Research Highlights
-
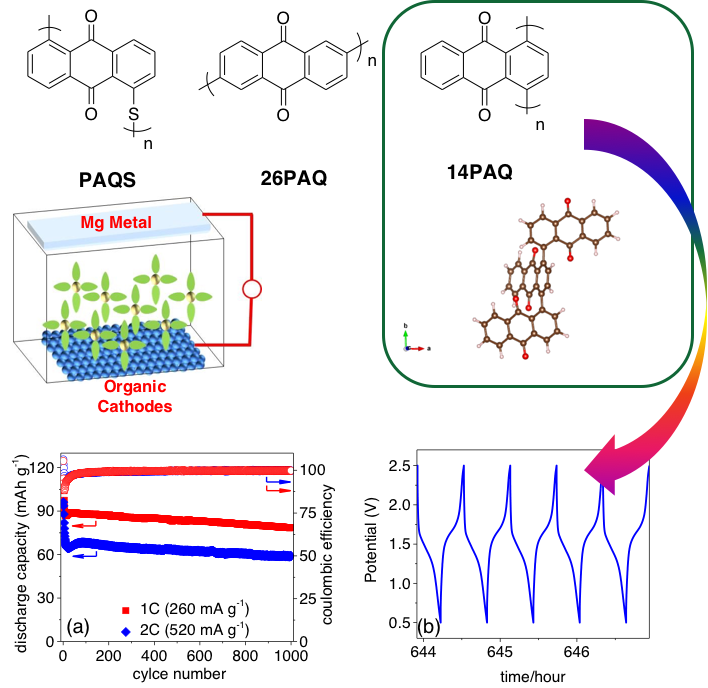
A Polyanthraquinone-based Organic Cathode for High-performance Rechargeable Magnesium-ion Batteries
A series of anthraquinonyl-based polymers (PAQS, 14PAQ, and 26PAQ) have been prepared and evaluated as high-performance organic cathodes for rechargeable Mg-ion batteries. Mg-14PAQ system shows the best cycling stability among the series: at the current rate of 0.5C, 104.9 mAh g-1 discharge capacity can be obtained in the 100th cycle; successful 1000 cycles with a small amount of capacity loss at 1C. Read More
-
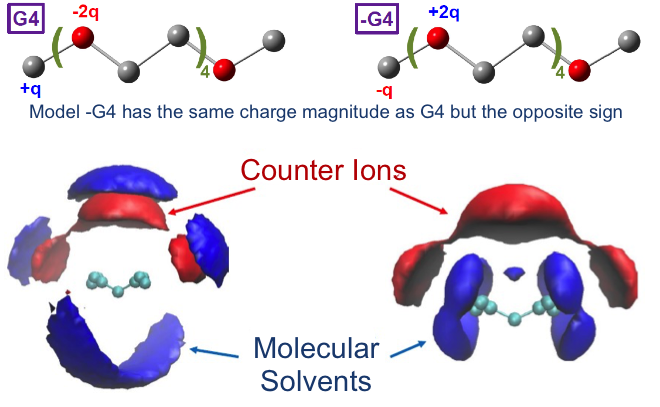
How Mixing Tetraglyme with Ionic Liquid Changes Volumetric and Transport Properties
Charge localization of the solvent molecules affects the liquid phase structure and transport properties in electrolyte solutions. Read More
-
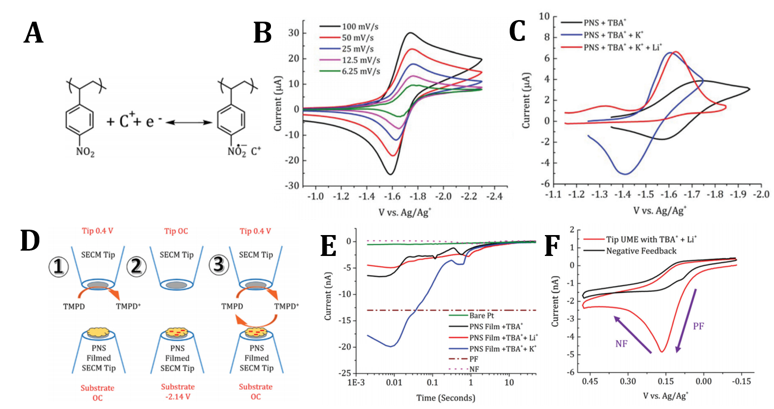
Enhancing the Performance of a Redox Active Polymer via Specific Electrolyte Interactions
A redox active polymer (RAP) with nitro- substituents was synthesized and studied via voltammetric analysis in combination with surface interrogation and ionic sensitive scanning electrochemical microscopy to reveal specific electrolyte interactions on the performance of the material. Read More
-

Effect of the Anion Activity on the Stability of Li Metal Anodes in Lithium-Sulfur Batteries
Discovered why the salt LiTFSI -- when added to the electrolyte of a Li-S battery -- allows the battery to hold a charge much longer than other salts Read More
-
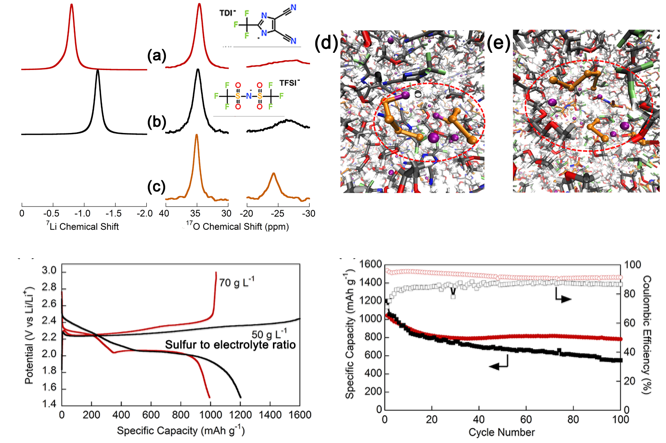
Restricting the Solubility of Polysulfides in Li-S Batteries Via Electrolyte Salt Selection
Lithium 2-trifluoromethyl-4,5-dicyanoimidazole (LiTDI) as a supporting salt in electrolytes suppresses the maximum solubility of Li2S8 by forming a Li4S8 dimer rather than the Li2S3 and Li2S5 observed in a LiTFSI electrolyte, which enables a cell with a high sulfur loading (3 mg-S cm-2) to deliver a 1.67 mAh cm-2 areal capacity after 300 stable cycles at a high current density (2.4 mA cm-2). Read More
-
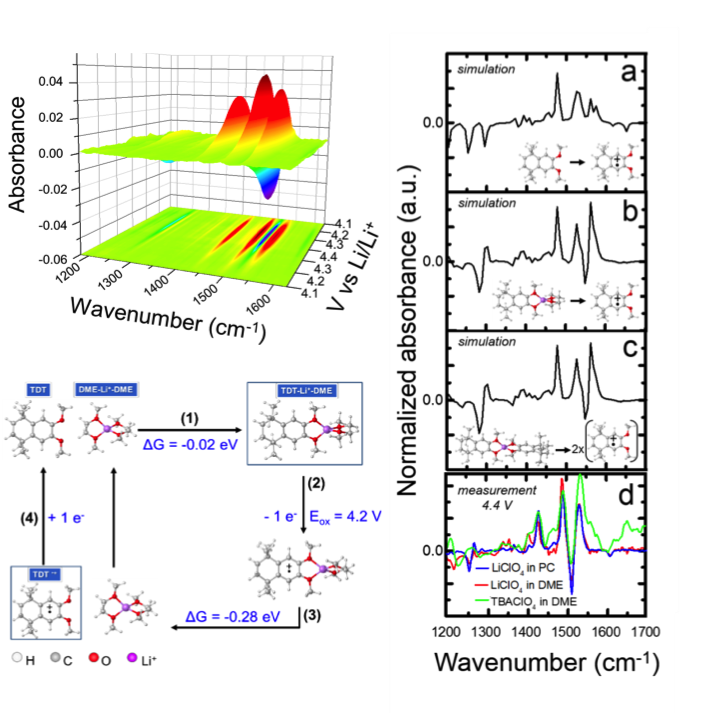
Controlling degradation pathways in organic electrochemistry via redox-mediated Li+ coordination
Exhaustive DFT experiments are well-matched to in-situ spectroscopic data showing Li+ coordination to basic methoxy groups. Li+ coordination promotes improved redox reversibility within Li+ electrolytes. Read More
-
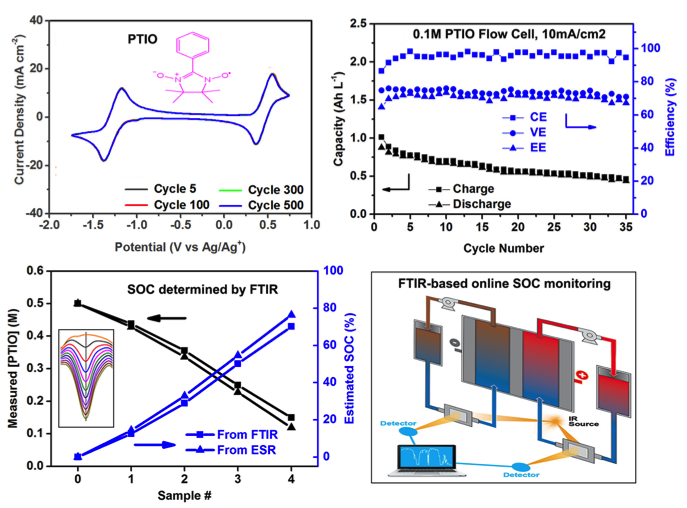
A Symmetric Organic - Based Nonaqueous Redox Flow Battery and Its State of Charge Diagnostics by FTIR
A symmetric nonaqueous flow battery based on the highly soluble, ambipolar PTIO achieved a cell voltage of ~1.7V and decent cyclability. We demonstrated FTIR as an effective method to monitor the state of charge (SOC) of this flow battery. Read More
-
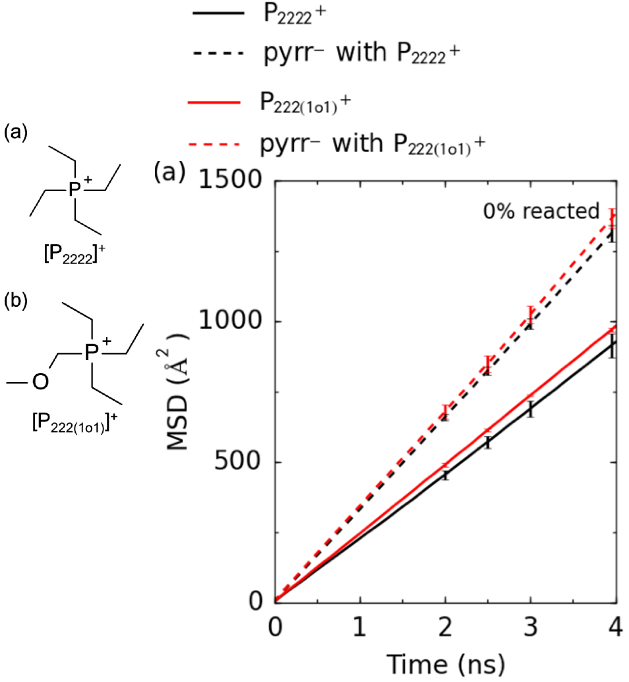
Molecular Design of Low Viscosity Ionic Liquid for High Performance Electrolyte
Computationally predicted viscosities confirmed by experimental measurements. Read More
-

Interface-Induced Renormalization of Electrolyte Energy Levels in Magnesium Batteries
Interface-induced changes to the stability of several electrolyte solvents on Mg-based electrodes were predicted using accurate many-body perturbation theory calculations. Read More
-
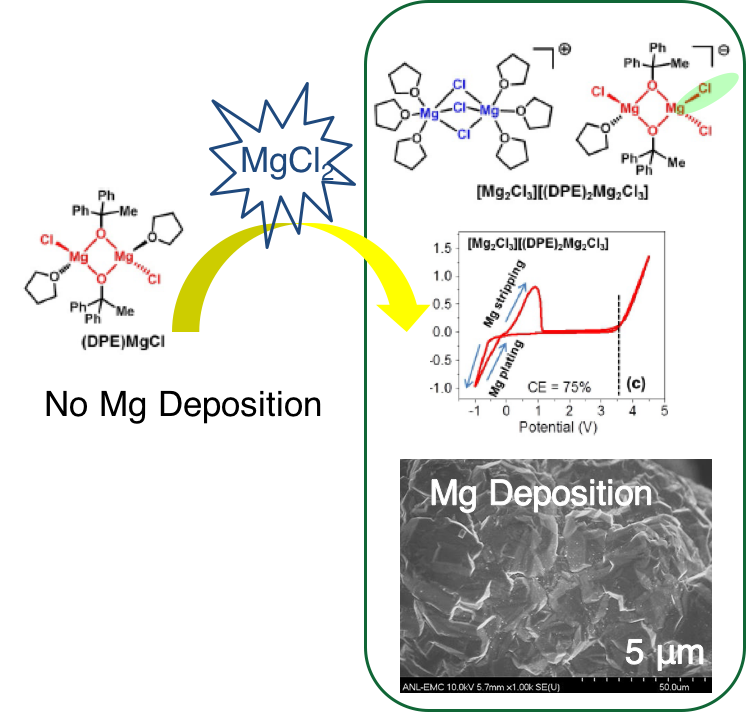
The Role of MgCl2 as a Lewis Base in ROMgCl–MgCl2 Electrolytes for Magnesium-Ion Batteries
A series of alkoxide/siloxide magnesium electrolytes were developed with environmentally benign MgCl2 and ROMgCl (R=alkyl or silyl). The resulting electrolytes possess oxidative stability up to about 3.5 V (vs. Mg/Mg2+), representing one of the best stability performing among all reported electrolytes for Mg-ion batteries. The mechanistic study towards the generation of active ionic species was extensively carried out. Read More
Latest Updates
-
You’re Invited - JCESR and Beyond: Translating the Basic Science of Batteries
Please join us at Argonne National Laboratory on Tuesday, April 4, 2023 for JCESR and Beyond: Translating the Basic Science of Batteries. Registration is now open. This in-person event will celebrate 10 years of research from the Joint Center… Read More
-
A Message from JCESR: In Memory of George Crabtree
It is with heavy hearts that we say goodbye to George Crabtree, a Senior Scientist and Distinguished Fellow at Argonne National Laboratory, and Director of the Joint Center for Energy Storage Research (JCESR), who passed away unexpectedly on January 23. Dr. Read More
-
Cyanopyridines As Extremely Low-Reduction-Potential Anolytes for Nonaqueous Redox Flow Batteries
Discovery of a cyanophenylpyridine derivative with a very low reduction potential and good stability during cycling. Read More
-
Characterizing Redoxmer – Electrode Kinetics Using a SECM-Based Spot Analysis Method
Identified asymmetries in electron transfer (ET) kinetics between the reduction and oxidation of ferrocene-based redoxmers by measuring the ET rate constants (kf/kb) as a function of electrode potential. Read More
-
Benzotriazoles as Low Potential Anolytes for Non-Aqueous Redox Flow Batteries
We developed an easy-to-synthesize benzotriazole-based anolyte with a high energy redox potential (-2.3 V vs Fc/Fc+) and high solubility that demonstrates stable electrochemical cycling performance. Read More

