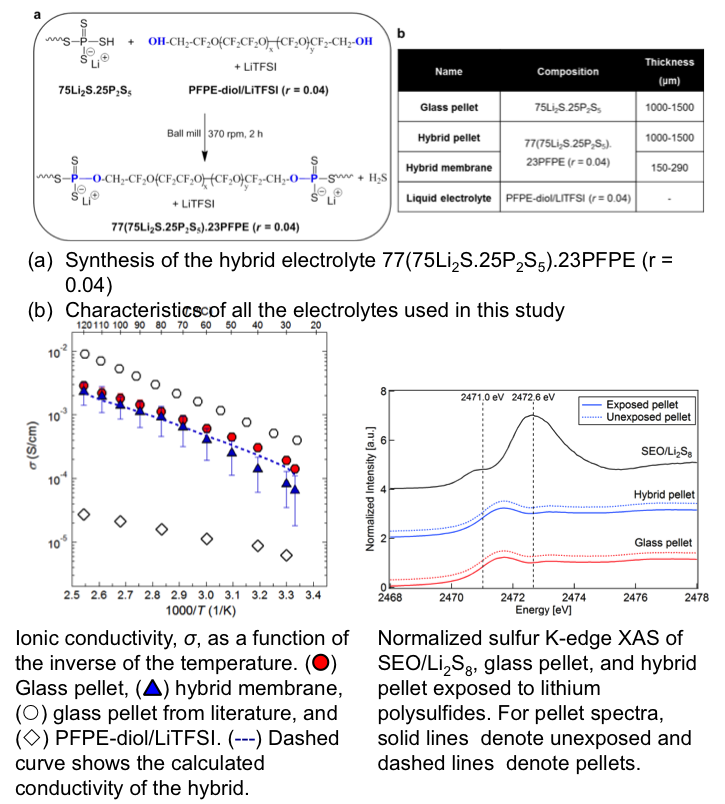
- The hybrid with 23 wt.% perfluoropolyether exhibits low shear modulus relative to neat glass electrolytes, ionic conductivity of 10-4 S/cm at room temperature, cation transference number close to unity, and a electrochemical stability window up to 5 V.
- X-ray absorption spectroscopy indicates that the hybrid electrolyte limits lithium polysulfide dissolution and is thus ideally suited for Li-S cells.
Work performed at Lawrence Berkeley National Laboratory (JCESR partner) by I. Villaluenga, K. H. Wujcik, W. Tong, D. Devaux, D. H. C. Wong, J. M. DeSimone and N. P. Balsara PNAS, 2015 (web publication)

