Research Highlights
-

Probing Electrolyte Structure at Interfaces: An Example of the Power of Variable Temperature and Pressure Operando MAS NMR
We have developed a robust method to analyze battery systems under finely-controlled environments of specified composition, temperature, and pressure to reveal the interactions of electrolytes with electrode surfaces. Read More
-
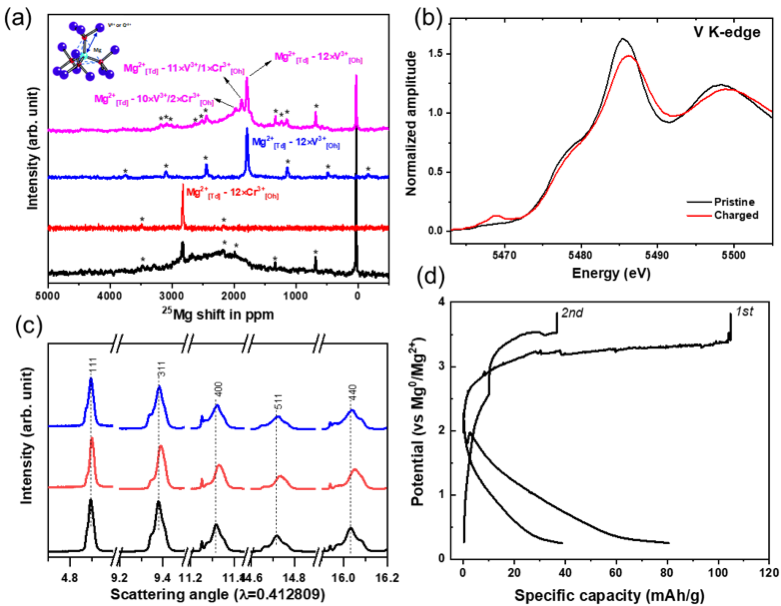
Electrochemical Mg-ion Activity in MgCr2-xVxO4 Spinel Oxides
Bulk demagnesiation in MgCrVO4 spinel lattice is observed in a full-cell configuration paired with a Mg metal anode in a chemically and anodically stable Mg(TPFA)2 electrolyte at 110 oC. Read More
-
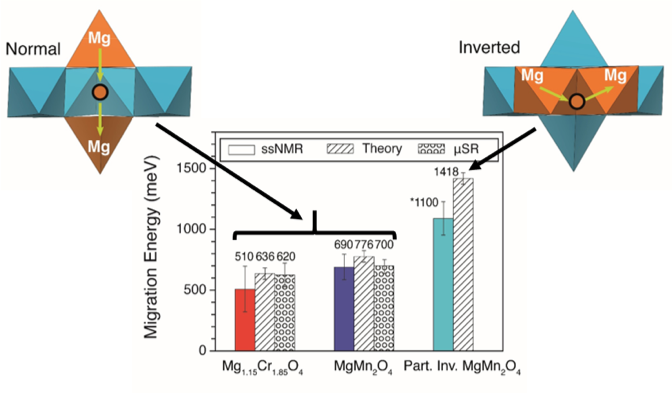
Probing migration of magnesium cations in spinel oxides
Measurements of 25Mg variable temperature solid-state nuclear magnetic resonance (VT ss-NMR), and muon spin relaxation (μSR) in 3 spinel oxides reveal experimental magnesium hoping barriers as low as ∼0.6 eV, in agreement with independent density functional theory (DFT) predictions. Read More
-
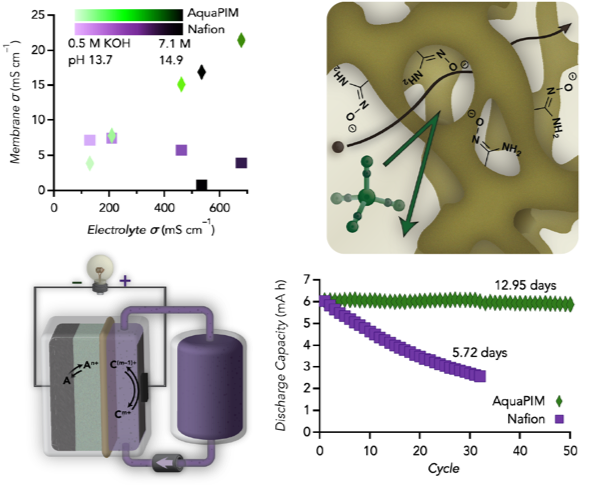
Designing Membranes for Aqueous Alkaline Flow Batteries from Polymers of Intrinsic Microporosity
The incorporation of amidoxime units along the rigid backbone of a polymer of intrinsic microporosity to make them aqueous-compatible (AquaPIMs) allows for exceptional conductivity and stability in harsh alkaline environments while blocking a variety of active materials. Read More
-
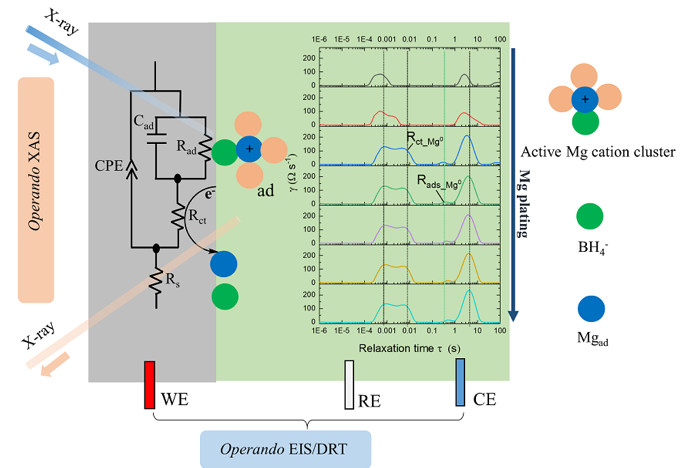
Reversible Electrochemical Interface of Mg Metal and a Conventional Electrolyte Enabled by Intermediate Adsorption
A detailed description of the complex charge-transfer process at a Mg/electrolyte interface is provided where an additional adsorption process, during Mg plating, is confirmed to be a key step. Read More
-
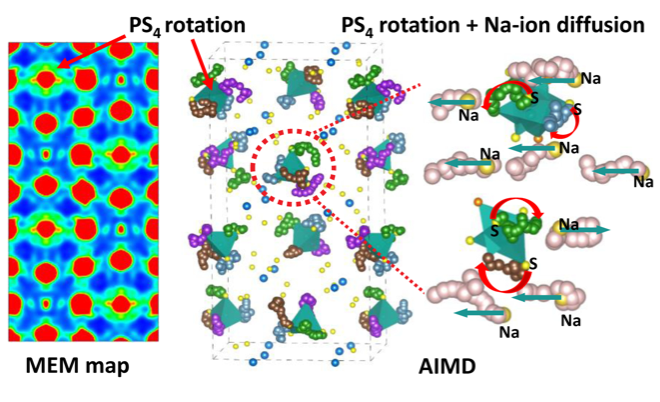
Coupled Cation-Anion Dynamics Enhances Cation Mobility in Room Temperature Superionic Solid-State Electrolytes
Our work reveals facile [PX4]3- anion rotation in superionic Na11Sn2PS12 and Na11Sn2PSe12, and greatly hindered [SbS4]3- rotational dynamics in their less conductive analogue, Na11Sn2SbS12. Along with introducing dynamic frustration in the energy landscape, the fluctuation caused by [PX4]3- anion rotation is firmly proved to couple to, and facilitate long range Na+-cation mobility, by transiently widening the bottlenecks for Na+-ion diffusion. Read More
-
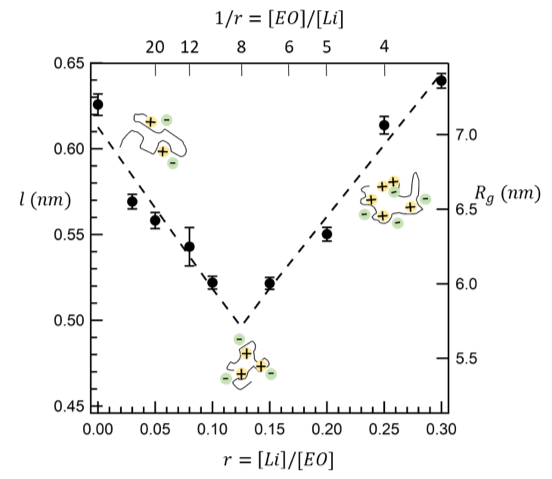
The Effect of Added Salt on the Size of Polymer Chains in Electrolytic Mixtures
The effect of salt concentration on the molecular conformation of a polymer was determined by small angle neutron scattering. Read More
-
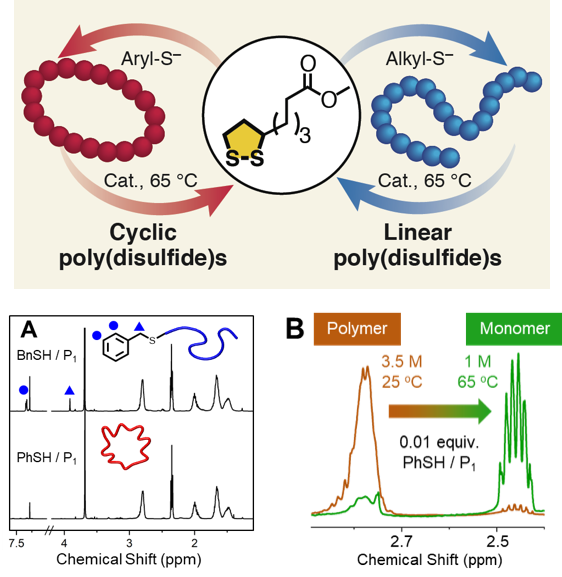
Architecture-Controlled Ring-Opening Polymerization for Dynamic Covalent Poly(disulfide)s
We reported a strategy to access different topologies of redox-active poly(disulfide)s by ring-opening polymerization. Control over polymerization enables synthesis of high molecular-weight polymers. The polymers undergo catalytic depolymerization to recycle monomer; a promising feature for sustainable flow batteries. Read More
-
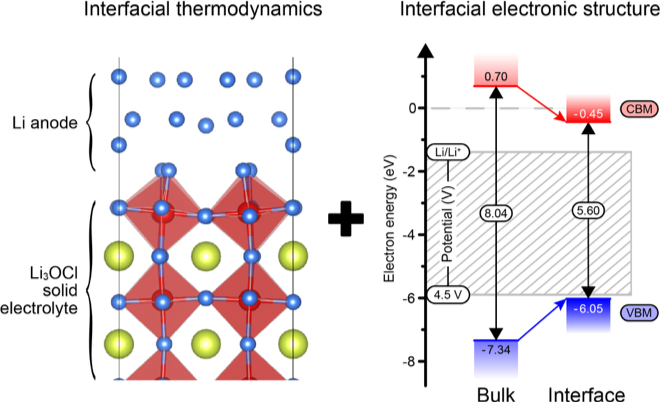
Predicting Wettability and the Electrochemical Window of Lithium- Metal/Solid Electrolyte Interfaces
This study demonstrates a comprehensive computational approach to predicting multiple electrode/electrolyte interfacial properties in solid-state batteries. Read More
-
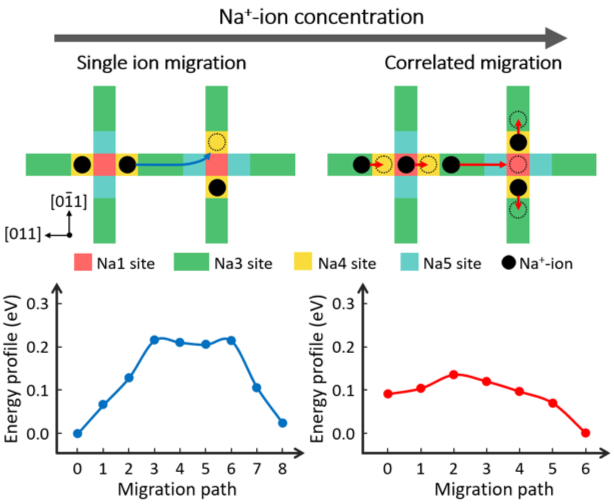
Correlated Migration Invokes Higher Na+‐Ion Conductivity in NaSICON‐Type Solid Electrolytes
Our theoretical studies reveal that correlated migration is the dominant conduction mechanism in NaSICON-type materials. This can be invoked by increasing the Na+-ion concentration, as confirmed by experimental X-ray diffraction and impedance studies, while neutron diffraction studies identified the existence of a new high energy Na site in the lattice. Most importantly, an important strategy was experimentally validated to enhance ion conductivity; namely by forcing cations into high-energy sites via repulsive forces, the energy barrier for cation migration can be lowered. Read More
Latest Updates
-
You’re Invited - JCESR and Beyond: Translating the Basic Science of Batteries
Please join us at Argonne National Laboratory on Tuesday, April 4, 2023 for JCESR and Beyond: Translating the Basic Science of Batteries. Registration is now open. This in-person event will celebrate 10 years of research from the Joint Center… Read More
-
A Message from JCESR: In Memory of George Crabtree
It is with heavy hearts that we say goodbye to George Crabtree, a Senior Scientist and Distinguished Fellow at Argonne National Laboratory, and Director of the Joint Center for Energy Storage Research (JCESR), who passed away unexpectedly on January 23. Dr. Read More
-
Cyanopyridines As Extremely Low-Reduction-Potential Anolytes for Nonaqueous Redox Flow Batteries
Discovery of a cyanophenylpyridine derivative with a very low reduction potential and good stability during cycling. Read More
-
Characterizing Redoxmer – Electrode Kinetics Using a SECM-Based Spot Analysis Method
Identified asymmetries in electron transfer (ET) kinetics between the reduction and oxidation of ferrocene-based redoxmers by measuring the ET rate constants (kf/kb) as a function of electrode potential. Read More
-
Benzotriazoles as Low Potential Anolytes for Non-Aqueous Redox Flow Batteries
We developed an easy-to-synthesize benzotriazole-based anolyte with a high energy redox potential (-2.3 V vs Fc/Fc+) and high solubility that demonstrates stable electrochemical cycling performance. Read More

