Research Highlights
-
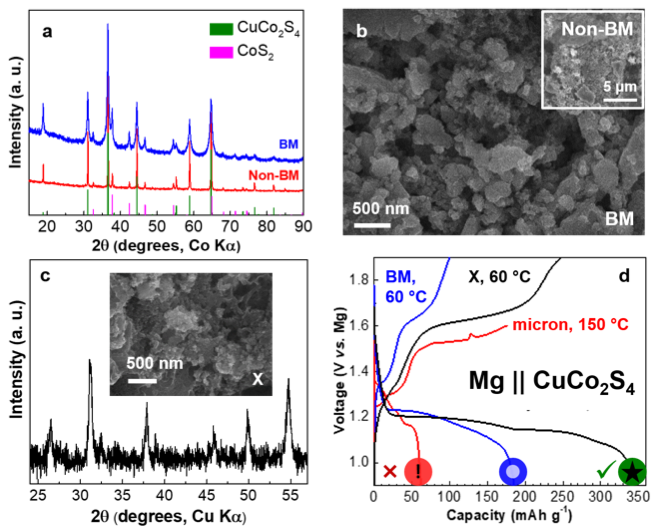
Direct Nano-Synthesis Methods Notably Benefit Mg-Battery Cathode Performance
A novel Mg cathode material – CuCo2S4 – was identified as a conversion material where direct nano-synthesis was required to provide the best electrochemical performance and deliver 350 mAh·g-1 at 60 °C, a capacity nearly double that of ball-milled material with similar dimensions. Read More
-
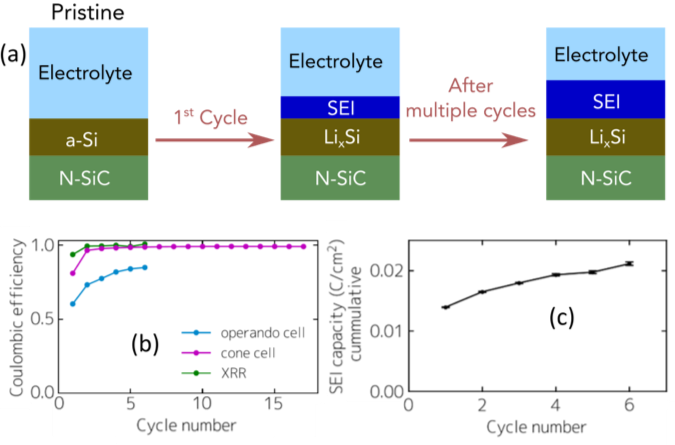
Quantifying Capacity Losses due to Solid Electrolyte Interphase Evolution
We quantified the capacity loss originating in solid electrolyte interphase (SEI) growth during each cycle and extracted the proportionality constant for SEI growth following a parabolic growth law. This continuous SEI growth contributes to the increasing overpotential, leading to capacity fading at a given constant current cycling rate. Read More
-
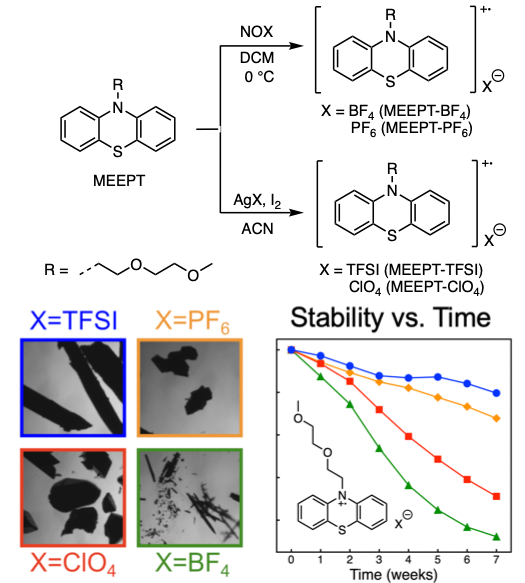
Quantifying Environmental Effects on the Solution and Solid-State Stability of a Phenothiazine Radical Cation
This study underscores the need to match the stability testing conditions for a phenothiazine electroactive derivative for RFB application. For analysis of stability, RRDE voltammetry can be used to rapidly screen molecules over seconds or minutes, providing stability trends that are similar to results that require several weeks to obtain using UV−vis spectroscopy. Read More
-
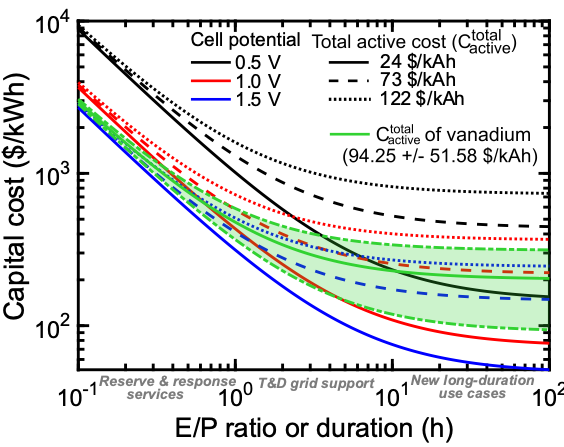
On Lifetime and Cost of Redox-Active Organics for Aqueous Flow Batteries
In this viewpoint, we recommend methodology for (1) testing aqueous organic flow batteries to better understand the fade mechanisms and failure modes, and for (2) techno-economic assessment of these batteries that incorporates the costs associated with electrolyte decay and replacement to articulate a feasible design space for the community to target. Read More
-
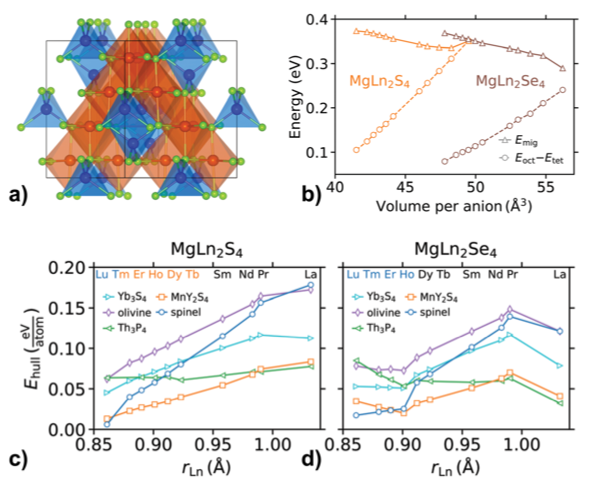
Computational investigation of chalcogenide spinel conductors for all-solid-state Mg batteries
(a) MgLn2X4 spinel structure with Mg (blue), lanthanoid Ln (orange), and chalcogenide X (light green). (b) Mg2+ migration energy, Emig, and the difference in octahedral and tetrahedral energies, Eoct - Etet. Stability with respect to competing phases, Ehull, for (c) each sulphide and (d) selenide. Read More
-
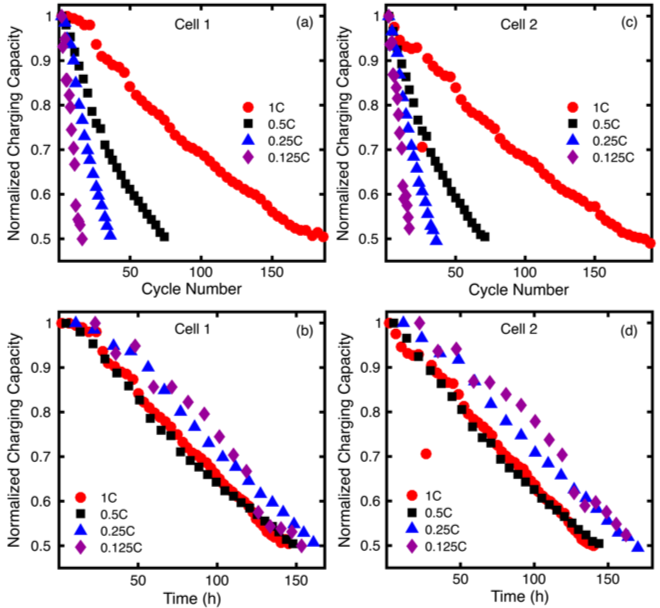
The impact of bulk electrolysis cycling conditions on the perceived stability of redox active materials
Under bulk electrolysis conditions, cycle time is a better measure of stability than cycle number and, for materials that are unstable in their charged state, the fractional capacity accessed is inversely related to cycle time until failure. Read More
-
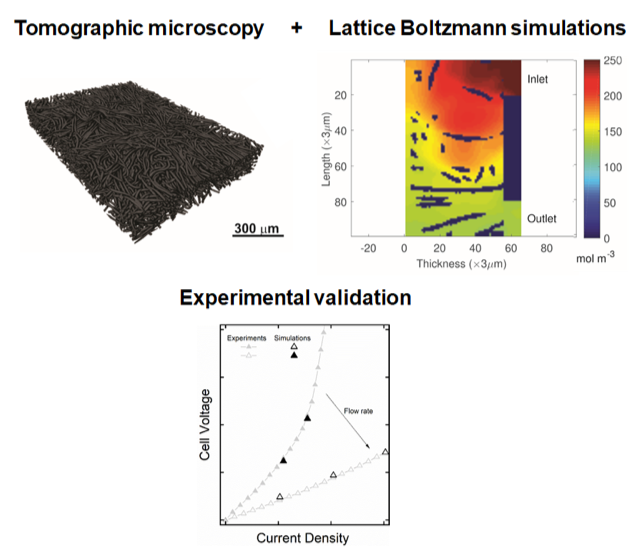
Experimentally validated 3D pore-scale lattice Boltzmann model for understanding porous electrode microstructure
Three dimensional (3D) multi-physics models of porous carbon electrodes are employed to understand the role of electrode microstructure on redox flow battery performance. Read More
-
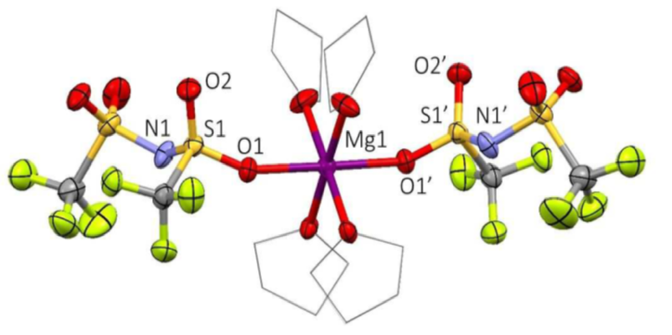
A Simple Halogen-free Magnesium Electrolyte for Reversible Magnesium Deposition through Co-solvent Assistance
Electrolytes for Mg-ion batteries are a complex mixture of salts, solvents, and additives that allow for the free-movement of Mg-ions across the cell. By addition of simple amines to the mixture, the internal equilibriums are shifted to allow formation of a simpler neutral complex in solution that does not require use of corrosive halide anions (Figure 1). Read More
-
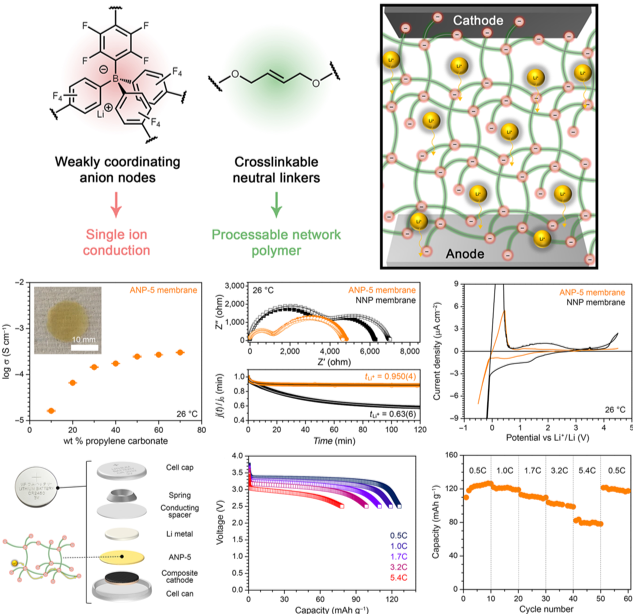
A Single-Ion Conducting Borate Network Polymer as a Viable Quasi-Solid Electrolyte for Lithium Metal Batteries
An anionic network polymer, ANP-5, membrane exhibits high room temperature ionic conductivity and selectivity for Li+ ion conduction, outstanding electrochemical stability, and flame retardancy. Read More
-
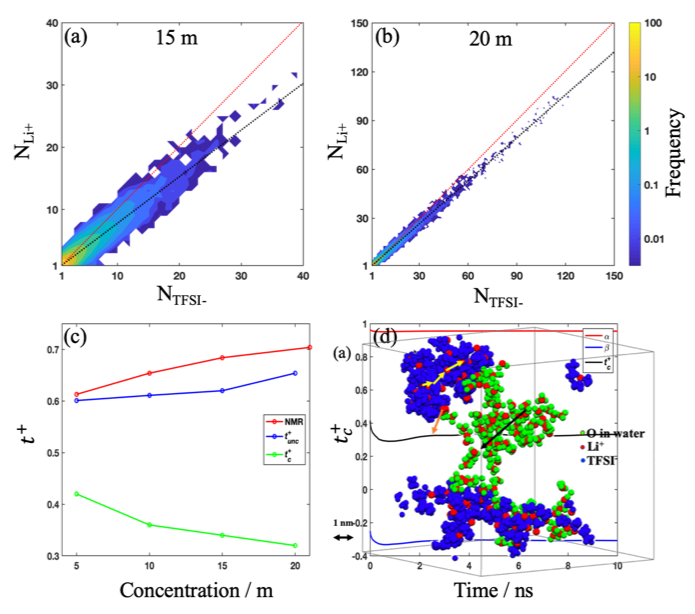
Asymmetric Composition of Ionic Aggregates and the Origin of High Correlated Transference Number in Water-in-Salt Electrolytes
MD simulations revealed the heterogeneous structure of ion and water domains/aggregates and fairly high correlated transference number in the water-in-salt electrolytes. Read More
Latest Updates
-
You’re Invited - JCESR and Beyond: Translating the Basic Science of Batteries
Please join us at Argonne National Laboratory on Tuesday, April 4, 2023 for JCESR and Beyond: Translating the Basic Science of Batteries. Registration is now open. This in-person event will celebrate 10 years of research from the Joint Center… Read More
-
A Message from JCESR: In Memory of George Crabtree
It is with heavy hearts that we say goodbye to George Crabtree, a Senior Scientist and Distinguished Fellow at Argonne National Laboratory, and Director of the Joint Center for Energy Storage Research (JCESR), who passed away unexpectedly on January 23. Dr. Read More
-
Cyanopyridines As Extremely Low-Reduction-Potential Anolytes for Nonaqueous Redox Flow Batteries
Discovery of a cyanophenylpyridine derivative with a very low reduction potential and good stability during cycling. Read More
-
Characterizing Redoxmer – Electrode Kinetics Using a SECM-Based Spot Analysis Method
Identified asymmetries in electron transfer (ET) kinetics between the reduction and oxidation of ferrocene-based redoxmers by measuring the ET rate constants (kf/kb) as a function of electrode potential. Read More
-
Benzotriazoles as Low Potential Anolytes for Non-Aqueous Redox Flow Batteries
We developed an easy-to-synthesize benzotriazole-based anolyte with a high energy redox potential (-2.3 V vs Fc/Fc+) and high solubility that demonstrates stable electrochemical cycling performance. Read More

