Latest Updates
-
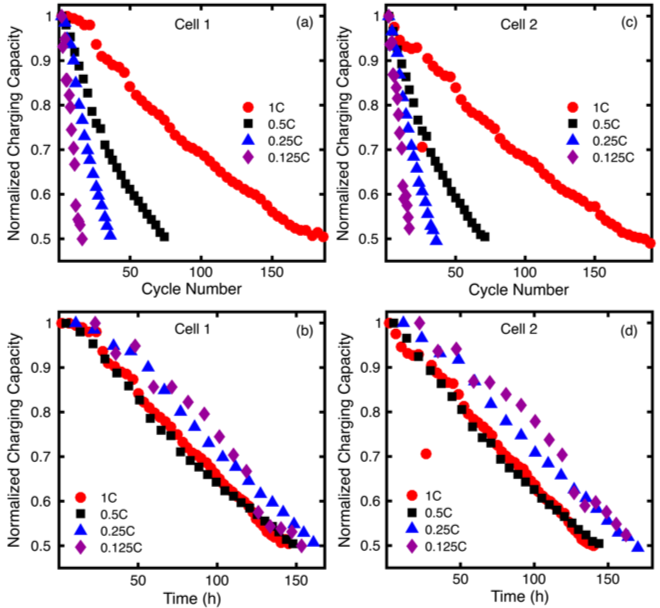
The impact of bulk electrolysis cycling conditions on the perceived stability of redox active materials
Under bulk electrolysis conditions, cycle time is a better measure of stability than cycle number and, for materials that are unstable in their charged state, the fractional capacity accessed is inversely related to cycle time until failure. Read More
-
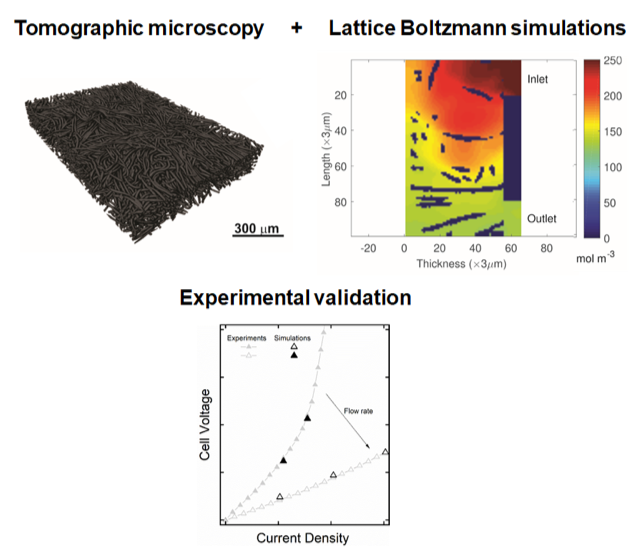
Experimentally validated 3D pore-scale lattice Boltzmann model for understanding porous electrode microstructure
Three dimensional (3D) multi-physics models of porous carbon electrodes are employed to understand the role of electrode microstructure on redox flow battery performance. Read More
-
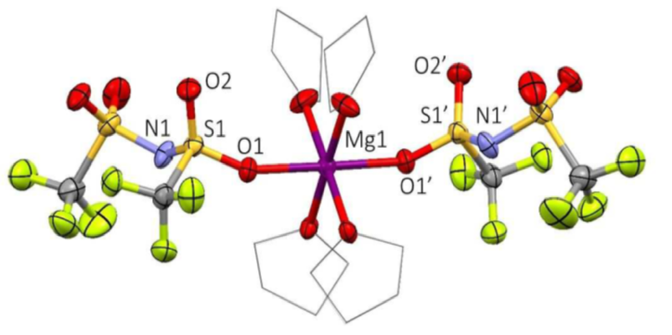
A Simple Halogen-free Magnesium Electrolyte for Reversible Magnesium Deposition through Co-solvent Assistance
Electrolytes for Mg-ion batteries are a complex mixture of salts, solvents, and additives that allow for the free-movement of Mg-ions across the cell. By addition of simple amines to the mixture, the internal equilibriums are shifted to allow formation of a simpler neutral complex in solution that does not require use of corrosive halide anions (Figure 1). Read More
-
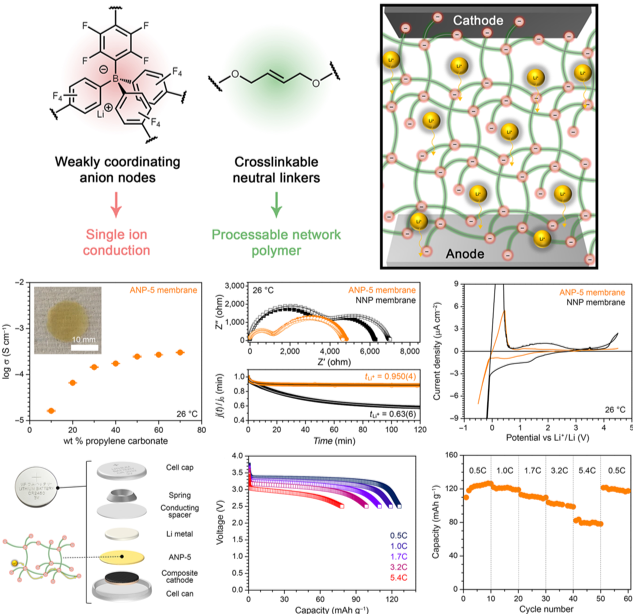
A Single-Ion Conducting Borate Network Polymer as a Viable Quasi-Solid Electrolyte for Lithium Metal Batteries
An anionic network polymer, ANP-5, membrane exhibits high room temperature ionic conductivity and selectivity for Li+ ion conduction, outstanding electrochemical stability, and flame retardancy. Read More
-
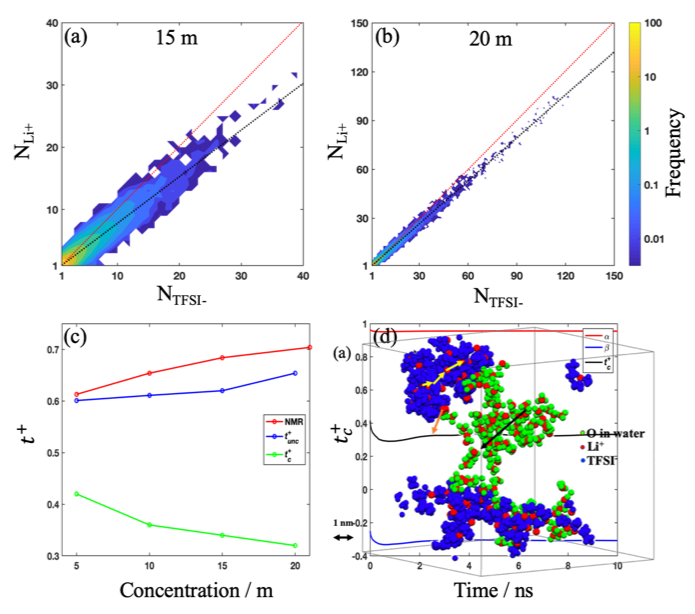
Asymmetric Composition of Ionic Aggregates and the Origin of High Correlated Transference Number in Water-in-Salt Electrolytes
MD simulations revealed the heterogeneous structure of ion and water domains/aggregates and fairly high correlated transference number in the water-in-salt electrolytes. Read More
-

Probing Electrolyte Structure at Interfaces: An Example of the Power of Variable Temperature and Pressure Operando MAS NMR
We have developed a robust method to analyze battery systems under finely-controlled environments of specified composition, temperature, and pressure to reveal the interactions of electrolytes with electrode surfaces. Read More
-
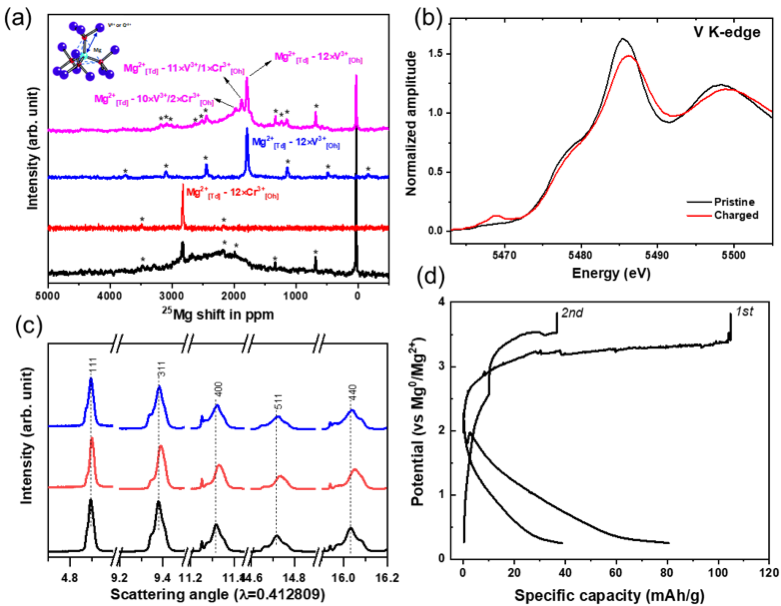
Electrochemical Mg-ion Activity in MgCr2-xVxO4 Spinel Oxides
Bulk demagnesiation in MgCrVO4 spinel lattice is observed in a full-cell configuration paired with a Mg metal anode in a chemically and anodically stable Mg(TPFA)2 electrolyte at 110 oC. Read More
-
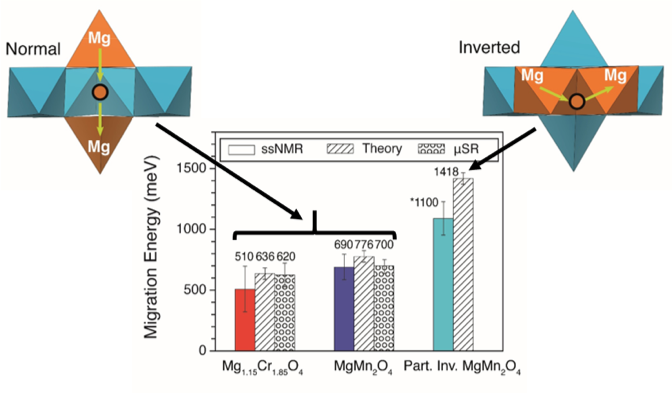
Probing migration of magnesium cations in spinel oxides
Measurements of 25Mg variable temperature solid-state nuclear magnetic resonance (VT ss-NMR), and muon spin relaxation (μSR) in 3 spinel oxides reveal experimental magnesium hoping barriers as low as ∼0.6 eV, in agreement with independent density functional theory (DFT) predictions. Read More
-
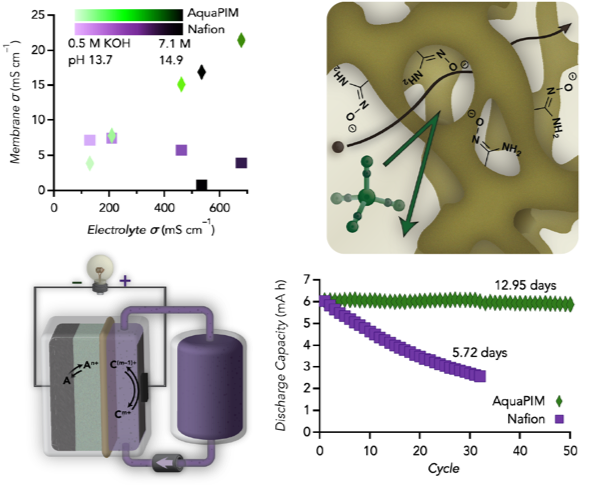
Designing Membranes for Aqueous Alkaline Flow Batteries from Polymers of Intrinsic Microporosity
The incorporation of amidoxime units along the rigid backbone of a polymer of intrinsic microporosity to make them aqueous-compatible (AquaPIMs) allows for exceptional conductivity and stability in harsh alkaline environments while blocking a variety of active materials. Read More
-
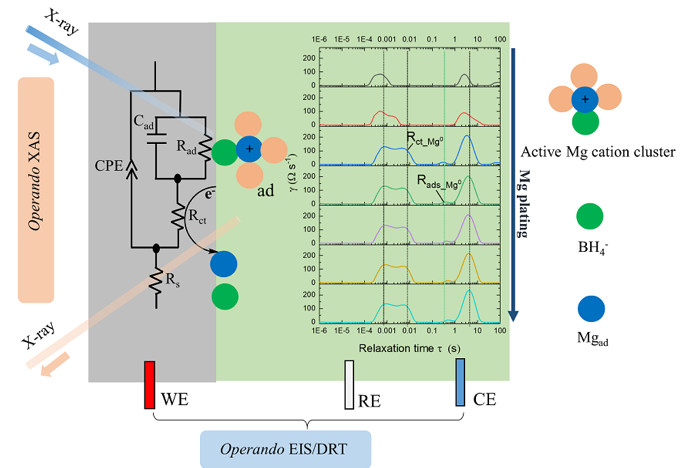
Reversible Electrochemical Interface of Mg Metal and a Conventional Electrolyte Enabled by Intermediate Adsorption
A detailed description of the complex charge-transfer process at a Mg/electrolyte interface is provided where an additional adsorption process, during Mg plating, is confirmed to be a key step. Read More
-
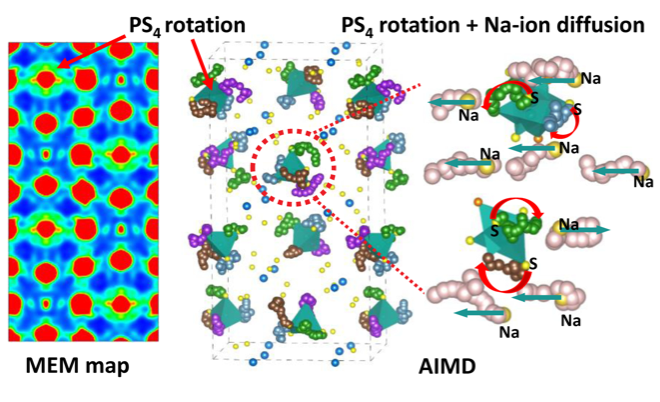
Coupled Cation-Anion Dynamics Enhances Cation Mobility in Room Temperature Superionic Solid-State Electrolytes
Our work reveals facile [PX4]3- anion rotation in superionic Na11Sn2PS12 and Na11Sn2PSe12, and greatly hindered [SbS4]3- rotational dynamics in their less conductive analogue, Na11Sn2SbS12. Along with introducing dynamic frustration in the energy landscape, the fluctuation caused by [PX4]3- anion rotation is firmly proved to couple to, and facilitate long range Na+-cation mobility, by transiently widening the bottlenecks for Na+-ion diffusion. Read More
-
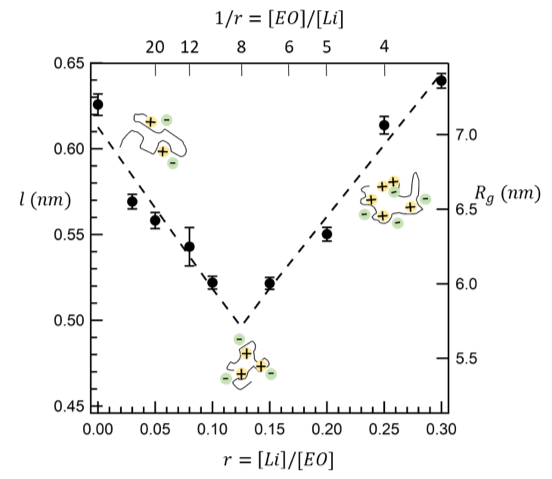
The Effect of Added Salt on the Size of Polymer Chains in Electrolytic Mixtures
The effect of salt concentration on the molecular conformation of a polymer was determined by small angle neutron scattering. Read More
-
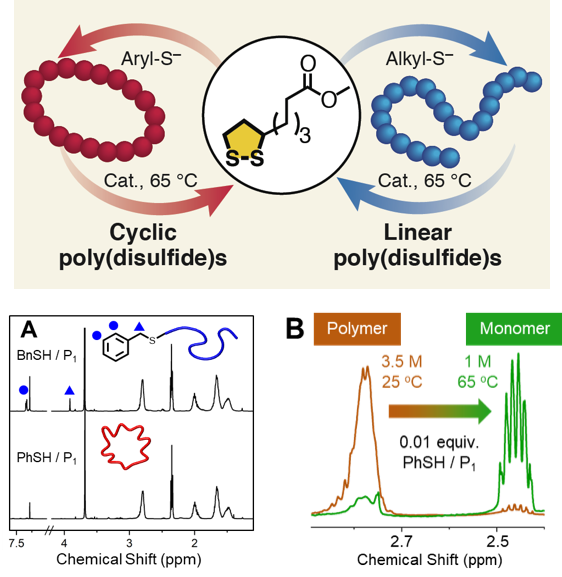
Architecture-Controlled Ring-Opening Polymerization for Dynamic Covalent Poly(disulfide)s
We reported a strategy to access different topologies of redox-active poly(disulfide)s by ring-opening polymerization. Control over polymerization enables synthesis of high molecular-weight polymers. The polymers undergo catalytic depolymerization to recycle monomer; a promising feature for sustainable flow batteries. Read More
-
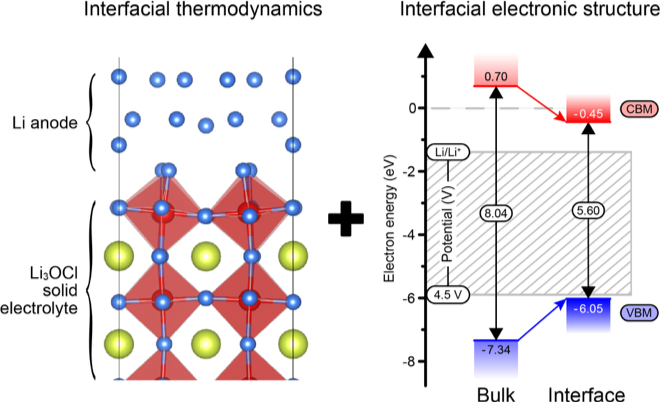
Predicting Wettability and the Electrochemical Window of Lithium- Metal/Solid Electrolyte Interfaces
This study demonstrates a comprehensive computational approach to predicting multiple electrode/electrolyte interfacial properties in solid-state batteries. Read More
-
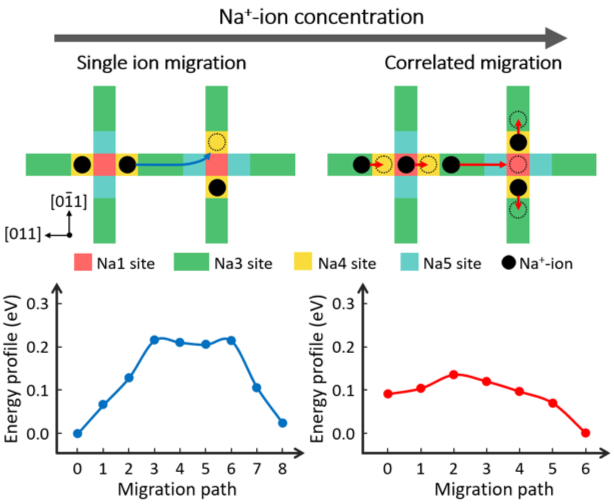
Correlated Migration Invokes Higher Na+‐Ion Conductivity in NaSICON‐Type Solid Electrolytes
Our theoretical studies reveal that correlated migration is the dominant conduction mechanism in NaSICON-type materials. This can be invoked by increasing the Na+-ion concentration, as confirmed by experimental X-ray diffraction and impedance studies, while neutron diffraction studies identified the existence of a new high energy Na site in the lattice. Most importantly, an important strategy was experimentally validated to enhance ion conductivity; namely by forcing cations into high-energy sites via repulsive forces, the energy barrier for cation migration can be lowered. Read More
-
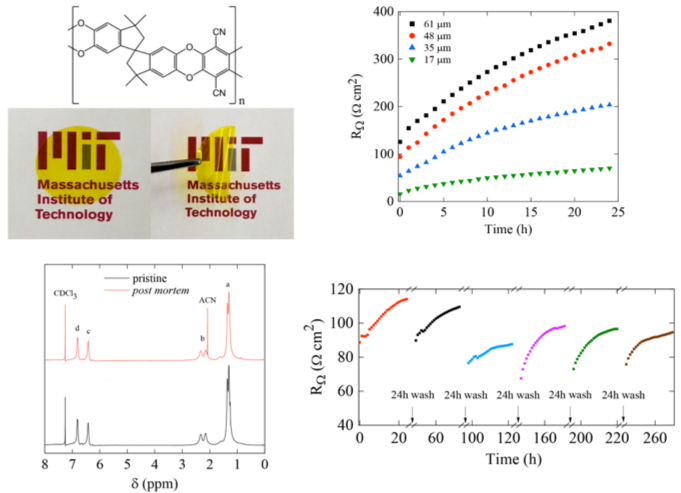
Investigating the factors that influence resistance rise of PIM-1 membranes in nonaqueous electrolytes
PIM-1 membranes undergo time-dependent resistance rise upon contact with flowing nonaqueous electrolytes; however, the increase in resistance is reversible and performance can be recovered by solvent washing. Read More
-
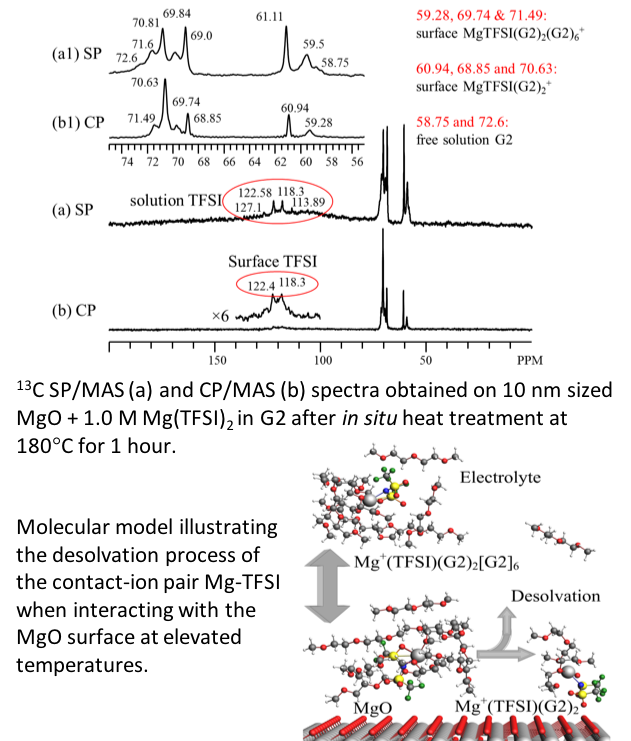
Adsorption and Thermal Decomposition of Electrolytes on Nanometer Magnesium Oxide: An in Situ 13C MAS NMR Study
The structural and chemical evolution of electrolyte constituents at the nanometric MgO surface were identified, providing a fundamental understanding of heterogeneous interphase evolution. Read More
-
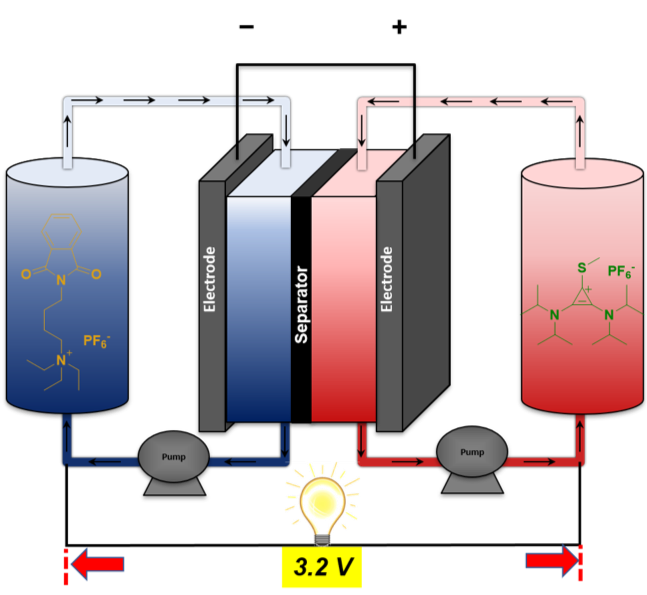
Mechanism-Based Design of a High-Potential Catholyte Enables a 3.2 V All-Organic Nonaqueous Redox Flow Battery
Development of an extremely high-potential catholyte leads to the first 3.2 V all-organic flow battery. Read More
-
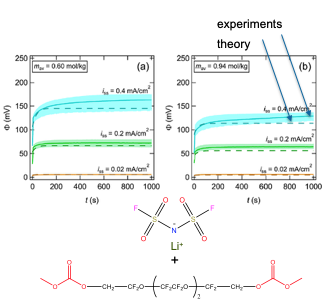
Comparing predictions and experiments for continuum ion transport fluorinated electrolyte
Experimentally measured ion transport rate in a fluorinated electrolyte is in quantitative agreement with theory from the dilute limit up to the salt solubility limit. Read More
-
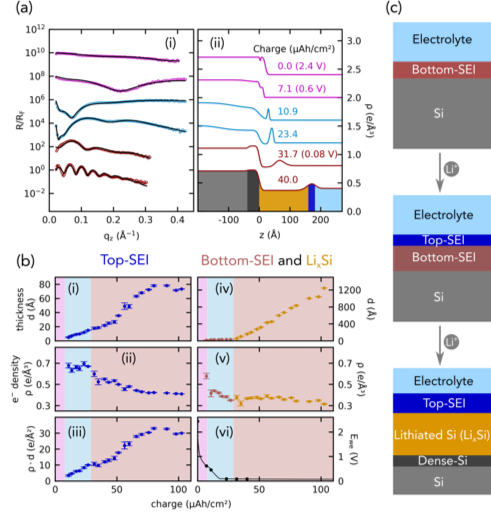
Shedding X‑ray Light on the Interfacial Electrochemistry of Silicon Anodes for Li-Ion Batteries
Our results shed light on the interfacial electrochemistry of silicon anodes for Lithium-ion batteries (LiBs), providing important mechanistic insight into nanometer scale phenomena and how these influence battery performance. Read More

