Research Highlights
-
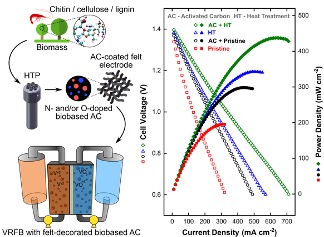
Exploration of Biomass-derived Activated Carbons for Use in Vanadium Redox Flow Batteries
Low-cost, sustainable, and elementally diverse biomass feedstocks are converted into activated carbons using a hydrothermal processing method. The suitability of the carbonaceous microparticles as catalysts for vanadium redox flow batteries is contemplated. Read More
-
Polymer Chain Dynamics in Block Copolymer Electrolytes
The effect of salt concentration on polymer chain dynamics was quantitatively determined in the range of = 0.1-100 ns. This study reports the first measurements of tube diameters in block copolymers (with or without salt). Read More
-
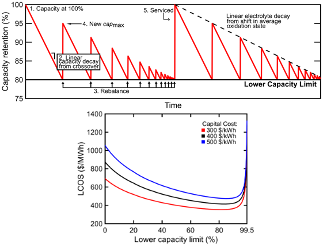
Assessing the levelized cost of vanadium redox flow batteries with capacity fade and rebalancing
Levelized cost is an important assessment metric that should be considered in addition to capital cost when determining the potential value of battery systems. Here, we contemplate the vanadium redox flow battery (VRFB) whose symmetric chemistry and open architecture enable periodic system maintenance (e.g. rebalancing) which, in turn, adds economic value. Read More
-
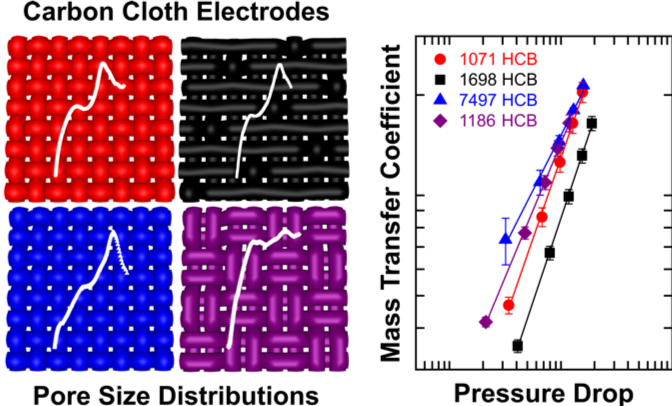
Comparing Physical and Electrochemical Properties of Different Weave Patterns for Carbon Cloth Electrodes in Redox Flow Batteries
In situ and ex situ characterization is combined with electrochemical analysis to elucidate mass transport and fluid dynamic relationships in carbon cloth electrodes with different weave patterns (plain, 8-satin harness, and 2×2 basket). Read More
-
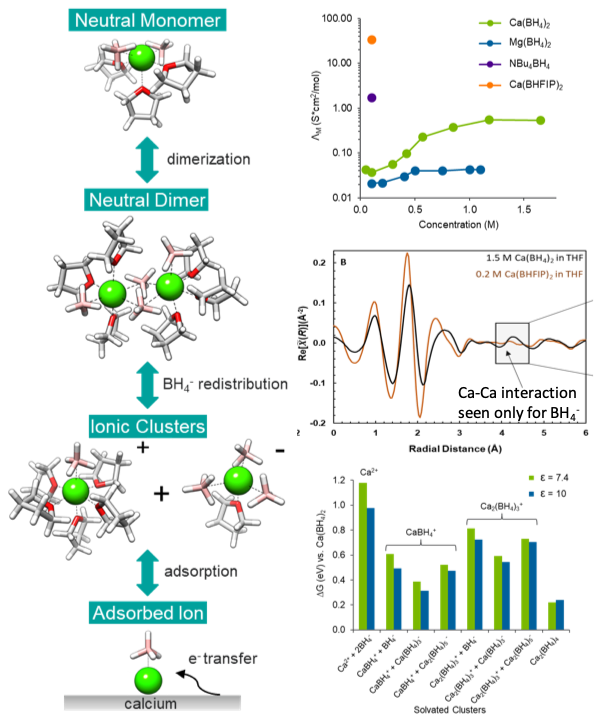
The critical role of configurational flexibility in facilitating reversible reactive metal deposition from borohydride solutions
Calcium borohydride in tetrahydrofuran is effective at reversible multivalent metal deposition where magnesium borohydride is not because the more polarizable Ca2+ cation enables greater coordination flexibility resulting in enhanced formation of CaBH4+ produced via multimer intermediates. Read More
-
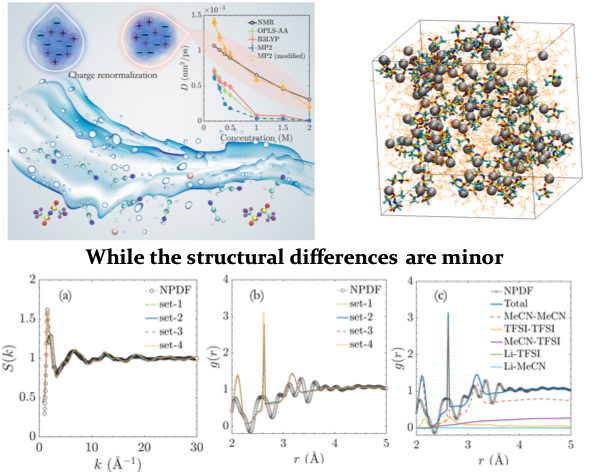
Realistic ion dynamics through charge renormalization in nonaqueous electrolytes
We demonstrate that a computationally inexpensive modification non-polarizable force fields: charge renormalization, can harmonize computed and experimentally determined structure and dynamic attributes of nonaqueous electrolytes. It is sufficient to reproduce dynamics over range of 0-2 M salt while not sacrificing quality of structural properties. Read More
-
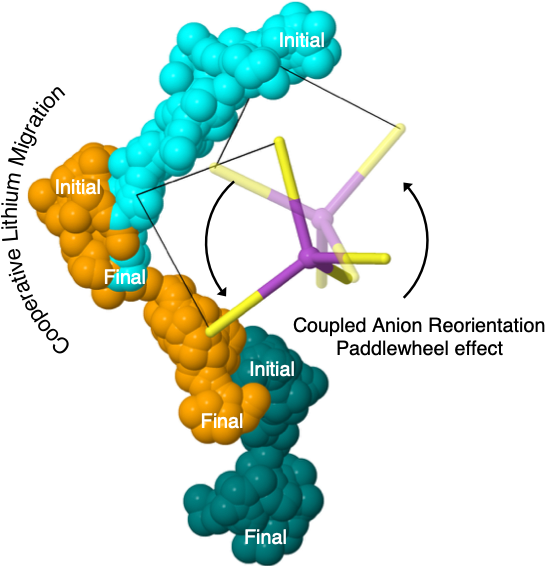
Low-temperature paddlewheel effect in glassy solid electrolytes
The high conductivity exhibited by glassy sulfide solid electrolytes is demonstrated to arise from the dynamic coupling of cation migration events with anion reorientations. Read More
-
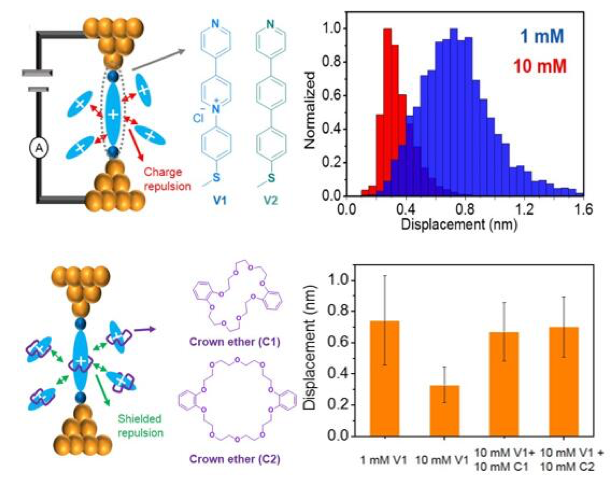
Direct Characterization of Intermolecular Interactions in Redox-active Pyridinium-based Molecules
Our results show that charge transport in redox-active pyridinium-based molecules can be modulated by host-guest interactions and intermolecular electrostatic effects. Read More
-
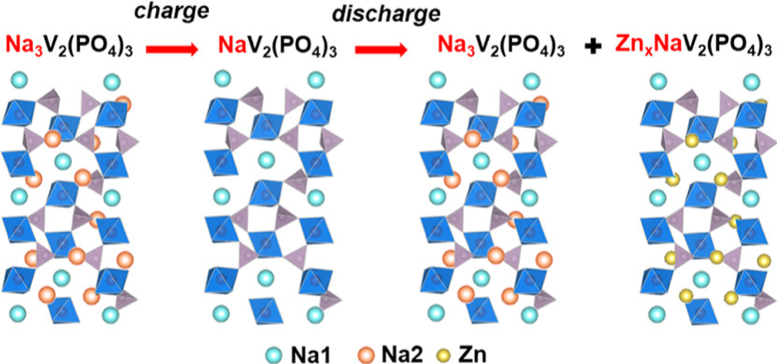
Understanding Zn2+ Charge-Storage Mechanism in Vanadium-based Na+ Superionic Conductor for Zn Batteries
Elucidated the complex, multivalent charge-storage behavior of Na3V2(PO4)3 with Na+ and Zn2+ insertion. Read More
-
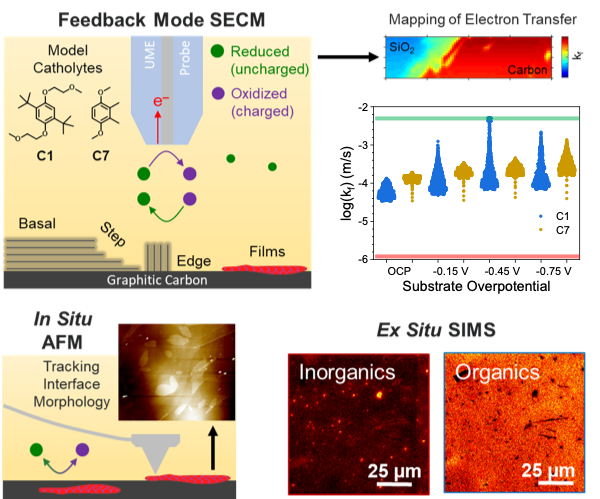
Combining SECM and AFM to Probe the Redoxmer-Electrode Interface
Dialkoxybenzene catholytes form electron transfer-limiting thin films preferentially on the basal plane of graphitic electrodes Read More
Latest Updates
-
You’re Invited - JCESR and Beyond: Translating the Basic Science of Batteries
Please join us at Argonne National Laboratory on Tuesday, April 4, 2023 for JCESR and Beyond: Translating the Basic Science of Batteries. Registration is now open. This in-person event will celebrate 10 years of research from the Joint Center… Read More
-
A Message from JCESR: In Memory of George Crabtree
It is with heavy hearts that we say goodbye to George Crabtree, a Senior Scientist and Distinguished Fellow at Argonne National Laboratory, and Director of the Joint Center for Energy Storage Research (JCESR), who passed away unexpectedly on January 23. Dr. Read More
-
Cyanopyridines As Extremely Low-Reduction-Potential Anolytes for Nonaqueous Redox Flow Batteries
Discovery of a cyanophenylpyridine derivative with a very low reduction potential and good stability during cycling. Read More
-
Characterizing Redoxmer – Electrode Kinetics Using a SECM-Based Spot Analysis Method
Identified asymmetries in electron transfer (ET) kinetics between the reduction and oxidation of ferrocene-based redoxmers by measuring the ET rate constants (kf/kb) as a function of electrode potential. Read More
-
Benzotriazoles as Low Potential Anolytes for Non-Aqueous Redox Flow Batteries
We developed an easy-to-synthesize benzotriazole-based anolyte with a high energy redox potential (-2.3 V vs Fc/Fc+) and high solubility that demonstrates stable electrochemical cycling performance. Read More

