Research Highlights
-
The Lightest Organic Radical Cation for Charge Storage in Redox Flow Batteries
By systematically shedding molecular fragments of 2,5-di-tert-butyl-1,4-bis(2-methoxyethoxy)benzene that are considered important for radical cation steric stabilization, we discovered a minimalistic structure that retains long-term stability in its oxidized form and exhibits the intrinsic capacity of 161 mAh/g. Read More
-
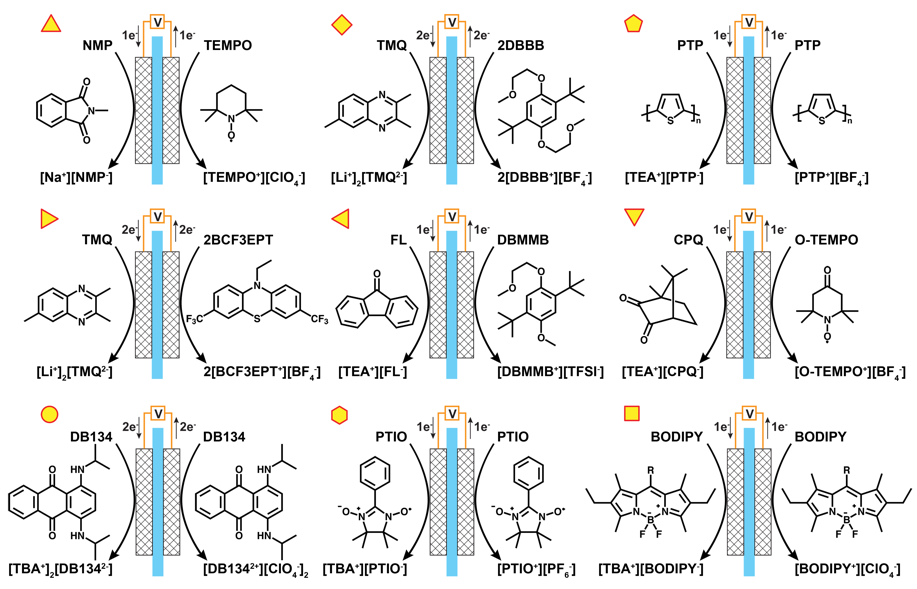
Recent Advances in Molecular Engineering of Redox Active Organic Molecules for Nonaqueous Flow Batteries
This review article summarizes the recent work on organic molecules for redox flow batteries, both redox couples and tailored redox active molecules. Read More
-

Evaluation of Sulfur Spinel Compounds for Multivalent Battery Cathode Applications
Systematic evaluations were performed to screen for feasible MV cathode candidates. Spinel MgCr2S4, MgTi2S4, MgMn2S4 and CaCr2S4 were found as the most promising systems for multivalent cathode applications due to their facile cation mobility (<650meV) among the 3d transition–metal sulfur spinel compounds. Read More
-

Molecular Dynamics Modeling of the Conductivity of Lithiated Nafion Containing Nonaqueous Solvents
Successfully predicted the ionic conductivities of lithiated Nafion membranes swollen in dimethyl sulfoxide (DMSO) and acetonitrile (ACN) using molecular dynamics and experimentally validated the results. Read More
-
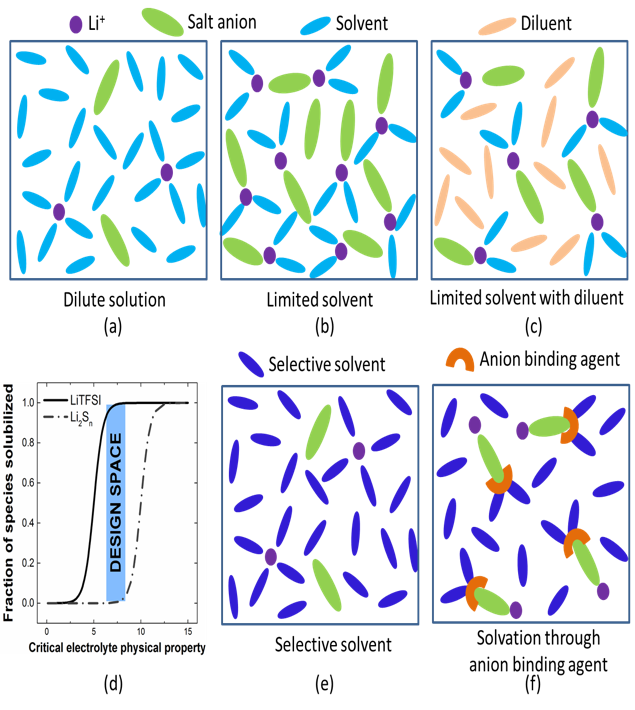
Sparingly Solvating Electrolytes for High Energy Density Lithium-Sulfur Batteries
This work presents the promising new concepts of using sparingly solvating electrolyte to enable Li-S battery operation at lean electrolyte condition, as well as the design rules for discovering new electrolyte systems. Read More
-
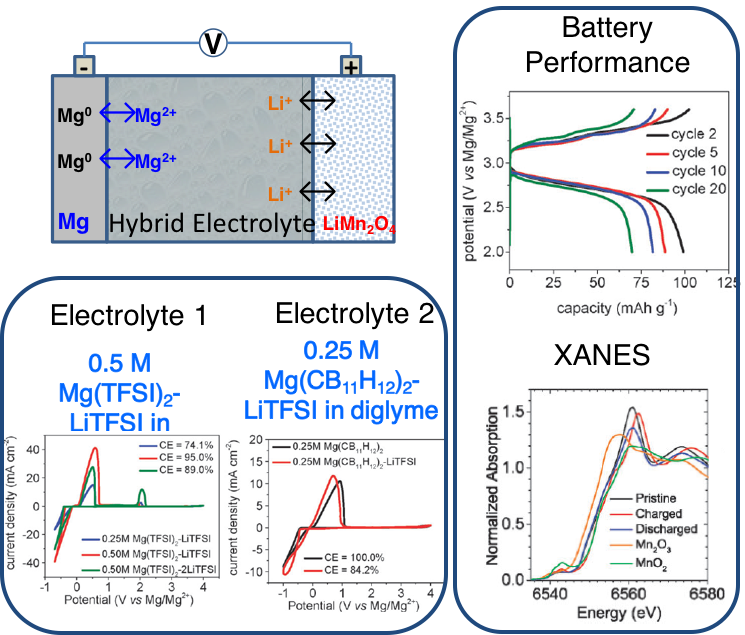
Advanced Hybrid Batteries with a Magnesium Metal Anode and Spinel LiMn₂O₄ Cathode
Two Mg-Li dual salt hybrid electrolytes were developed with excellent oxidative stability up to around 3.8 V (vs Mg/Mg2+) on a aluminum current collector, enabling the successful coupling of several state-of-the-art lithium-ion intercalation cathodes (LiMn2O4, LiCoO2 and LiNi1/3Mn1/3Co1/3O2) with magnesium metal anodes. Read More
-
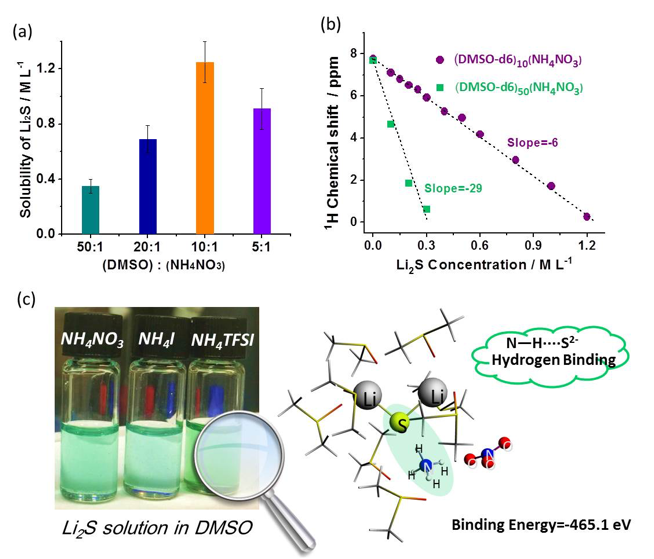
Ammonium Additives to Dissolve Li2S through Hydrogen Binding for High Energy Li-S Batteries
Ammonium salts are demonstrated as effective additives to promote the dissolution of Li2S (up to concentrations of 1.25 M) in DMSO solvent at room temperature through hydrogen binding between N-H groups and S2- anions. Read More
-
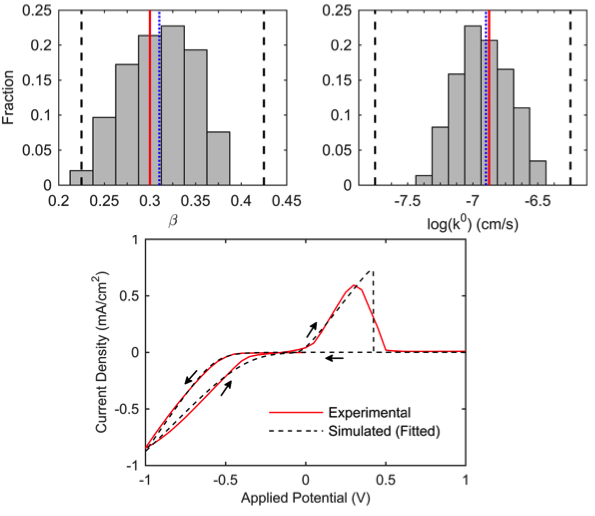
Computational Model of Magnesium Deposition and Dissolution for Property Determination via Cyclic Voltammetry
A computationally inexpensive model has been developed to parametrically study the cyclic voltammetry behavior of magnesium-based battery electrolytes. Read More
-
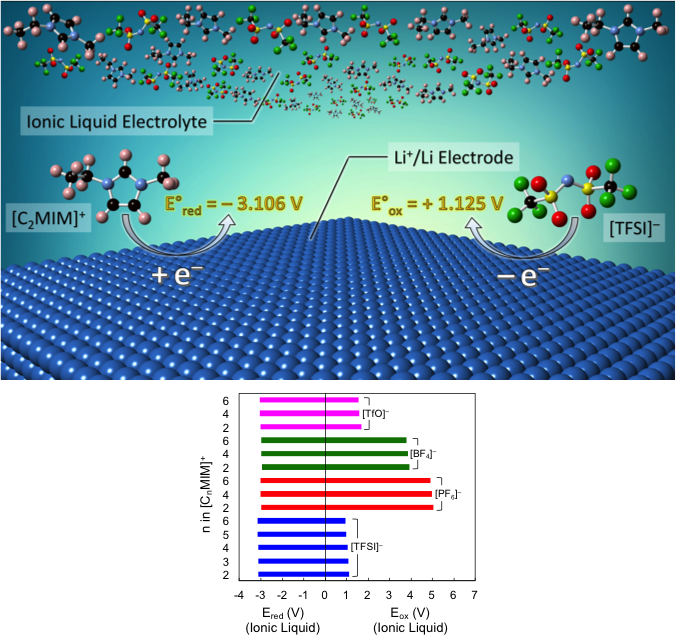
Electrochemical Stability Window of Imidazolium-Based Ionic Liquids as Electrolytes for Lithium Batteries
The oxidation and reduction potentials of the constituent cations and anions of a series of imidazolium-based ionic liquids with respect to a Li+/Li reference electrode were calculated using density functional theory (DFT) following the method of thermodynamic cycles. Read More
-
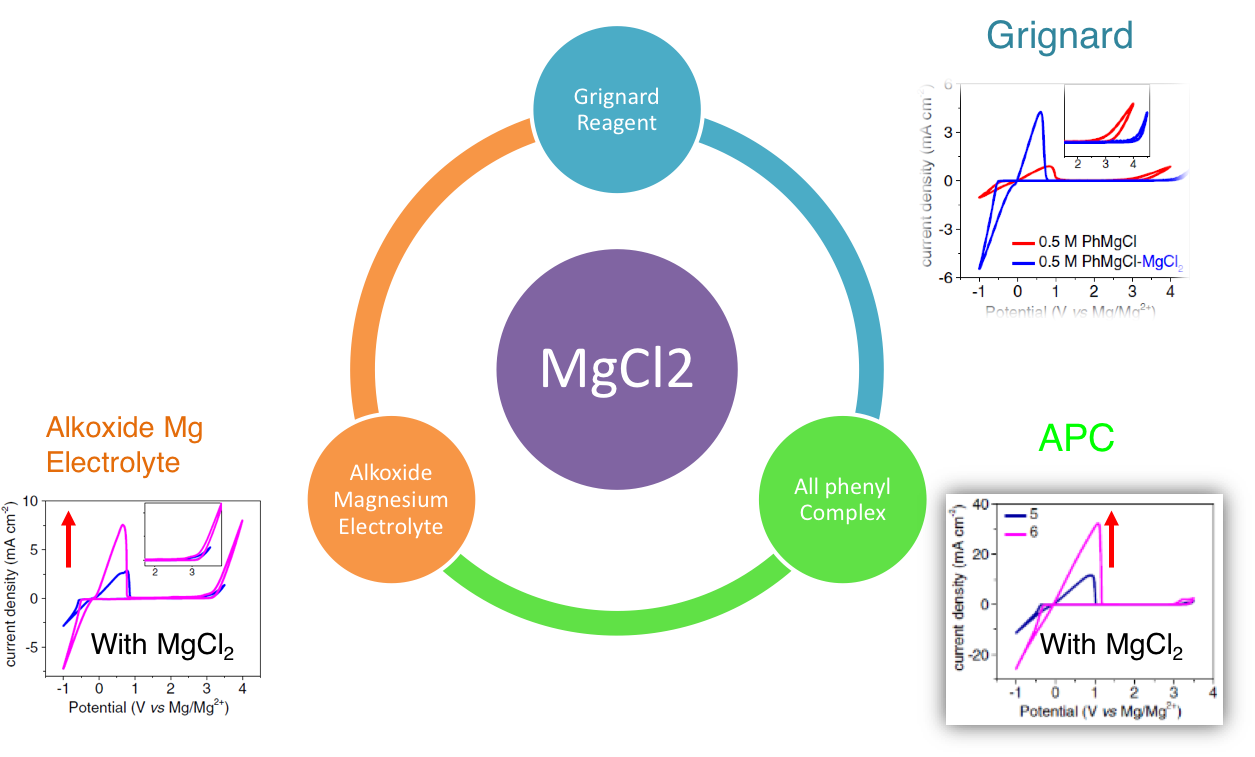
MgCl2: The Key Ingredient to Improve Chloride Containing Electrolytes for Rechargeable Magnesium-Ion Batteries
The effect of MgCl2 on a series of chloride containing magnesium electrolytes has been experimentally investigated. The incorporation of MgCl2 into these electrolytes results in the significant improvement in all aspects of electrochemical properties. Read More
Latest Updates
-
You’re Invited - JCESR and Beyond: Translating the Basic Science of Batteries
Please join us at Argonne National Laboratory on Tuesday, April 4, 2023 for JCESR and Beyond: Translating the Basic Science of Batteries. Registration is now open. This in-person event will celebrate 10 years of research from the Joint Center… Read More
-
A Message from JCESR: In Memory of George Crabtree
It is with heavy hearts that we say goodbye to George Crabtree, a Senior Scientist and Distinguished Fellow at Argonne National Laboratory, and Director of the Joint Center for Energy Storage Research (JCESR), who passed away unexpectedly on January 23. Dr. Read More
-
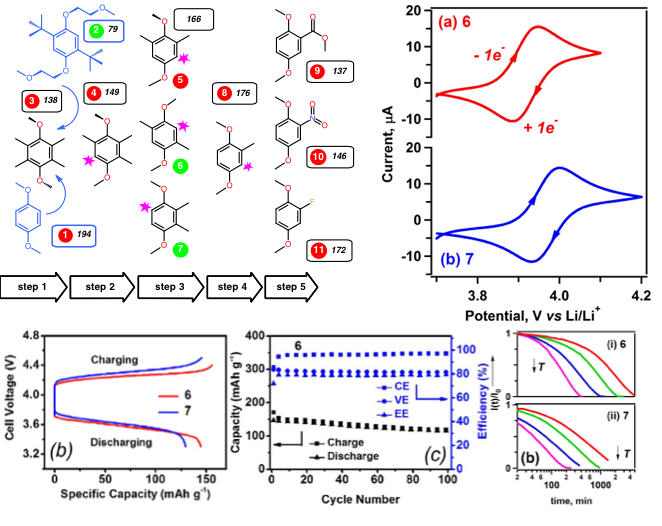
Cyanopyridines As Extremely Low-Reduction-Potential Anolytes for Nonaqueous Redox Flow Batteries
Discovery of a cyanophenylpyridine derivative with a very low reduction potential and good stability during cycling. Read More
-
Characterizing Redoxmer – Electrode Kinetics Using a SECM-Based Spot Analysis Method
Identified asymmetries in electron transfer (ET) kinetics between the reduction and oxidation of ferrocene-based redoxmers by measuring the ET rate constants (kf/kb) as a function of electrode potential. Read More
-
Benzotriazoles as Low Potential Anolytes for Non-Aqueous Redox Flow Batteries
We developed an easy-to-synthesize benzotriazole-based anolyte with a high energy redox potential (-2.3 V vs Fc/Fc+) and high solubility that demonstrates stable electrochemical cycling performance. Read More

