Research Highlights
-
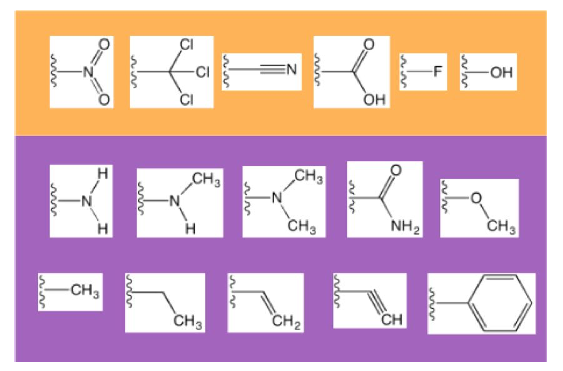
The Effects of Functional Groups in Redox-Active Organic Molecules: A High-Throughput Screening Approach
This work utilized high-throughput screening of thousands of molecules to explore the relationship between molecular structure and the reduction/oxidation potentials (ROPs) of organic electrolytes. Read More
-
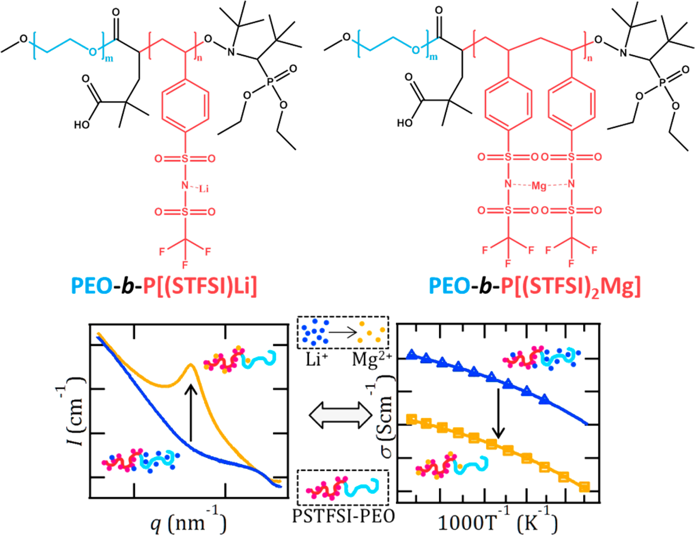
Interaction Parameter (χ) and Ion Dissociation in Lithium and Magnesium Single-Ion-Conducting Block Copolymers Electrolytes
Ion dissociation and block copolymer thermodynamics are intimately coupled: ion dissociation in lithiated systems suppresses microphase separation. Read More
-
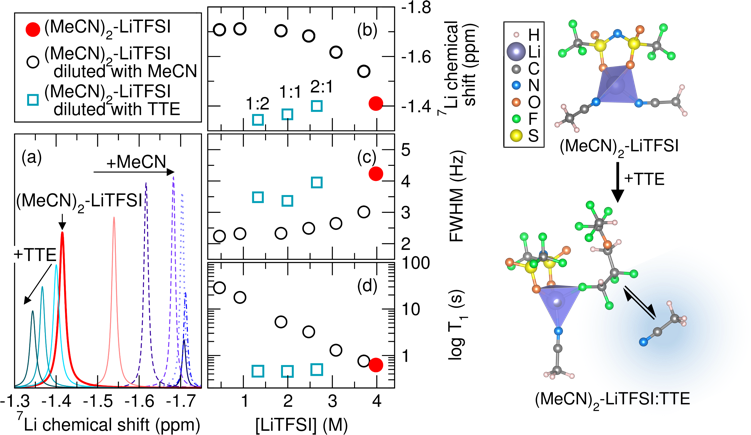
The Effect of Hydrofluoroether Addition on S8 Reduction and the Li+ Solvation Structure in the Solvate Electrolyte
The hydrofluoroether, TTE, competes with MeCN coordination to Li+ in the solvate electrolyte resulting in a higher free MeCN content as TTE is added. The content of free MeCN affects S8 reduction kinetics likely through facilitation of polysulfide formation and enhanced local solvation effects. Read More
-
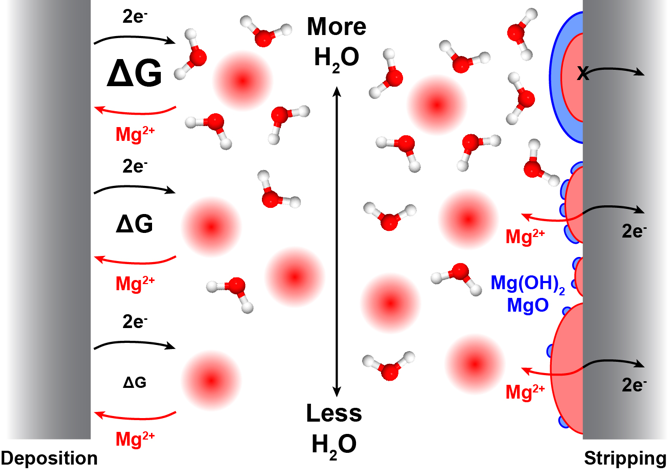
Impact of Trace Impurities on the Reversibility of Mg Anodes
Trace amounts of water (≤ 3 ppm) are shown to strongly influence both the kinetics of Mg deposition and the reversibility of Mg stripping, with Cl- mitigating passivation by H2O by strongly modifying the Mg surface chemistry. Read More
-

Dendrites and Pits: Untangling the Complex Behavior of Lithium Metal Anodes through Operando Video Microscopy
A mechanistic understanding of the complex cycling behavior of Li metal anodes has been gained by combining operando video microscopy with continuum-scale modeling of Li/Li symmetric cells. Read More
-
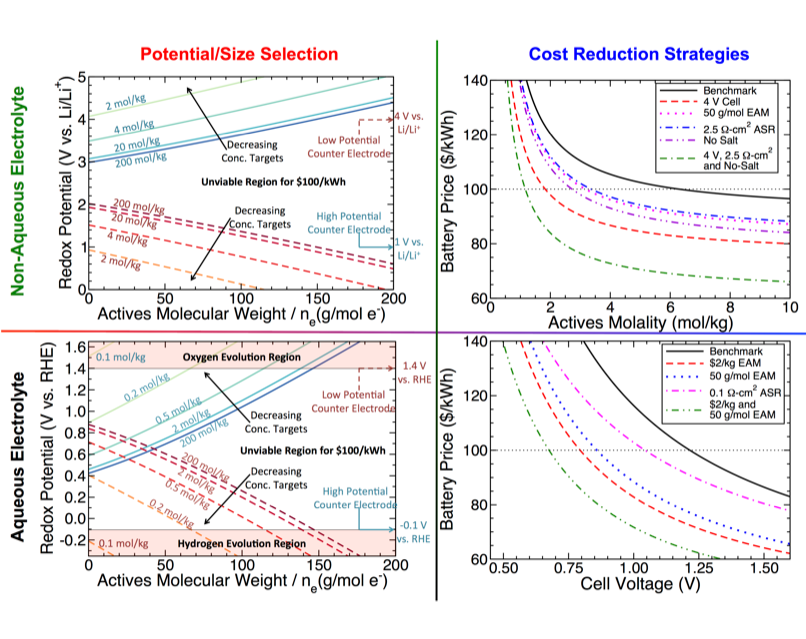
Cost-Driven Materials Selection Criteria for Redox Flow Battery Electrolytes
Techno-economic analysis shows that design challenges for non-aqueous batteries include minimizing salt content and dropping redox-active species concentration to moderate levels, while aqueous batteries are sensitive to only redox-active material cost and cell voltage due to low area-specific resistance and supporting electrolyte costs. Read More
-
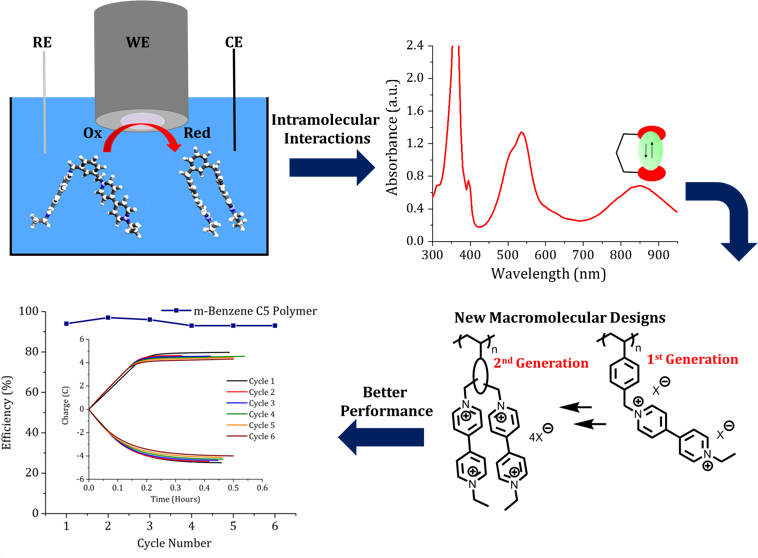
Impact of Backbone Tether Length and Structure on the Electrochemical Performance of Viologen RAPs
Fundamental inter-valence interactions are used as starting point for improving the electrochemical properties of redox active polymers and explore structure/activity relationships. Read More
-
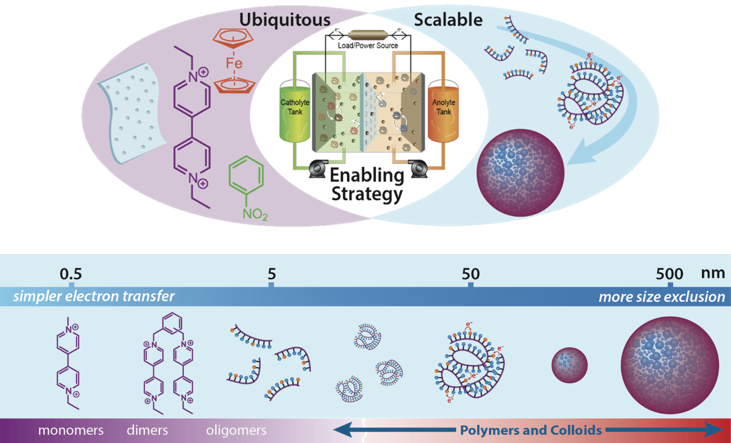
The Evolution of Redox Active Polymers as Soluble Nanomaterials for Energy Storage
Redox active polymers (RAPs) are highlighted for use as new soluble energy storage materials. This review article shines light on some of the major work from the RAP flow battery sprint in JCESR and provides perspective on future studies of RAPs. Read More
-
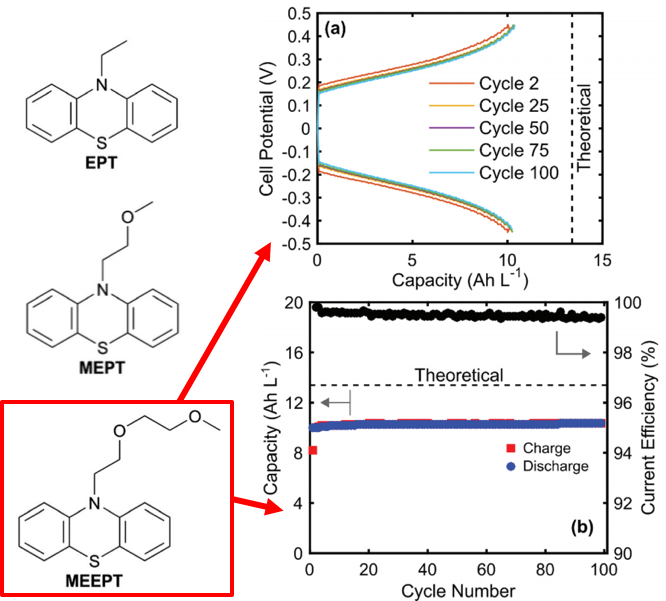
High Current Density, Long Duration Cycling of Soluble Organic Active Species for Non-Aqueous Redox Flow Batteries
We simultaneously engineered a soluble catholyte molecule and low resistance flow cell, enabling unprecedented high current density and undetectable capacity fade cycling of an organic molecule in a non-aqueous flow cell. Read More
-
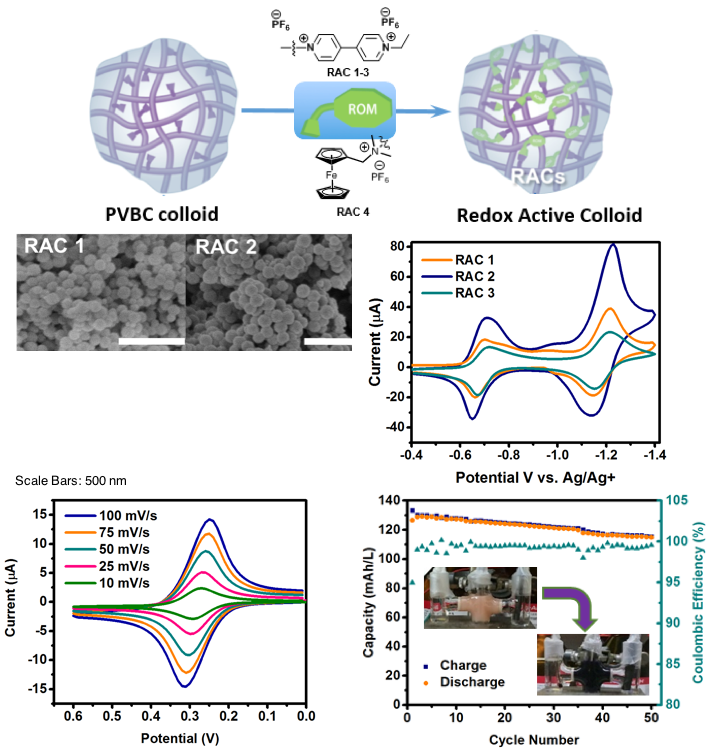
Redox Active Colloids as Discrete Energy Storage Carriers
Redox active colloids (RACs) were introduced as a promising class of energy storage materials. These were synthesized and electrochemically studied for their charge transfer properties as well as charge storage capabilities. Read More
Latest Updates
-
You’re Invited - JCESR and Beyond: Translating the Basic Science of Batteries
Please join us at Argonne National Laboratory on Tuesday, April 4, 2023 for JCESR and Beyond: Translating the Basic Science of Batteries. Registration is now open. This in-person event will celebrate 10 years of research from the Joint Center… Read More
-
A Message from JCESR: In Memory of George Crabtree
It is with heavy hearts that we say goodbye to George Crabtree, a Senior Scientist and Distinguished Fellow at Argonne National Laboratory, and Director of the Joint Center for Energy Storage Research (JCESR), who passed away unexpectedly on January 23. Dr. Read More
-
Cyanopyridines As Extremely Low-Reduction-Potential Anolytes for Nonaqueous Redox Flow Batteries
Discovery of a cyanophenylpyridine derivative with a very low reduction potential and good stability during cycling. Read More
-
Characterizing Redoxmer – Electrode Kinetics Using a SECM-Based Spot Analysis Method
Identified asymmetries in electron transfer (ET) kinetics between the reduction and oxidation of ferrocene-based redoxmers by measuring the ET rate constants (kf/kb) as a function of electrode potential. Read More
-
Benzotriazoles as Low Potential Anolytes for Non-Aqueous Redox Flow Batteries
We developed an easy-to-synthesize benzotriazole-based anolyte with a high energy redox potential (-2.3 V vs Fc/Fc+) and high solubility that demonstrates stable electrochemical cycling performance. Read More

