Scientific Achievement
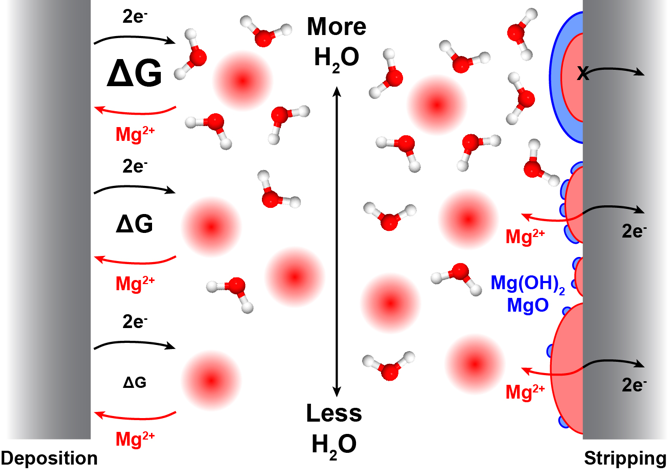
Trace amounts of water (≤ 3 ppm) are shown to strongly influence both the kinetics of Mg deposition and the reversibility of Mg stripping, with Cl- mitigating passivation by H2O by strongly modifying the Mg surface chemistry.
Significance and Impact
Understanding the underlying phenomena that govern surface chemistry can point to new strategies for designing new electrolytes for more efficient and reversible Mg-ion batteries.
Research Details
- As H2O content increases, (i) the Mg surface becomes increasingly oxidized eventually blocking reversible stripping (schematic, right), and (ii) the kinetics of Mg desolvation during deposition become more sluggish due to higher energy required to desolvate Mg2+ from tightly-bound H2O molecules (schematic, left).
- Cl- ions strongly adsorb to the Mg surface, preventing passivation by H2O oxidation and improving the reversibility of Mg anodes.

