Latest Updates
-
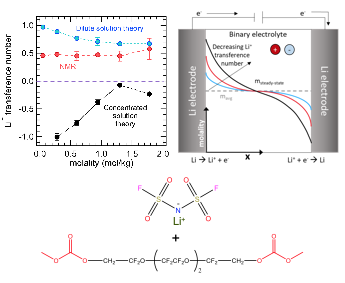
Complete electrochemical characterization of a fluorinated electrolyte
Common approaches to measuring the lithium-ion transference number for fluorinated electrolytes leads to severe over-estimation of this value. Read More
-
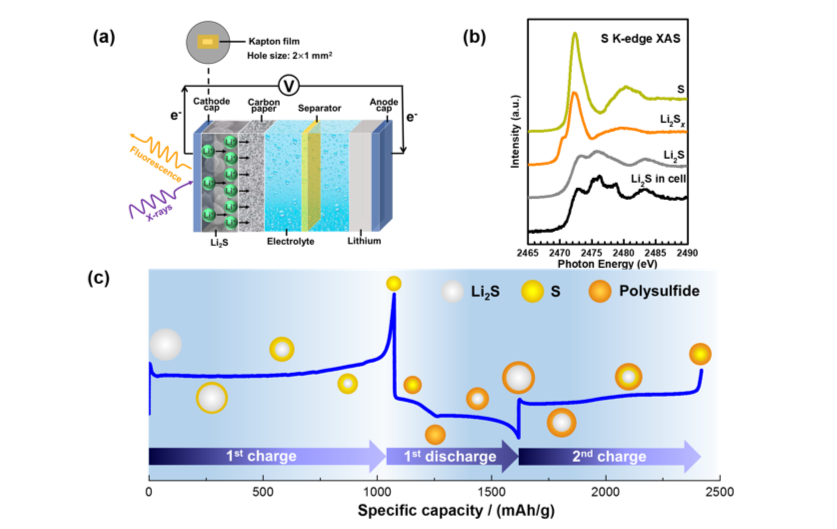
Understanding the Reaction Mechanism of Lithium-Sulfur Batteries by In situ/Operando XAS
We presented the recent progress in the application of in situ/operando XAS in the understanding of the redox mechanism of lithium sulfur batteries, which is of great significance for the development of high-performance lithium sulfur batteries. Read More
-
PyLAT: Python LAMMPS Analysis Tools
A series of tools was developed to perform post-analysis on molecular dynamics trajectories from the LAMMPS software. Read More
-
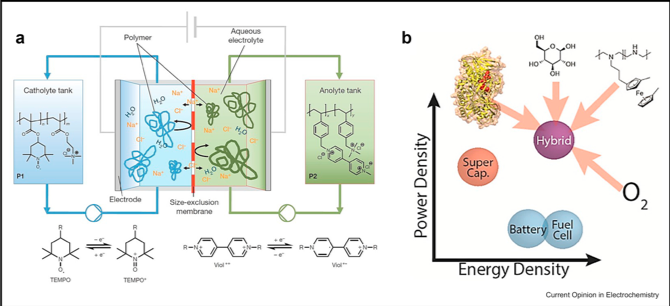
Redox Polymers in Electrochemical Systems: From Methods of Mediation to Energy Storage
Redox polymers were designed for mediation in sensors in the 1980s, but it was not until the last five years that researchers realized that they could play a critical role in energy storage, including redox flow batteries and supercapacitors. Read More
-
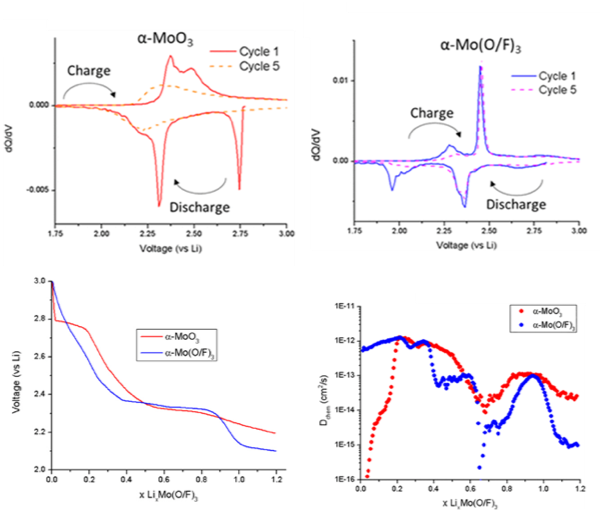
Fluoride Doping Enhances Lithium Diffusivity in Layered Molybdenum Oxide
By fluoride doping MoO3 to MoO2.8F0.2 the reversibility of lithium intercalation could be greatly increased and a sluggish intercalation step could be bypassed. Read More
-
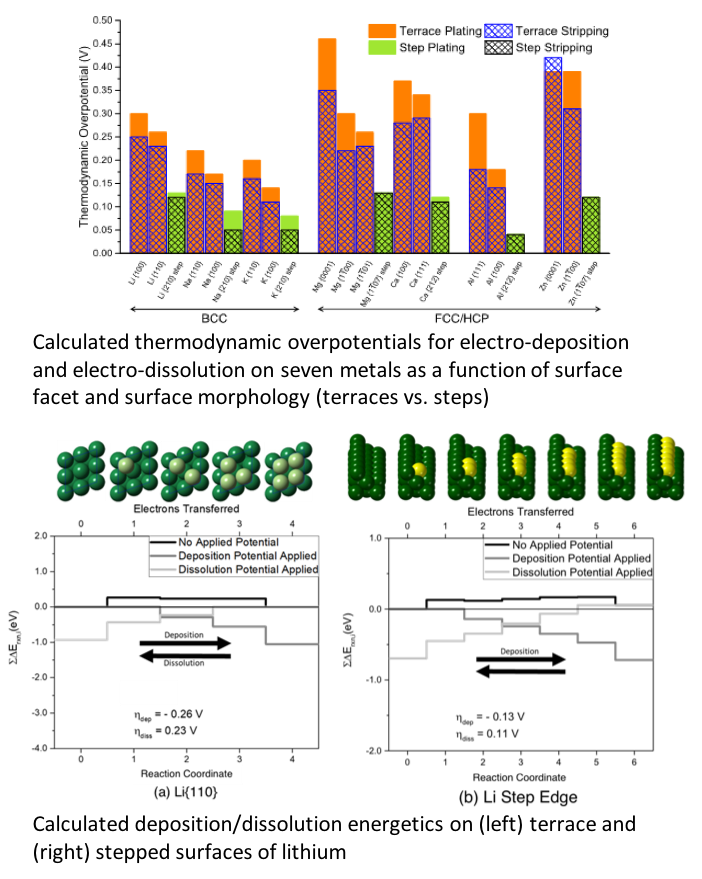
Thermodynamic Overpotentials and Nucleation Rates for Electrodeposition on Metal Anodes
Thermodynamic contributions to overpotentials are predicted for seven metals relevant for next-generation metal anode batteries: Al, Ca, K, Li, Mg, Na, and Zn. In addition, a multi-scale model that combines these predictions with nucleation theory is used to estimate nucleation rates for electrodeposits on these electrodes. Read More
-
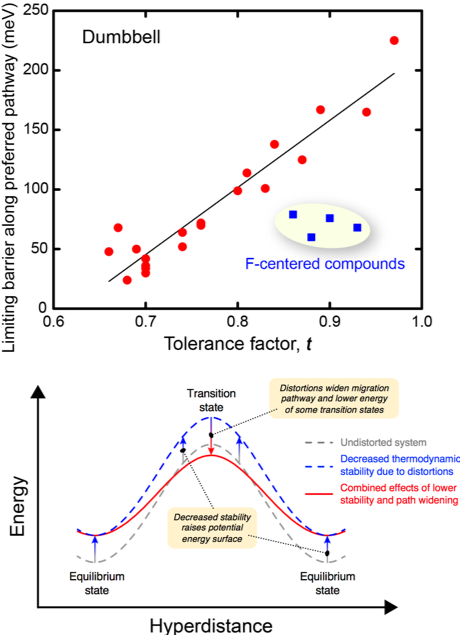
Correlating Lattice Distortions, Ion Migration Barriers, and Stability in Solid Electrolytes
The discovery of solid electrolytes remains an important scientific goal. This search has been slowed, however, by incomplete understanding of the chemical and structural features that give rise to high ionic mobility. Read More
-
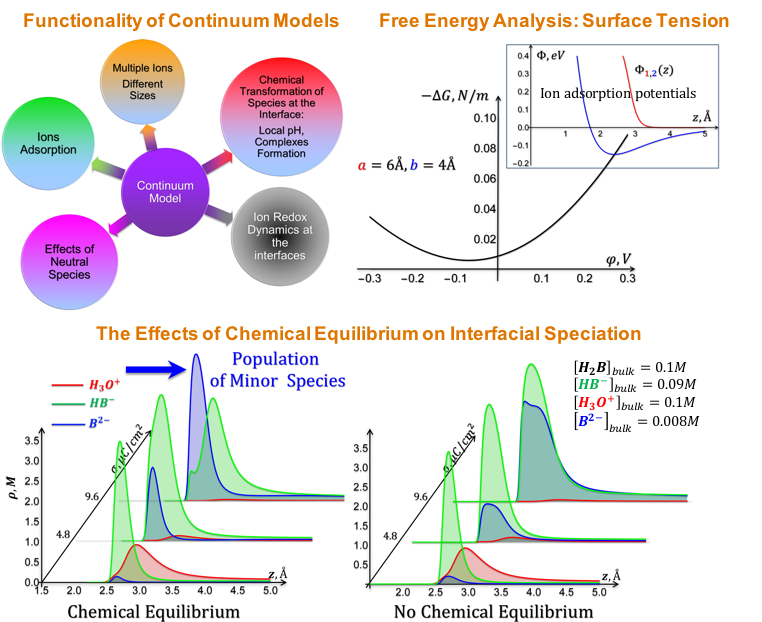
Advanced Continuum Models for Simulations of Electrified Interfaces
Development of a novel formalism and methodology to model electrolyte composition at electrified (electrode) interfaces. Predictions that minor species (from the bulk perspective) may become dominant at the electrified interface. Read More
-
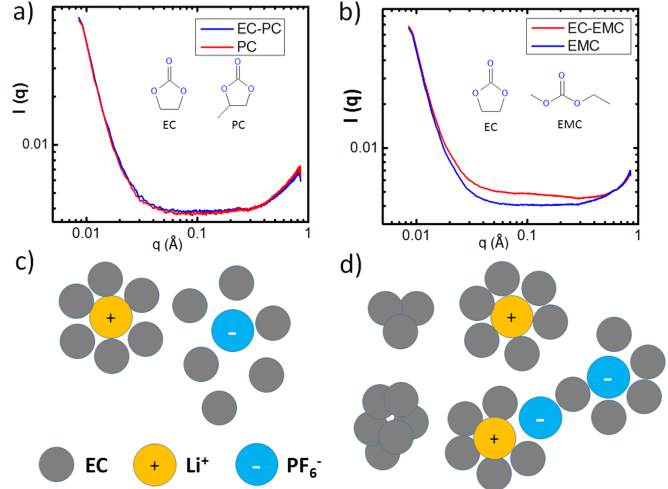
Microscopic View of the Ethylene Carbonate Based Lithium-Ion Battery Electrolyte by Small Angle X-ray Scattering
We report, for the first time, the observation of EC nanoclusters in both EC/EMC solvent mixture and the LiPF6/EC/EMC electrolyte through Small Angle X-ray Scattering (SAXS) studies. We detect and estimate the size of LiPF6 ion pair in these carbonate-based electrolytes. Read More
-
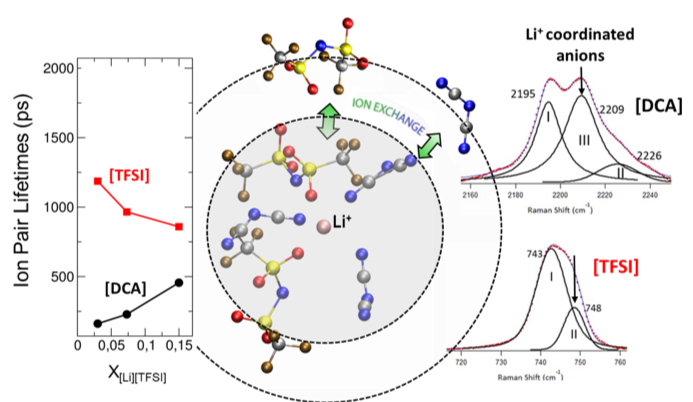
Solvation Structure and Dynamics of Li+ in Ternary Ionic Liquid-Lithium Salt Electrolytes
The local structure of Li+ in this eutectic is found to be heterogenous and preferentially solvated by [DCA], which is related to the transport properties. Read More
-
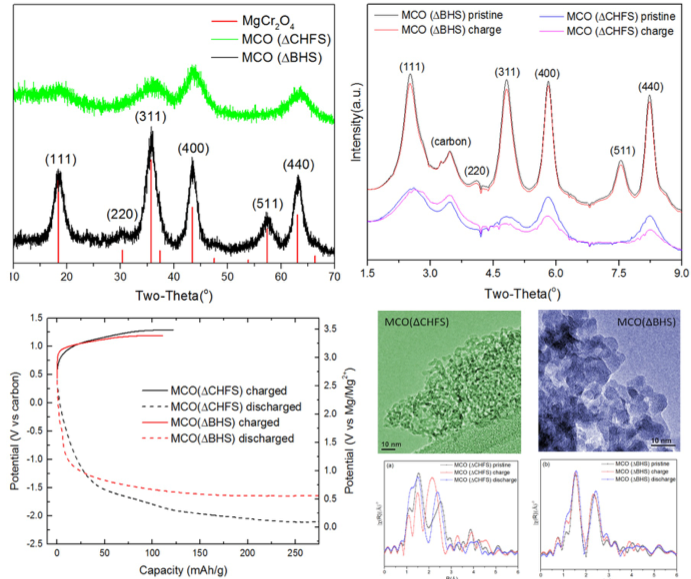
Tailoring the Electrochemical Activity of Magnesium Chromium Oxide Towards Mg Batteries Through Control of Size and Crystal Structure
Reversible magnesium removal from MgCr2O4 was induced by nanosizing and introducing significant structural defects to reduce diffusion the distance and overcome the activation energy barrier. Read More
-
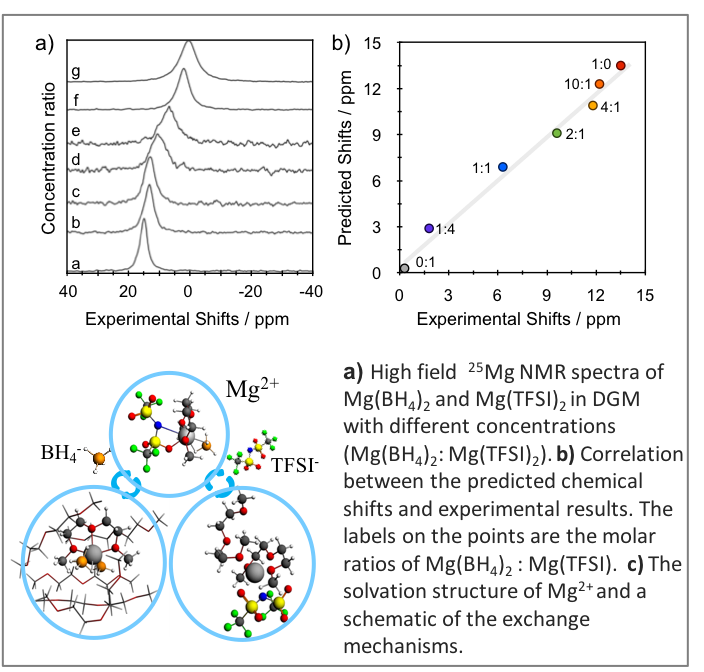
Topical Review: In Situ and Ex Situ NMR for Battery Research
We review scientific studies where nuclear magnetic resonance (NMR) proves an indispensable tool for obtaining detailed molecular level understanding of electrochemical processes for energy storage. Featured advances include in situ NMR analyses where the chemistries within a functioning battery are probed during cell cycling. Read More
-

Technology
JCESR’s focus has changed to building transformational materials from the bottom up, atom-by-atom and molecule-by-molecule, where each atom or molecule plays a prescribed role in producing the desired overall materials performance. We could not have done this 10 years ago when the advanced scientific… Read More
-

Team Approach
JCESR is a collaborative team of engineers and scientists with very broad backgrounds. In the battery space we are now facing challenges that required a multidisciplinary approach that no single group can achieve. Made up of 18 partner institutions, JCESR’s diversity and the opportunity… Read More
-

Renewed Focus
JCESR has had a very successful first five years. The personal relationships we’ve formed now enable us to move forward with even more momentum. Recently, the team of more than 150 came together for its first full program meeting since renewal. As stated by… Read More
-
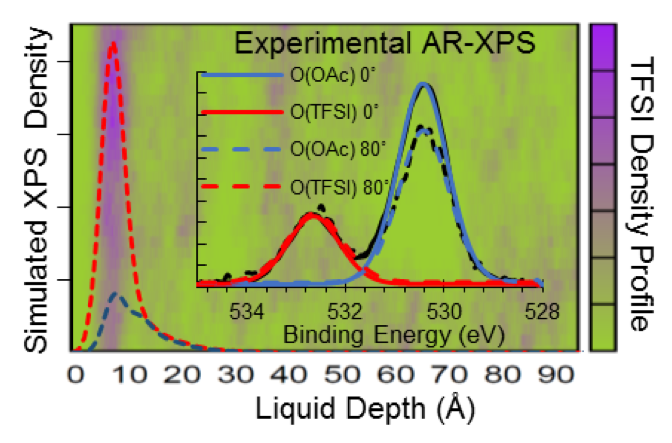
Anion Enhancement at the Liquid-Vacuum Interface of an Ionic Liquid Mixture
IL-vacuum interface was studied and a quantitative connection between molecular simulations and angle-resolved X-ray photoemission was achieved. Read More
-

Simulation and Measurement of Water-induced Liquid-liquid Phase Separation of Imidazolium Ionic Liquid Mixtures
Computationally predicted liquid-liquid phase equilibrium confirmed by experimental measurements. Read More
-
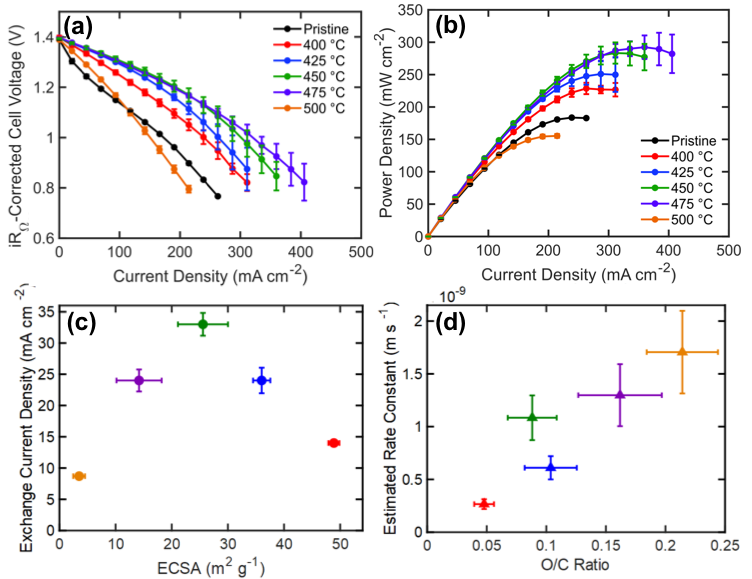
Elucidating the Nuanced Effects of Thermal Pretreatment on Carbon Paper Electrodes for Vanadium Redox Flow Batteries
The chemical and physical properties of carbon paper electrodes treated at temperatures between 400-500 °C for 30 hours were quantified, and the subsequent impact of these properties on the performance of vanadium redox flow batteries explained. We find that the oxygen content and electrochemically active surface area, two parameters that govern the kinetic performance of the vanadium flow battery, follow opposing temperature trends; thus, optimal performance is achieved at a balance of these properties rather than a maximization of each. Read More
-
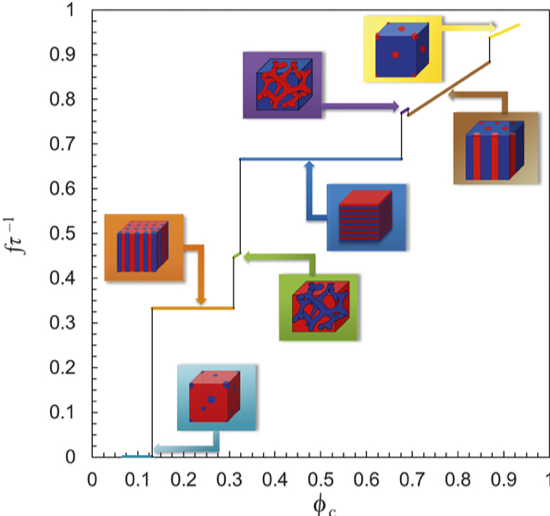
Unified Platform for Ion Transport in Inorganic Glasses, Polymers and Composite Solid Electrolytes
In this review paper, ion transport parameters in seemingly different solid electrolytes – glasses, polymers, and composites - were presented on a unified platform. Read More
-
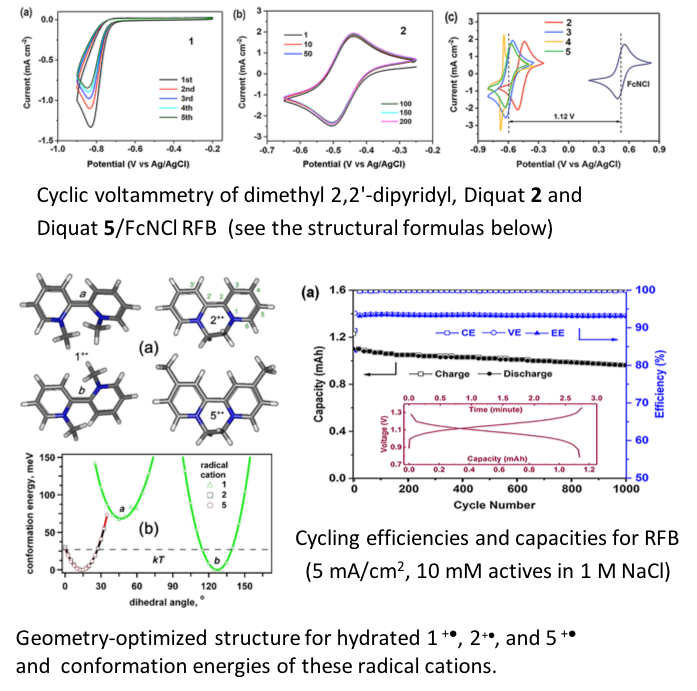
Diquat Anolytes for Sustainable Use of Grid-Scale Aqueous Redox Flow Batteries
Diquats are shown to be exceptionally stable anolyte (negative charge storing) materials for aqueous redox flow batteries (RFBs) having low redox potentials, extremely stable reduced states, and outstanding electrochemical cycling performance (>1,000 cycles with 0.016% capacity loss per cycle). Read More

