Redox Flow
-
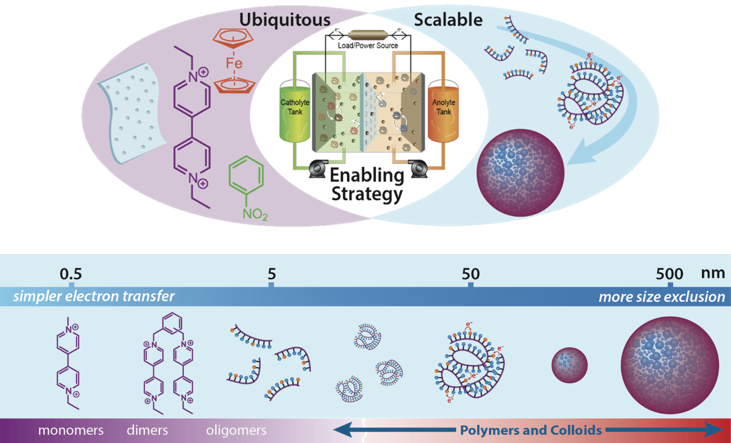
The Evolution of Redox Active Polymers as Soluble Nanomaterials for Energy Storage
Redox active polymers (RAPs) are highlighted for use as new soluble energy storage materials. This review article shines light on some of the major work from the RAP flow battery sprint in JCESR and provides perspective on future studies of RAPs. Read More
-
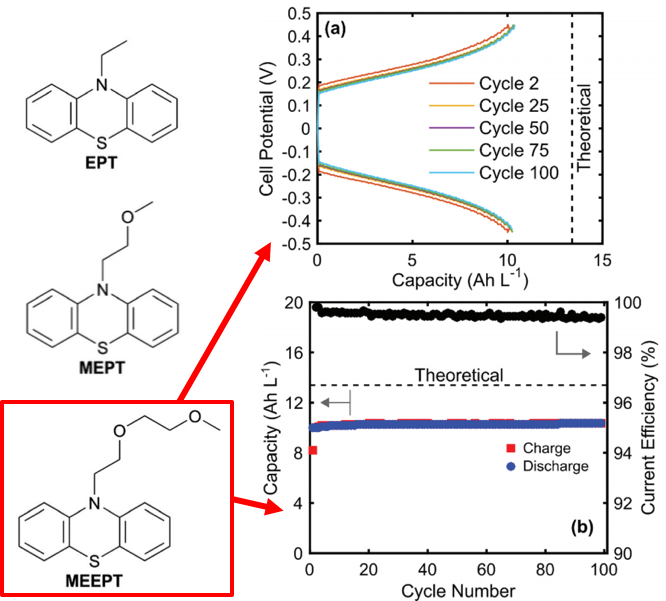
High Current Density, Long Duration Cycling of Soluble Organic Active Species for Non-Aqueous Redox Flow Batteries
We simultaneously engineered a soluble catholyte molecule and low resistance flow cell, enabling unprecedented high current density and undetectable capacity fade cycling of an organic molecule in a non-aqueous flow cell. Read More
-
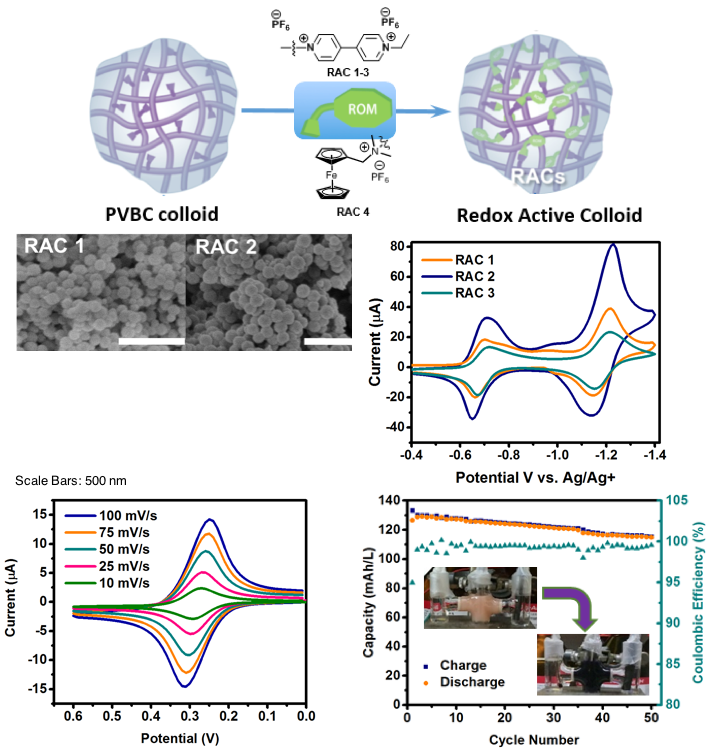
Redox Active Colloids as Discrete Energy Storage Carriers
Redox active colloids (RACs) were introduced as a promising class of energy storage materials. These were synthesized and electrochemically studied for their charge transfer properties as well as charge storage capabilities. Read More
-
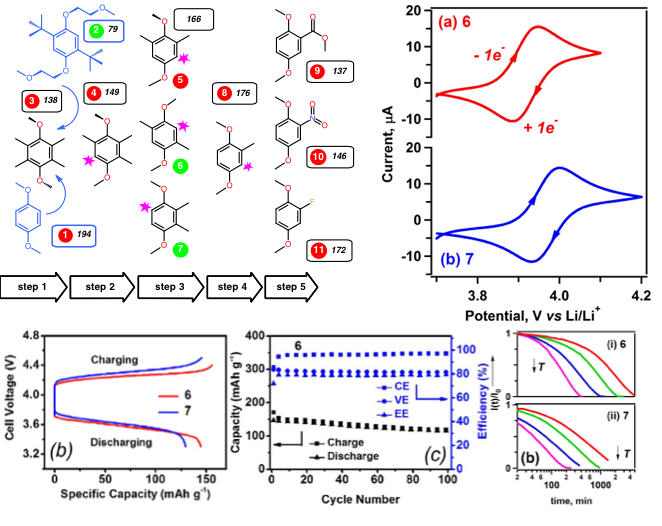
The Lightest Organic Radical Cation for Charge Storage in Redox Flow Batteries
By systematically shedding molecular fragments of 2,5-di-tert-butyl-1,4-bis(2-methoxyethoxy)benzene that are considered important for radical cation steric stabilization, we discovered a minimalistic structure that retains long-term stability in its oxidized form and exhibits the intrinsic capacity of 161 mAh/g. Read More
-
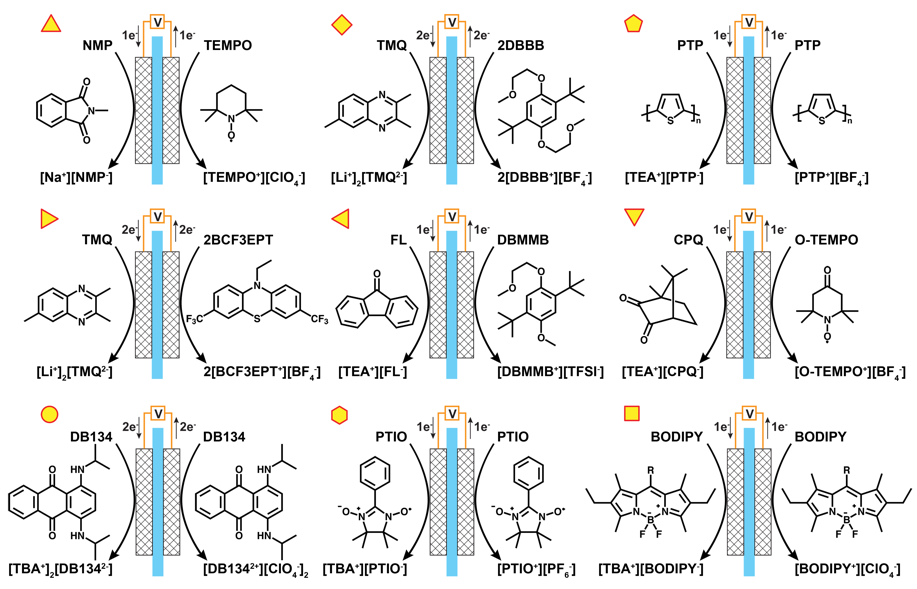
Recent Advances in Molecular Engineering of Redox Active Organic Molecules for Nonaqueous Flow Batteries
This review article summarizes the recent work on organic molecules for redox flow batteries, both redox couples and tailored redox active molecules. Read More
-

Molecular Dynamics Modeling of the Conductivity of Lithiated Nafion Containing Nonaqueous Solvents
Successfully predicted the ionic conductivities of lithiated Nafion membranes swollen in dimethyl sulfoxide (DMSO) and acetonitrile (ACN) using molecular dynamics and experimentally validated the results. Read More
-
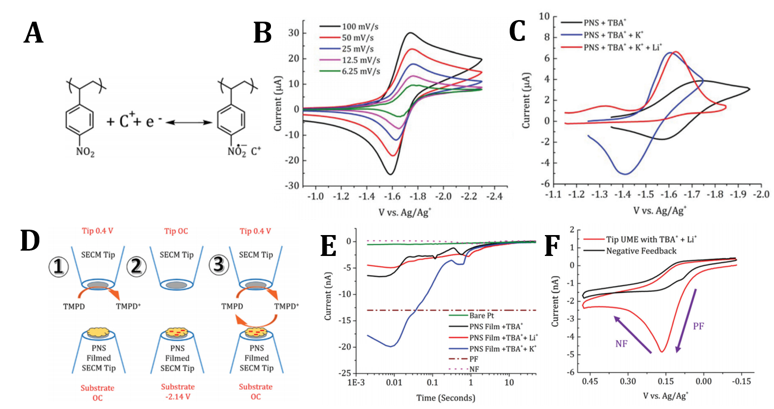
Enhancing the Performance of a Redox Active Polymer via Specific Electrolyte Interactions
A redox active polymer (RAP) with nitro- substituents was synthesized and studied via voltammetric analysis in combination with surface interrogation and ionic sensitive scanning electrochemical microscopy to reveal specific electrolyte interactions on the performance of the material. Read More
-
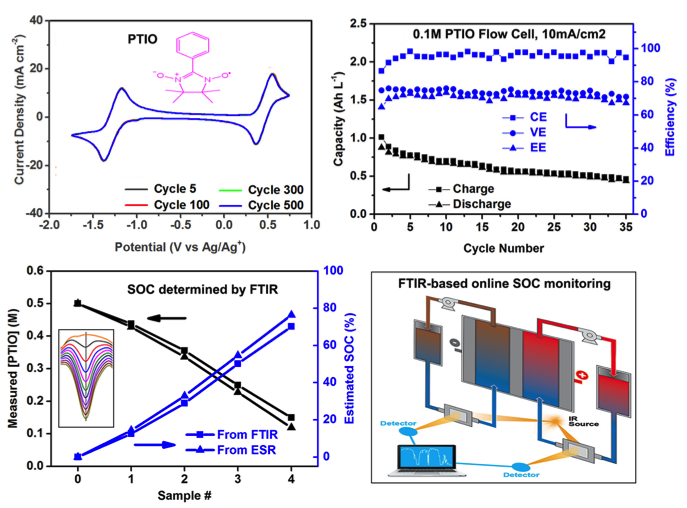
A Symmetric Organic - Based Nonaqueous Redox Flow Battery and Its State of Charge Diagnostics by FTIR
A symmetric nonaqueous flow battery based on the highly soluble, ambipolar PTIO achieved a cell voltage of ~1.7V and decent cyclability. We demonstrated FTIR as an effective method to monitor the state of charge (SOC) of this flow battery. Read More
-
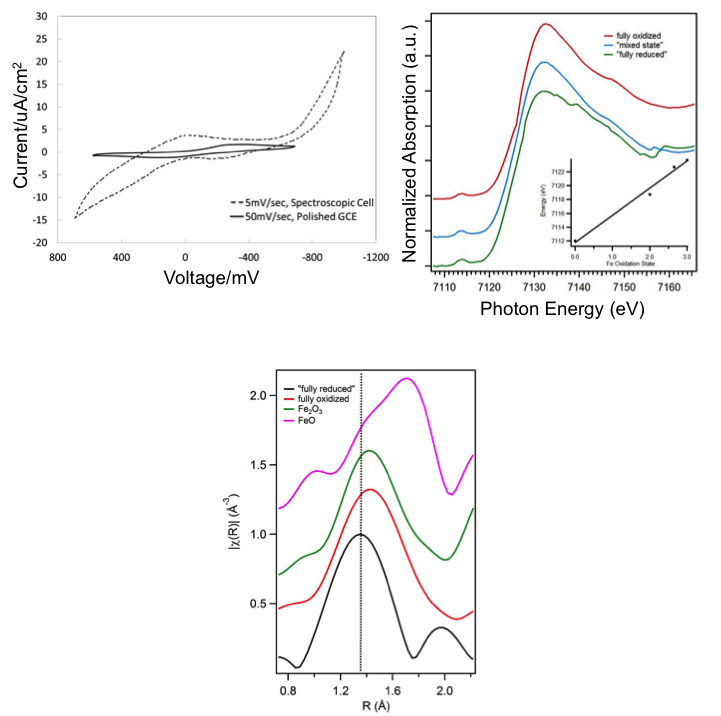
In-Situ XANES and EXAFS Analysis of Redox Active Fe Center Ionic Liquids
Redox active ionic liquids have been studied using in situ electrochemical and spectroscopic techniques to demonstrate conformational change in ligand structure during redox behavior of the iron center. Read More
-
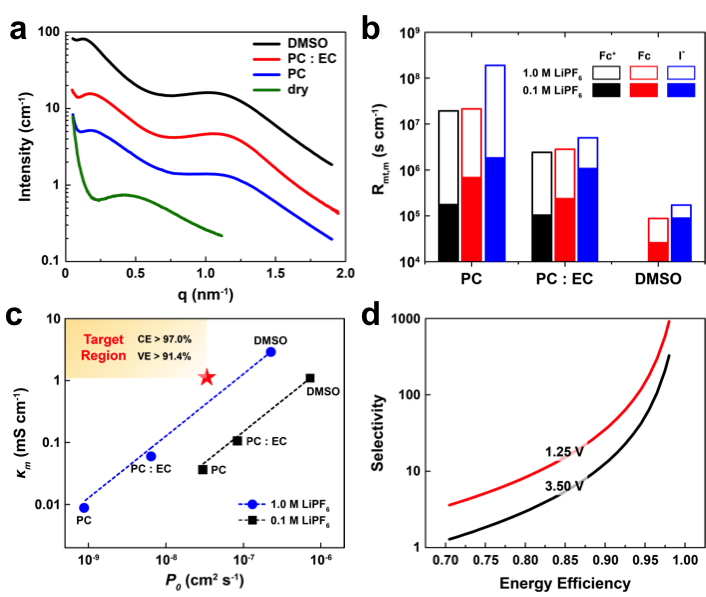
An Investigation of the Ionic Conductivity and Species Crossover of Lithiated Nafion 117 in Nonaqueous Electrolytes
Solvent volume fraction within the membrane has the greatest effect on both conductivity and crossover; as a secondary effect, the charge on redox species modifies crossover rates in accordance with Donnan exclusion. Read More
Latest Updates
-
You’re Invited - JCESR and Beyond: Translating the Basic Science of Batteries
Please join us at Argonne National Laboratory on Tuesday, April 4, 2023 for JCESR and Beyond: Translating the Basic Science of Batteries. Registration is now open. This in-person event will celebrate 10 years of research from the Joint Center… Read More
-
A Message from JCESR: In Memory of George Crabtree
It is with heavy hearts that we say goodbye to George Crabtree, a Senior Scientist and Distinguished Fellow at Argonne National Laboratory, and Director of the Joint Center for Energy Storage Research (JCESR), who passed away unexpectedly on January 23. Dr. Read More
-
Cyanopyridines As Extremely Low-Reduction-Potential Anolytes for Nonaqueous Redox Flow Batteries
Discovery of a cyanophenylpyridine derivative with a very low reduction potential and good stability during cycling. Read More
-
Characterizing Redoxmer – Electrode Kinetics Using a SECM-Based Spot Analysis Method
Identified asymmetries in electron transfer (ET) kinetics between the reduction and oxidation of ferrocene-based redoxmers by measuring the ET rate constants (kf/kb) as a function of electrode potential. Read More
-
Benzotriazoles as Low Potential Anolytes for Non-Aqueous Redox Flow Batteries
We developed an easy-to-synthesize benzotriazole-based anolyte with a high energy redox potential (-2.3 V vs Fc/Fc+) and high solubility that demonstrates stable electrochemical cycling performance. Read More

