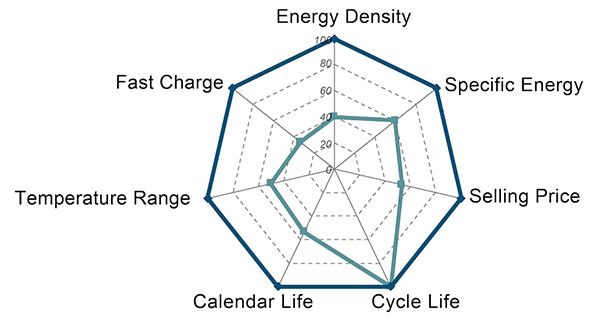
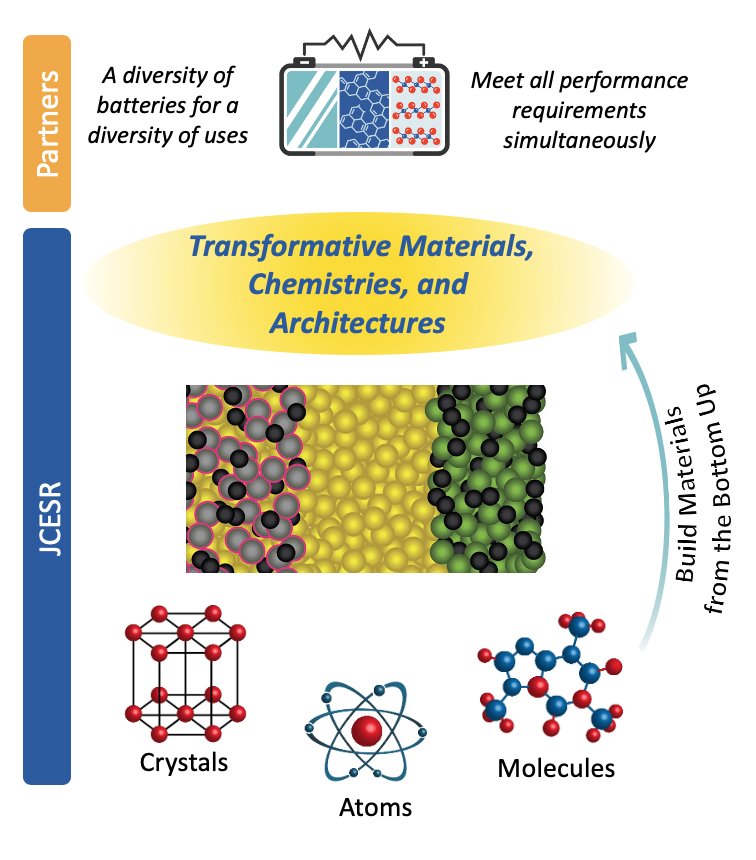
Today’s batteries meet only some of their targeted performance metrics. Meeting all of the metrics for a given application requires new materials with transformative behavior, such as simultaneously achieving high reactivity, high mobility and high stability in electrodes, or high selectivity and stability at low cost in membranes. Such transformative materials are not readily available in nature; they must be designed based on an intimate knowledge of the atomic and molecular origins of the targeted behavior.
Imagine the impact on battery science and technology of a unified description of ion solvation in liquids and solids at the atomic level, a template for designing redox active multimers (redoxmers) that allows adjusting reactivity, solubility, and stability independently, or a full understanding of the roles of defects in crystals and heterogeneity at interfaces on overall materials behavior.
Breakthroughs in these areas would permanently alter the face of battery and electrochemical science and would lead to a blossoming of new materials with unique electroactive function. Most importantly, these three fundamental breakthroughs would usher in a new era of constructionist “bottom up” atom-by-atom and molecule-by-molecule materials design, with each atom or molecule playing a prescribed role in producing a targeted overall behavior.
The bottom up approach enables making materials that simultaneously meet multiple performance requirements that are often anti-correlated such as high reactivity and long lifetime, a pervasive challenge for electrochemistry. JCESR takes concrete steps toward achieving fundamental breakthroughs like these and demonstrating a new bottom up constructionist approach to designing and discovering electrochemical materials.
To achieve these objectives JCESR is organized around five research Thrusts that, taken together, will create transformative materials that meet all the performance metrics for a given application. These Thrusts and the specific science directions are: Liquid Solvation Science, Solid Solvation Science, Flowable Redoxmer Science, Charge Transfer at Dynamic Interfaces, and Science of Material Complexity.