Research Highlights
-
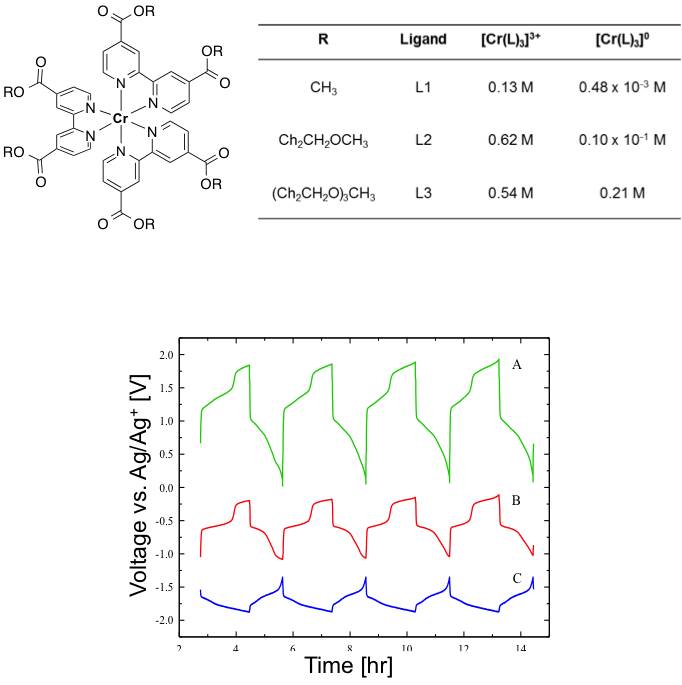
Complexes Containing Redox Non-Innocent Ligands for Symmetric, Multi-Electron Transfer Non-Aqueous Redox Flow Batteries
Designed and synthesized functionalized chromium-bipyridine complexes with high solubilities that undergo up to 6 electron transfers over a ~2 V window Read More
-
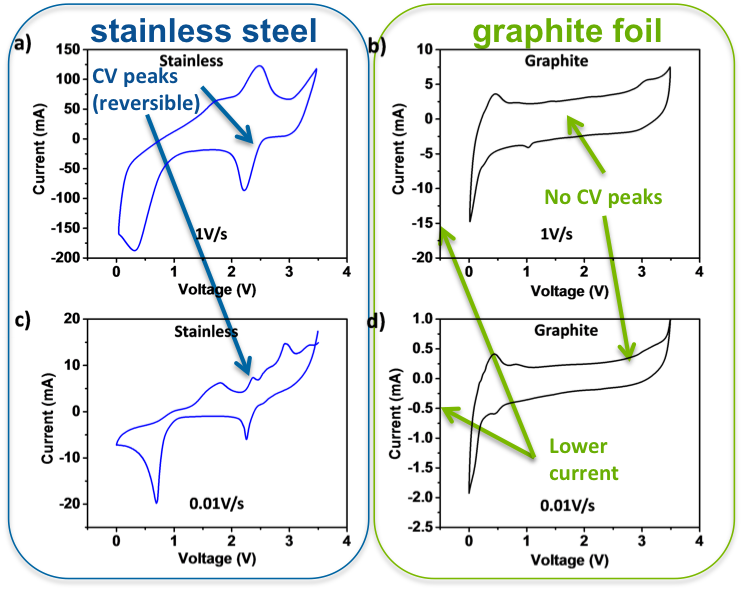
Current Collector Corrosion in Ca-Ion Batteries
This study identified and verified corrosion of the metallic current collectors in a series of multivalent electrolyte/electrode systems; calcium based systems were shown to be particularly susceptible to corrosion. Read More
-
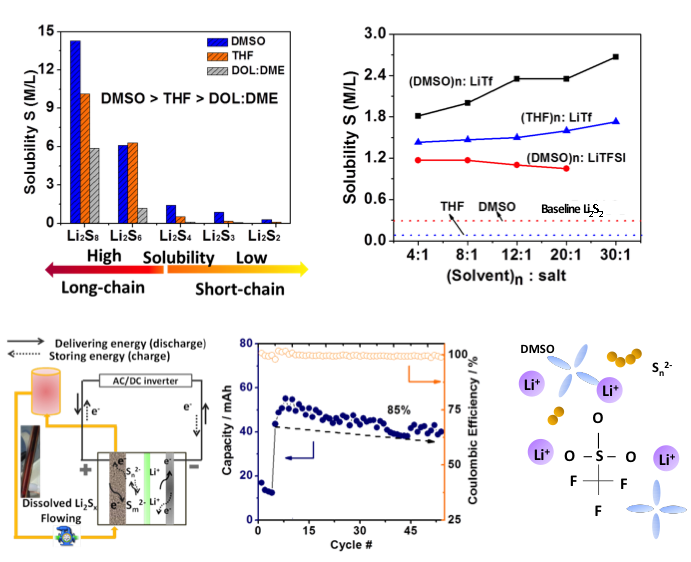
Understanding and Manipulating Solution Chemistry of Polysulfides for Lithium Sulfur Batteries
The factors that govern the solubility of various polysulfides were identified. LiTf with strong ionic association strength increases the solubility of short-chain species significantly. A solution Li-S flow cell was demonstrated using LiTf as additive in the polysulfide catholyte. Read More
-
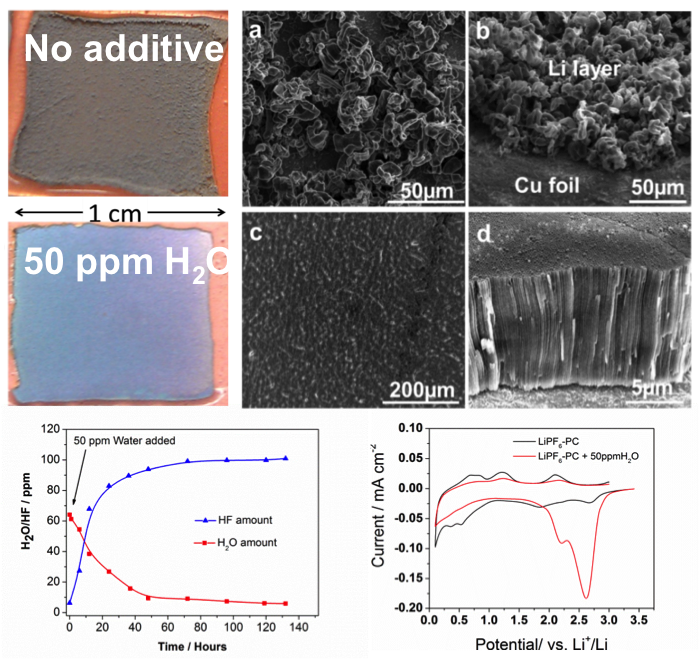
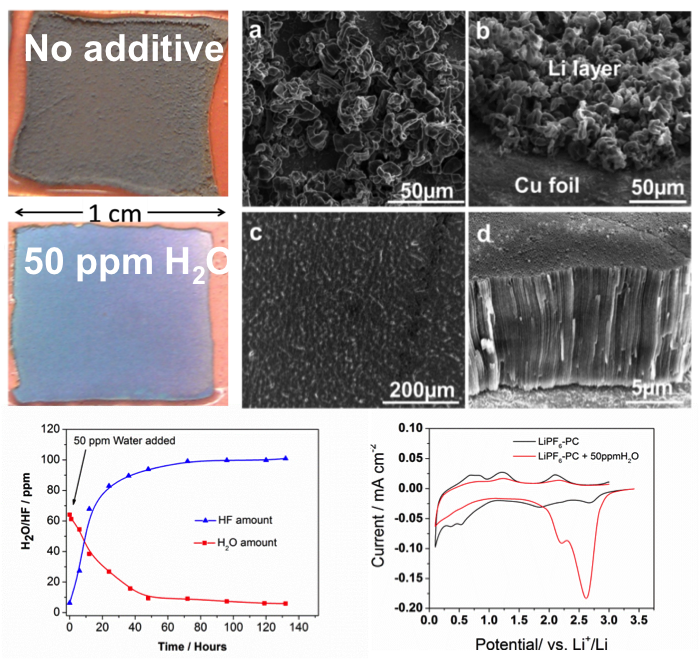
Dendrite-Free Li Deposition Using Trace-Amounts of Water as an Electrolyte Additive
Residual water (H2O) present in nonaqueous electrolytes has been widely regarded as a detrimental factor for lithium (Li) batteries. However, dendrite-free Li film can be obtained with trace H2O as an electrolyte additive. Read More
-
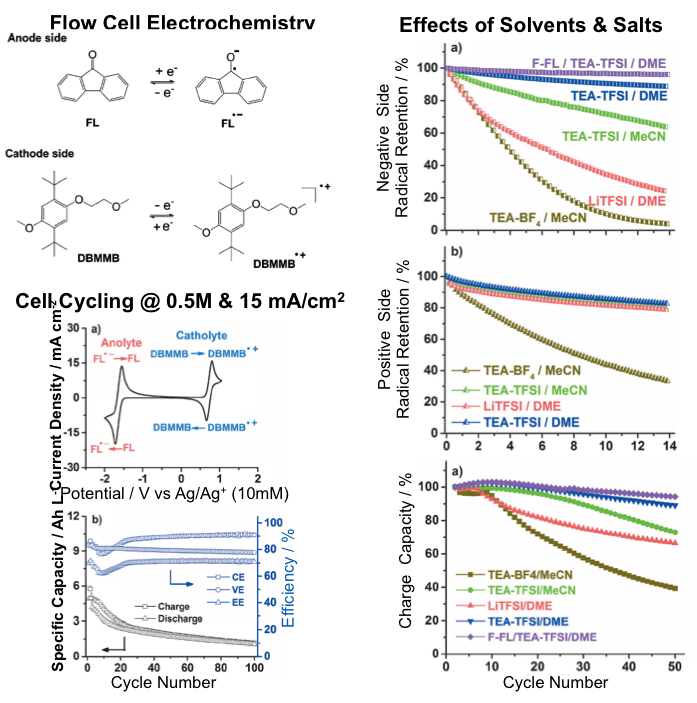
Radical Compatibility with Nonaqueous Electrolytes and Its Impact on an All-Organic Redox Flow Battery
A new nonaqueous all-organic redox flow chemistry was developed with a cell voltage of ~2.4V, energy efficiency of >70%, and operational current density of >10mA/cm2. Electron spin resonance (ESR) study reveals that the chemical stability of both charged organic radical species dictates the cycling stability of flow cells and is highly dependent on the supporting solvents and salts. Read More
-
Demonstration of Magnesium Intercalation into a High-Voltage Oxide Electrode
First demonstration of reversible insertion of multivalent magnesium ions (Mg2+) into a spinel-type manganese oxide (Mn2O4), using multi-modal characterization Read More
-
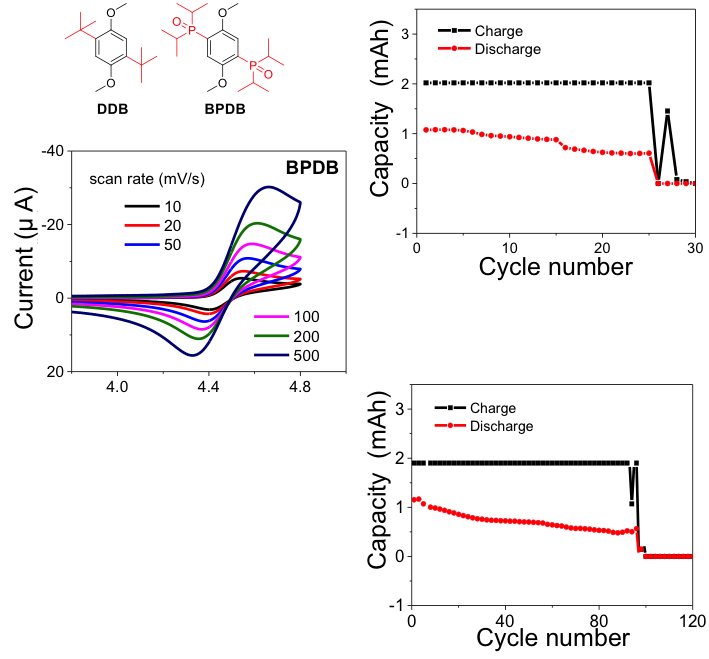
An Organophosphine Oxide Redox Shuttle Additive that Delivers Long-term Overcharge Protection for 4 V Lithium-ion Batteries
A novel redox shuttle molecule, BPDB, was developed through replacing the tert-butyl group in DDB by bulky organophosphine oxide group Read More
-
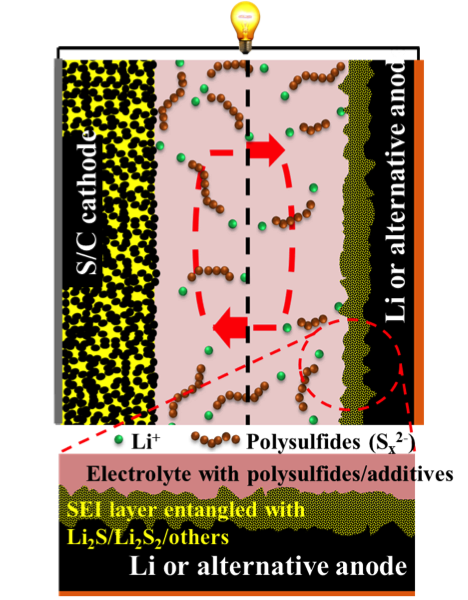
Anodes for Rechargeable Lithium-Sulfur Batteries
Recent developments on the protection of the Li metal anode in Li-S batteries are reviewed Read More
-
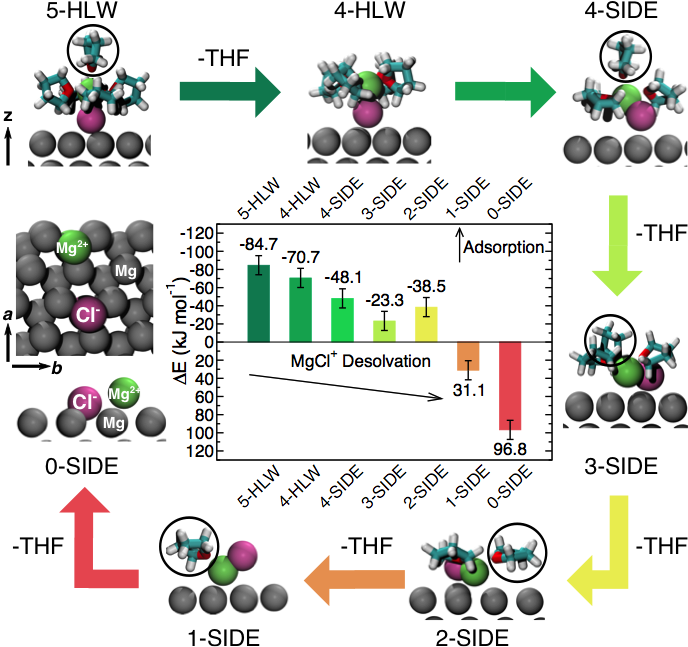
Understanding the Initial Stages of Reversible Mg Deposition and Stripping in Inorganic Nonaqueous Electrolytes
The chemical species at the Mg-anode surface in the presence of Magnesium Aluminum-Chloro complex (MACC) electrolyte were identified. While solvent molecules (THF and DME) are loosely bound at the Mg(0001) surface, the constituents of the MACC electrolyte (e.g. MgCl+, see diagram) are strongly anchored at the electrode. Read More
-
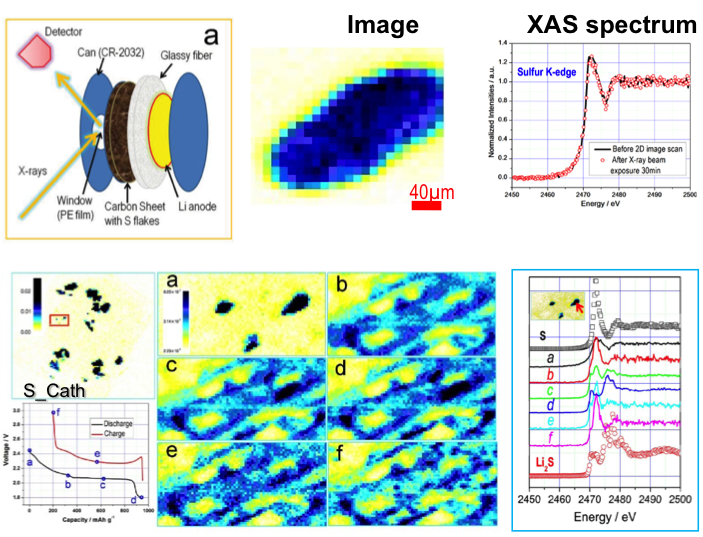
Direct Observation of the Redistribution of Sulfur and Polysufides in Li-S Batteries by In Situ X-Ray Fluorescence Microscopy
The dissolution and redistribution of sulfur and polysulfides were directly observed by XRF imaging. The long-chain polysulfides were generated as soon as discharge was initiated. The dissolved polysulfide diffused to the anode side and deposited onto (and reacted with) lithium metal. Read More
Latest Updates
-
You’re Invited - JCESR and Beyond: Translating the Basic Science of Batteries
Please join us at Argonne National Laboratory on Tuesday, April 4, 2023 for JCESR and Beyond: Translating the Basic Science of Batteries. Registration is now open. This in-person event will celebrate 10 years of research from the Joint Center… Read More
-
A Message from JCESR: In Memory of George Crabtree
It is with heavy hearts that we say goodbye to George Crabtree, a Senior Scientist and Distinguished Fellow at Argonne National Laboratory, and Director of the Joint Center for Energy Storage Research (JCESR), who passed away unexpectedly on January 23. Dr. Read More
-
Cyanopyridines As Extremely Low-Reduction-Potential Anolytes for Nonaqueous Redox Flow Batteries
Discovery of a cyanophenylpyridine derivative with a very low reduction potential and good stability during cycling. Read More
-
Characterizing Redoxmer – Electrode Kinetics Using a SECM-Based Spot Analysis Method
Identified asymmetries in electron transfer (ET) kinetics between the reduction and oxidation of ferrocene-based redoxmers by measuring the ET rate constants (kf/kb) as a function of electrode potential. Read More
-
Benzotriazoles as Low Potential Anolytes for Non-Aqueous Redox Flow Batteries
We developed an easy-to-synthesize benzotriazole-based anolyte with a high energy redox potential (-2.3 V vs Fc/Fc+) and high solubility that demonstrates stable electrochemical cycling performance. Read More

