Research Highlights
-
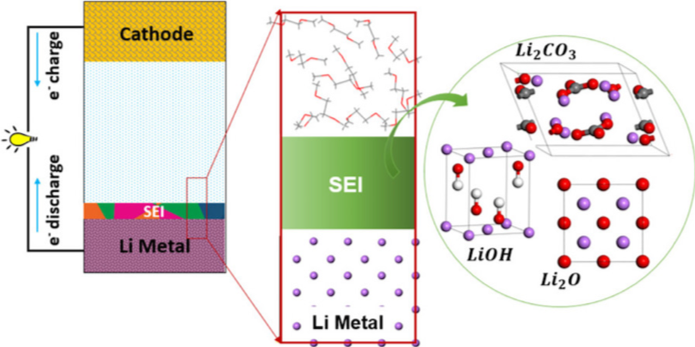
Roles of inorganic surface layers on solid electrolyte interphase evolution at Li-metal anodes
We uncovered the effects of nanometer-sized lithium oxide, lithium hydroxide, and lithium carbonate as surface passivation layers on the interfacial reactivity of Li-metal anodes. Read More
-
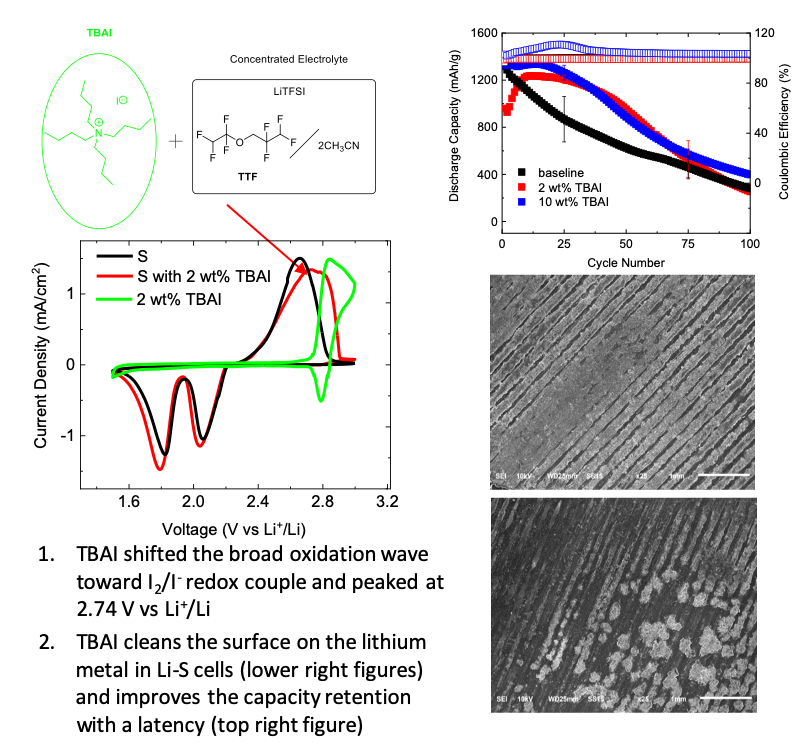
Lipophilic Additives for Highly Concentrated Electrolytes in Lithium-Sulfur Batteries
Developed a lipophilic additive, tetrabutylammonium iodide (TBAI), for highly concentrated electrolytes and tested its electrochemical performance in lithium-sulfur batteries. Read More
-
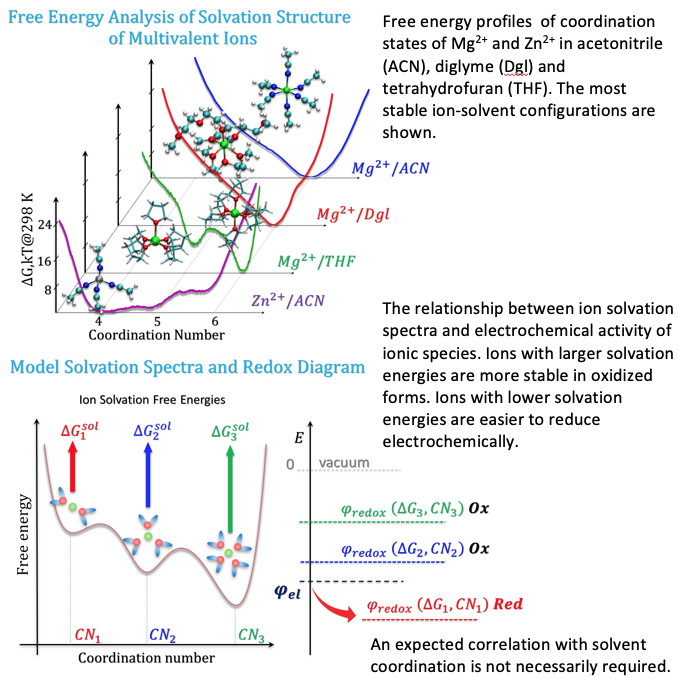
“Ion Solvation Spectra”: Free Energy Analysis of Solvation Structures of Multivalent Cations in Aprotic Solvents
Advanced free-energy sampling techniques reveal that monoatomic divalent cations in organic solvents may have multiple well-defined minima or plateaus of free energy with respect to the number of coordinating solvent molecules. We introduce the concept of “ion solvation spectra” to highlight non-trivial phenomenology related to a continuum of ion solvation environments, as opposed to a single coordination minimum. Read More
-

Modified Ohm’s Law in Lithium and Beyond-lithium Battery Electrolytes
Battery electrolytes contain two mobile charged species of opposite charge and thus the traditional Ohm’s law, which applies to one mobile charged species, must be modified. This review uses a modified Ohm’s law to rank order electrolytes. Read More
-
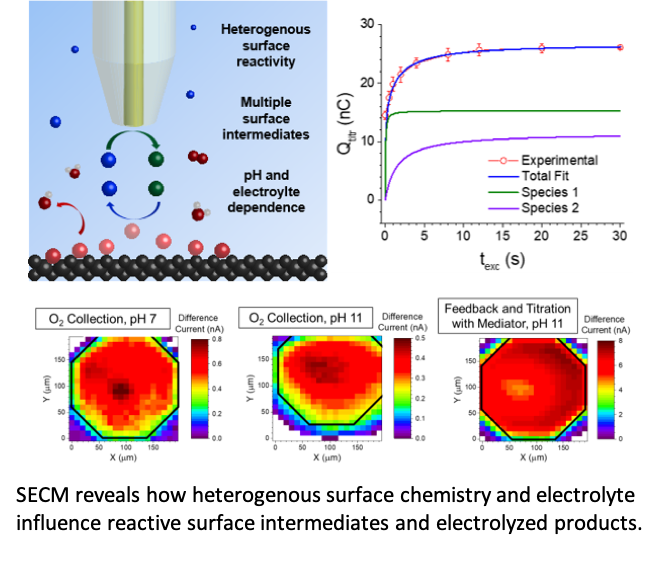
Probing Reactive Oxygen Species on Boron-Doped Diamond Anodes
Multiple surface oxygen species form and react at different rates on sp2 carbon-containing boron-doped diamond electrodes during water oxidation. Read More
-
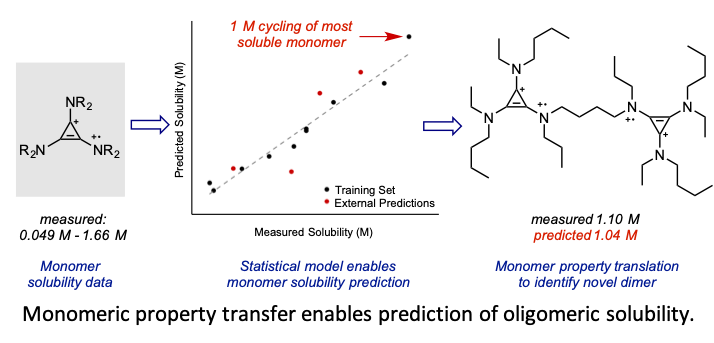
Developing a Predictive Solubility Model for Monomeric and Oligomeric Cyclopropenium-Based Flow Battery Catholytes
Two important field-wide goals are met through this study: the development of a statistical model for the solubility of conformationally flexible molecules in acetonitrile and the operation of a high concentration (1 M) nonaqueous organic symmetric flow cell. Read More
-
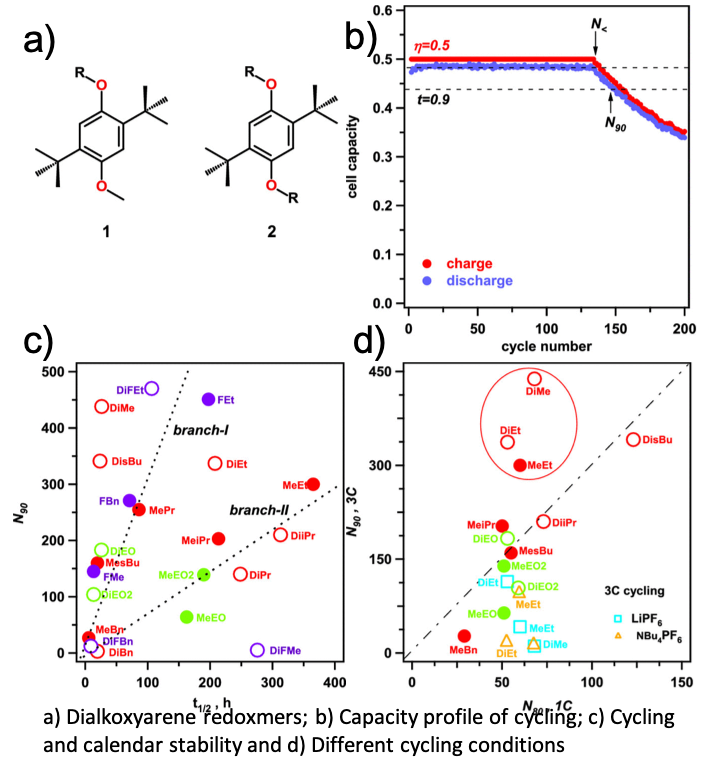
On Transferability of Performance Metrics for Redox-active Molecules
It is shown that two commonly used performance metrics for charged redoxmers in flow batteries, viz. their cycling and calendar lifetimes, generally do not correlate with each other. It is also demonstrated that the cycling stability is extremely sensitive to experimental detail; unless the conditions are tightly controlled, this metric is unsuitable as a guide for materials development. Read More
-
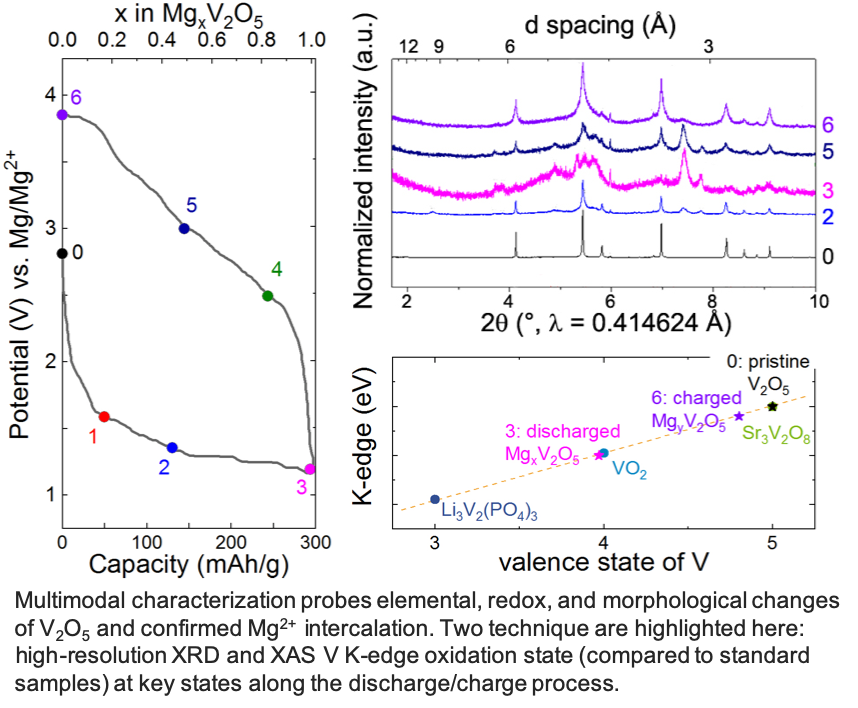
Intercalation of Magnesium into a Layered V2O5
Layered a-V2O5 reversibly intercalates 1 Mg2+ per unit formula to achieve 280 mAh/g at 110°C in a chemically and anodically stable ionic liquid electrolyte. Read More
-
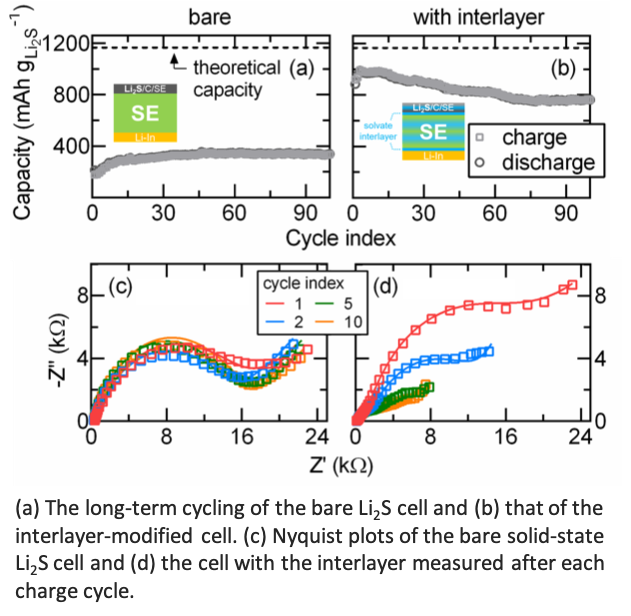
Incorporating Solvate and Solid Electrolytes for All-Solid-State Li2S Batteries with High Capacity and Long Cycle Life
Incorporating highly concentrated solvate electrolyte into all‐solid‐state Li2S batteries significantly improves the battery performance compared to its bare counterpart. The origin of the improved battery cyclability is the favorable interfacial contact, where the ion conducting network surrounding the active material remains intact even after the significant volume contraction of the Li2S cathode. Read More
-
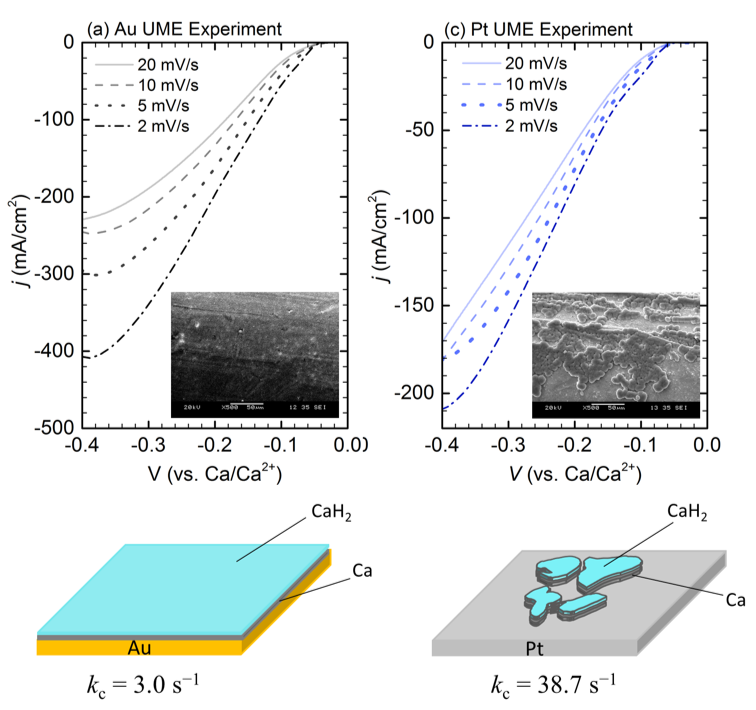
Understanding Ca Electrodeposition and Speciation Processes in Nonaqueous Electrolytes for Next-Generation Ca-Ion Batteries
Rechargeable Ca batteries have attracted little interest due to the extreme difficulty to perform reversible Ca electrodeposition. There is currently only one electrolyte, Ca(BH4)2 in THF, that exhibits reversible Ca deposition at room temperature. However, the underlying deposition mechanism and the interaction of Ca2+ ions with the surrounding molecules are still mostly unknown. Our research explores Ca deposition and speciation processes by using electrochemical and physical characterization techniques. Read More
Latest Updates
-
You’re Invited - JCESR and Beyond: Translating the Basic Science of Batteries
Please join us at Argonne National Laboratory on Tuesday, April 4, 2023 for JCESR and Beyond: Translating the Basic Science of Batteries. Registration is now open. This in-person event will celebrate 10 years of research from the Joint Center… Read More
-
A Message from JCESR: In Memory of George Crabtree
It is with heavy hearts that we say goodbye to George Crabtree, a Senior Scientist and Distinguished Fellow at Argonne National Laboratory, and Director of the Joint Center for Energy Storage Research (JCESR), who passed away unexpectedly on January 23. Dr. Read More
-
Cyanopyridines As Extremely Low-Reduction-Potential Anolytes for Nonaqueous Redox Flow Batteries
Discovery of a cyanophenylpyridine derivative with a very low reduction potential and good stability during cycling. Read More
-
Characterizing Redoxmer – Electrode Kinetics Using a SECM-Based Spot Analysis Method
Identified asymmetries in electron transfer (ET) kinetics between the reduction and oxidation of ferrocene-based redoxmers by measuring the ET rate constants (kf/kb) as a function of electrode potential. Read More
-
Benzotriazoles as Low Potential Anolytes for Non-Aqueous Redox Flow Batteries
We developed an easy-to-synthesize benzotriazole-based anolyte with a high energy redox potential (-2.3 V vs Fc/Fc+) and high solubility that demonstrates stable electrochemical cycling performance. Read More

